IDVYNSO- doravirine, islatravir tablet, film coated
Drug Labeling and Warnings
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use IDVYNSO safely and effectively. See full prescribing information for IDVYNSO.
IDVYNSO™ (doravirine and islatravir) tablets, for oral use
Initial U.S. Approval: 2026INDICATIONS AND USAGE
IDVYNSO is a two-drug combination of doravirine, a HIV-1 non-nucleoside reverse transcriptase inhibitor (NNRTI), and islatravir, a nucleoside analog reverse transcriptase inhibitor (NRTI), and is indicated as a complete regimen for the treatment of HIV-1 infection in adults to replace the current antiretroviral regimen in those who are virologically-suppressed (HIV-1 RNA less than 50 copies per mL) on a stable antiretroviral regimen with no history of virologic treatment failure and no known substitutions associated with resistance to doravirine. (1)
DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHS
Tablets: 100 mg doravirine and 0.25 mg islatravir. (3)
CONTRAINDICATIONS
- IDVYNSO is contraindicated when co-administered with drugs that are strong cytochrome P450 (CYP)3A enzyme inducers as significant decreases in doravirine plasma concentrations may occur, which may decrease the effectiveness of IDVYNSO. (4)
- IDVYNSO is contraindicated when co-administered with lamivudine (3TC) or emtricitabine (FTC), which are deoxycytidine kinase (dCK) substrates, as a decrease in islatravir-triphosphate (ISL-TP) levels may occur, which may decrease the effectiveness of IDVYNSO. (4)
WARNINGS AND PRECAUTIONS
Severe skin reactions, including Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN), and Drug Rash with Eosinophilia and Systemic Symptoms (DRESS), have been reported. Discontinue IDVYNSO immediately if signs or symptoms of severe skin reactions develop. (5.1)
ADVERSE REACTIONS
Most common adverse reactions (incidence greater than or equal to 2%, all grades): diarrhea, dizziness, fatigue, abdominal distension, headache, and weight increased. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Merck Sharp & Dohme LLC at 1-877-888-4231 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 4/2026
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage
2.2 Dosage Adjustment with Rifabutin
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Skin and Hypersensitivity Reactions
5.2 Risk of Adverse Reactions or Loss of Virologic Response Due to Drug Interactions
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Concomitant Use with Other Antiretroviral Medications
7.2 Effects of Other Drugs on IDVYNSO
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
8.7 Hepatic Impairment
8.8 HBV Co-Infection
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
12.4 Microbiology
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
IDVYNSO™ is indicated as a complete two-drug regimen for the treatment of human immunodeficiency virus type 1 (HIV-1) infection in adults to replace the current antiretroviral regimen in those who are virologically-suppressed (HIV-1 RNA less than 50 copies/mL) on a stable antiretroviral regimen with no history of virologic treatment failure and no known substitutions associated with resistance to doravirine [see Microbiology (12.4) and Clinical Studies (14)].
-
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage
The recommended dosage of IDVYNSO is one tablet taken orally once daily with or without food [see Clinical Pharmacology (12.3)]. One tablet of IDVYNSO contains 100 mg doravirine and 0.25 mg islatravir.
2.2 Dosage Adjustment with Rifabutin
If IDVYNSO is co-administered with rifabutin, take one tablet of IDVYNSO once daily as recommended, followed by one tablet of doravirine (PIFELTRO) 100 mg approximately 12 hours after the dose of IDVYNSO for the duration of rifabutin co-administration [see Drug Interactions (7.2) and Clinical Pharmacology (12.3)].
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
IDVYNSO is contraindicated when co-administered with:
- drugs that are strong cytochrome P450 (CYP)3A enzyme inducers as significant decreases in doravirine plasma concentrations may occur, which may decrease the effectiveness of IDVYNSO [see Warnings and Precautions (5.2), Drug Interactions (7.2), and Clinical Pharmacology (12.3)].
