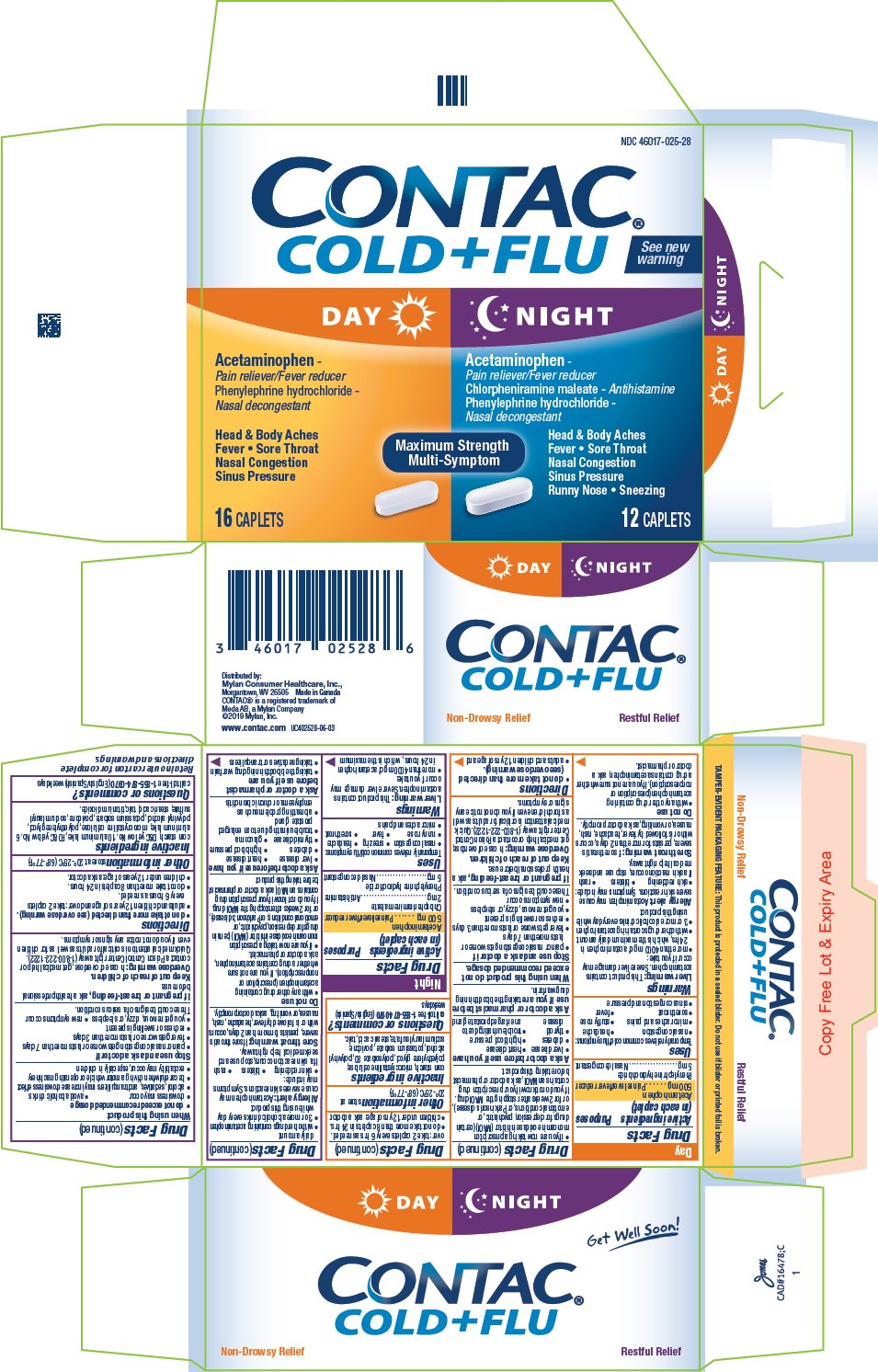
CONTAC COLD AND FLU DAY NIGHT- acetaminophen and phenylephrine hydrochloride/ acetaminophen, chlorpheniramine maleate, and phenylephrine hydrochloride kit
Contac Cold and Flu by
Drug Labeling and Warnings
Contac Cold and Flu by is a Otc medication manufactured, distributed, or labeled by Mylan Consumer Healthcare, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Day
- Active ingredients (in each caplet)
- Purposes
- Uses
-
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take:
- more than 4000 mg of acetaminophen in 24 hrs, which is the maximum daily amount
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Sore throat warning: If sore throat is severe, persists for more than 2 days, occurs with or is followed by fever, headache, rash, nausea, or vomiting, ask a doctor promptly.
- DO NOT USE
-
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
- ASK DOCTOR
- Ask a doctor before use if you have
- ASK DOCTOR/PHARMACIST
- Ask a doctor or pharmacist before use if you are
- WHEN USING
- When using this product do not exceed recommended dosage.