- lamivudine (3TC) or emtricitabine (FTC) as significant decreases in islatravir-triphosphate (ISL-TP) concentrations may occur, which may decrease the effectiveness of IDVYNSO [see Warnings and Precautions (5.2), Drug Interactions (7.2) and Clinical Pharmacology (12.3)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Skin and Hypersensitivity Reactions
Severe skin reactions, including Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN), have been reported during postmarketing experience with doravirine-containing regimens [see Adverse Reactions (6.2)]. In addition, Drug Rash with Eosinophilia and Systemic Symptoms (DRESS syndrome) was reported with IDVYNSO in a clinical trial [see Adverse Reactions (6.1)]. Discontinue IDVYNSO, and other medications known to be associated with severe skin reactions, immediately if a painful rash with mucosal involvement, a progressive severe rash, or a rash with constitutional symptoms, eosinophilia, lymphadenopathy, or other organ involvement develops. Clinical status should be closely monitored, and appropriate therapy should be initiated.
5.2 Risk of Adverse Reactions or Loss of Virologic Response Due to Drug Interactions
The concomitant use of IDVYNSO and certain other drugs may result in known or potentially significant drug interactions, some of which may lead to [see Dosage and Administration (2.2), Contraindications (4), Drug Interactions (7.2), and Clinical Pharmacology (12.3)]:
- Loss of therapeutic effect of IDVYNSO and possible development of resistance.
- Possible clinically significant adverse reactions from greater exposures of a component of IDVYNSO.
See Table 3 for steps to prevent or manage these possible and known significant drug interactions, including dosing recommendations. Consider the potential for drug interactions prior to and during IDVYNSO therapy, review concomitant medications during IDVYNSO therapy, and monitor for adverse reactions.
-
6 ADVERSE REACTIONS
The following serious adverse reactions are described in greater detail in other sections of the labeling:
- Skin and Hypersensitivity Reactions [see Warnings and Precautions (5.1)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Adverse Reactions in Virologically-Suppressed Adults Living with HIV-1 Who Switched to IDVYNSO
The safety assessment of IDVYNSO in virologically-suppressed (HIV-1 RNA less than 50 copies/mL) participants living with HIV was based on Week 48 data from two Phase 3, randomized trials, Trial 051 and Trial 052. A total of 708 participants received once-daily IDVYNSO [see Clinical Studies (14)].
In Trial 051, an open-label trial with 551 participants, 366 participants were switched to IDVYNSO and 185 participants continued their baseline antiretroviral therapy (ART). By Week 48, 0.5% in the IDVYNSO group and 2% in the baseline ART group had adverse events leading to discontinuation of study medication.
In Trial 052, a double-blinded trial with 513 participants, 342 participants were switched to IDVYNSO and 171 participants continued on bictegravir/emtricitabine/tenofovir alafenamide (BIC/FTC/TAF). By Week 48, 3% in the IDVYNSO group and 2% in the BIC/FTC/TAF group had adverse events leading to discontinuation of study medication.
Among participants who received IDVYNSO and experienced at least one adverse event in Trial 051 or Trial 052, 88% experienced only adverse events that were mild (Grade 1) or moderate (Grade 2).
The most common adverse reactions (all grades) reported in greater than or equal to 2% of participants in any treatment group from Trial 051 and Trial 052 through Week 48 are presented in Table 1.
Table 1: Adverse Reactions* (All Grades) Reported in ≥2% of Participants in Any Treatment Group in Trials 051 and 052 in HIV-1 Virologically-Suppressed Adults (Week 48) Adverse Reactions Trial 051 Trial 052 IDVYNSO
N=366Baseline ART
N=185IDVYNSO
N=342BIC/FTC/TAF
N=171- * Frequencies based on all adverse events attributed to study drugs by the investigator
- † Fatigue includes fatigue and asthenia.
- ‡ The mean change in weight from baseline at Week 48 was 0.94 kg in the IDVYNSO group vs. -0.15 kg in the baseline ART group in Trial 051, and -0.03 kg in the IDVYNSO group vs. 0.28 kg in the BIC/FTC/TAF group in Trial 052.
- § 4 of the 6 participants with adverse reactions of weight increased switched from a baseline ART regimen containing efavirenz and/or tenofovir disoproxil fumarate in Trial 051.
Diarrhea 3% 0 1% 1% Dizziness 2% 1% 1% 0 Fatigue† 2% 1% 1% 1% Abdominal distension 2% 0 1% 0 Headache 2% 1% 1% 0 Weight increased‡ 2%§ 0 <1% 0 A single case of severe immune thrombocytopenia (platelet count nadir of 2 x109/L) characterized by abrupt onset of subcutaneous hematoma, petechiae, and hematuria was reported in a participant 32 days after initiating IDVYNSO in Trial 052. This serious adverse reaction resolved with discontinuation of IDVYNSO, in conjunction with treatments including corticosteroids and IVIG. Among all participants in Trial 051 and Trial 052, there were no patterns of platelet decreases over time with IDVYNSO and no differences between treatment arms in mean change from baseline in platelet count.
Less Common Adverse Reactions
The following select adverse reactions were observed in less than 2% of participants administered IDVYNSO:
- Gastrointestinal disorders: Abdominal pain (includes abdominal pain, abdominal pain upper, abdominal pain lower, and abdominal discomfort), flatulence, nausea
- Psychiatric disorders: Abnormal dreams, Insomnia (includes insomnia, initial insomnia, middle insomnia and terminal insomnia)
- Skin and Subcutaneous Tissue Disorders: Pruritus, Rash (includes rash, rash papular, rash maculo-papular and rash pruritic)
In an ongoing clinical trial, a participant developed Grade 4 DRESS syndrome ten weeks after starting IDVYNSO that was characterized by a diffuse pruritic maculopapular rash covering 60% of the body, lymphadenopathy, eosinophilia (peak 20,000 cells/µL), and an episode of hypotension. This serious adverse reaction resolved with discontinuation of IDVYNSO [see Warnings and Precautions (5.1)].
Laboratory Abnormalities
Hepatic laboratory data from Trials 051 and Trial 052 are presented in Table 2. The hepatic laboratory abnormalities were generally asymptomatic and resolved without discontinuation of IDVYNSO.
Table 2: Hepatic Laboratory Abnormalities (Grades 2-4) Worsened from Baseline Reported in Adults Receiving IDVYNSO in Trial 051 and Trial 052 Laboratory Parameter Trial 051 Trial 052 IDVYNSO
N=366Baseline ART
N=185IDVYNSO
N=340BIC/FTC/TAF
N=171Alanine Aminotransferase Grade 2: 2.5 - <5.0 x ULN 2% 1% 1% 1% Grade 3: 5.0 - <10.0 x ULN <1% 0 0 0 Grade 4: ≥10.0 x ULN 0 1% 1% 0 Aspartate Aminotransferase Grade 2: 2.5 - <5.0 x ULN 2% 2% 2% 1% Grade 3: 5.0 - <10.0 x ULN 0 0 <1% 0 Grade 4: ≥10.0 x ULN 0 1% 1% 1% Total Bilirubin Grade 2: 1.6 - <2.6 x ULN 1% 1% 1% 1% Grade 3: 2.6 - <5.0 x ULN <1% 0 1% 0 Grade 4: ≥5.0 x ULN 0 0 0 0 Across the clinical development program for the combination of doravirine and islatravir, there were 3 participants with elevations in alanine aminotransferase and/or aspartate aminotransferase greater than 10X ULN attributed to study drug that resolved with discontinuation of treatment. Of the 3 participants, one received doravirine 100 mg with islatravir 0.75 mg (three times the recommended dosage of islatravir) and had transient elevation in total bilirubin 2X ULN and Grade 1 (mild) nausea. The other 2 participants received IDVYNSO and were asymptomatic.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postmarketing experience in patients receiving doravirine-containing regimens. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Doravirine:
Hepatobiliary Disorders: hepatitis
Investigations: hepatic enzyme increased
Skin and Subcutaneous Tissue Disorders: Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN)
-
7 DRUG INTERACTIONS
7.1 Concomitant Use with Other Antiretroviral Medications
Because IDVYNSO is a complete regimen for the treatment of HIV-1 infection, co-administration with other antiretroviral medications for treatment of HIV-1 infection is not recommended.
7.2 Effects of Other Drugs on IDVYNSO
Doravirine
Co-administration of IDVYNSO with a CYP3A inducer decreases doravirine plasma concentrations, which may reduce the efficacy of IDVYNSO [see Contraindications (4), Warnings and Precautions (5.2), and Clinical Pharmacology (12.3)].