- STOP USE
- Stop use and ask a doctor if
- PREGNANCY OR BREAST FEEDING
- Keep out of reach of children.
- Overdose warning:
-
Directions
- do not take more than directed (see overdose warning).
- adults and children 12 yrs of age and over: take 2 caplets every 6 hours as needed.
- do not take more than 8 caplets in 24 hrs.
- children under 12 yrs of age: ask a doctor
- Other information
- INACTIVE INGREDIENT
- Inactive ingredients
- Questions or comments?
- Night
- Active ingredients (in each caplet)
- Purposes
- Uses
-
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take:
- more than 4000 mg of acetaminophen in 24 hours, which is the maximum daily amount
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Sore throat warning: If sore throat is severe, persists for more than 2 days, occurs with or is followed by fever, headache, rash, nausea, or vomiting, ask a doctor promptly.
-
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
- Ask a doctor before use if you have
- Ask a doctor or pharmacist before use if you are
- When using this product
-
Stop use and ask a doctor if
- pain or nasal congestion gets worse or lasts more than 7 days
- fever gets worse or lasts more than 3 days
- redness or swelling is present
- you get nervous, dizzy, or sleepless
- new symptoms occur
These could be signs of a serious condition.
If pregnant or breast-feeding, ask a health professional before use.
- Keep out of reach of children.
- Overdose warning:
-
Directions
- do not take more than directed (see overdose warning).
- adults and children 12 years of age and over: take 2 caplets every 6 hours as needed.
- do not take more than 8 caplets in 24 hrs.
- children under 12 years of age: ask a doctor
- Other information
- Inactive ingredients
- Questions or comments?
-
PRINCIPAL DISPLAY PANEL
Display Panel CONTAC Cold+Flu Day Night Caplets: 28 count Package
NDC: 46017-025-28
CONTAC®
COLD + FLU See new warningDAY
Acetaminophen -
Pain reliever/Fever reducer
Phenylephrine hydrochloride -
Nasal decongestantHead & Body Aches
Fever Sore Throat
Nasal Congestion
Sinus Pressure16 CAPLETS
NIGHT
Acetaminophen -
Pain reliever/Fever reducer
Chlorpheniramine maleate – Antihistamine
Phenylephrine hydrochloride -
Nasal decongestantHead & Body Aches
Fever Sore Throat
Nasal Congestion
Sinus Pressure
Runny Nose Sneezing12 CAPLETS
Distributed by:
Mylan Consumer Healthcare, Inc.,
Morgantown, WV 26505 Made in Canada
CONTAC® is a registered trademark of Meda AB, a Mylan Company
© 2019 Mylan, Inc.
www.contac.com UC402528-06-03 -
INGREDIENTS AND APPEARANCE
CONTAC COLD AND FLU DAY NIGHT
acetaminophen and phenylephrine hydrochloride/ acetaminophen, chlorpheniramine maleate, and phenylephrine hydrochloride kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 46017-025 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 46017-025-28 1 in 1 CARTON; Type 0: Not a Combination Product 07/01/2014 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 16 Part 2 12 Part 1 of 2 CONTAC COLD AND FLU DAY
acetaminophen and phenylephrine hydrochloride tabletProduct Information Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 500 mg PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE HYDROCHLORIDE 5 mg Inactive Ingredients Ingredient Name Strength STARCH, CORN (UNII: O8232NY3SJ) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYSORBATE 80 (UNII: 6OZP39ZG8H) POLYVINYL ALCOHOL, UNSPECIFIED (UNII: 532B59J990) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) SODIUM LAURYL SULFATE (UNII: 368GB5141J) STEARIC ACID (UNII: 4ELV7Z65AP) TALC (UNII: 7SEV7J4R1U) Product Characteristics Color WHITE Score no score Shape CAPSULE Size 21mm Flavor Imprint Code sun;symbol Contains Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 07/01/2014 Part 2 of 2 CONTAC COLD AND FLU NIGHT
acetaminophen, chlorpheniramine maleate, and phenylephrine hydrochloride tabletProduct Information Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 500 mg CHLORPHENIRAMINE MALEATE (UNII: V1Q0O9OJ9Z) (CHLORPHENIRAMINE - UNII:3U6IO1965U) CHLORPHENIRAMINE MALEATE 2 mg PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE HYDROCHLORIDE 5 mg Inactive Ingredients Ingredient Name Strength STARCH, CORN (UNII: O8232NY3SJ) ALUMINUM OXIDE (UNII: LMI26O6933) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYVINYL ALCOHOL, UNSPECIFIED (UNII: 532B59J990) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) SODIUM LAURYL SULFATE (UNII: 368GB5141J) STEARIC ACID (UNII: 4ELV7Z65AP) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color YELLOW Score no score Shape CAPSULE Size 21mm Flavor Imprint Code moon;symbol Contains Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 07/01/2014 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 07/01/2014 Labeler - Mylan Consumer Healthcare, Inc. (831410522)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.