Co-administration of IDVYNSO and drugs that are inhibitors of CYP3A may result in increased plasma concentrations of doravirine.
Islatravir
Islatravir requires phosphorylation by cellular kinases to form the pharmacologically active ISL-TP. Co-administration of IDVYNSO and drugs that are substrates of dCK (e.g., certain nucleoside antiviral agents and nucleoside antimetabolites) may result in a decrease in ISL-TP concentrations and may reduce the therapeutic effect of islatravir [see Contraindications (4), Warnings and Precautions (5.2), and Clinical Pharmacology (12.3)].
Islatravir is subject to ADA-mediated metabolism (approximately 53%). Co-administration of IDVYNSO and drugs that are ADA inhibitors (e.g., pentostatin) may result in increased plasma concentrations of islatravir and may increase the risk of adverse reactions [see Warnings and Precautions (5.2) and Clinical Pharmacology (12.3)].
The drug interactions described in Table 3 are based on studies conducted with IDVYNSO, its components, or are predicted drug interactions that may occur with IDVYNSO.
Table 3: Drug Interactions with IDVYNSO* Concomitant Drug Class: Drug Name Effect on Concentration Clinical Comment ↑ = increase, ↓ = decrease - * This table is not all inclusive.
- † Interactions were evaluated in clinical studies. All other drug-drug interactions are predicted.
- ‡ May include other nucleoside drugs
Strong CYP3A Inducers ↓ doravirine Co-administration with strong CYP3A inducers is contraindicated
At least a 4-week cessation period is recommended prior to initiation of IDVYNSO.Moderate CYP3A Inducers ↓ doravirine If IDVYNSO is co-administered with rifabutin†, one tablet of doravirine (PIFELTRO) should be taken approximately 12 hours after the dose of IDVYNSO [see Dosage and Administration (2.2)].
Co-administration with other moderate CYP3A inducers is not recommended.dCK Substrates‡ Anti-virals
lamivudine†
emtricitabine
↓ ISL-TP
Co-administration is contraindicated with lamivudine or emtricitabine.Nucleoside Antimetabolites
cladribine
clofarabine
cytarabine
fludarabine
gemcitabine
↓ ISL-TP
Co-administration with these nucleoside antimetabolites is not recommended as dCK substrates may cause a decrease in the intracellular concentration of ISL-TP.ADA Inhibitors pentostatin ↑ islatravir Co-administration with pentostatin is not recommended as it may cause an increase in plasma concentrations of islatravir. -
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors pregnancy outcomes in individuals exposed to IDVYNSO during pregnancy. Healthcare providers are encouraged to register patients by calling the Antiretroviral Pregnancy Registry (APR) at 1-800-258-4263.
Risk Summary
There are insufficient human data on the use of IDVYNSO during pregnancy to inform a drug-associated risk of birth defects and miscarriage. In animal reproduction studies, no developmental effects were observed when the components of IDVYNSO were administered separately at exposures (AUC) at least 8 (doravirine) and 500 (islatravir) times the exposure at the recommended human dose (RHD) of these components in IDVYNSO (see Data.)
The background rate of major birth defects is 2.7% in a U.S. reference population of the Metropolitan Atlanta Congenital Defects Program (MACDP). The rate of miscarriage is not reported in the APR. The estimated background rate of miscarriage in the clinically recognized pregnancies in the U.S. general population is 15-20%. Methodological limitations of the APR include the use of MACDP as the external comparator group. The MACDP population is not disease-specific, evaluates individuals and infants from the limited geographic area, and does not include outcomes for births that occurred at less than 20 weeks gestation.
Animal Data
Doravirine
Doravirine was administered orally to pregnant rabbits (up to 300 mg/kg/day on Gestation Days (GD) 7 to 20) and rats (up to 450 mg/kg/day on GD 6 to 20 and separately from GD 6 to Lactation/Postpartum Day 20). No significant toxicological effects on embryo-fetal (rats and rabbits) or pre/post-natal (rats) development were observed at exposures (AUC) approximately 9 times (rats) and 8 times (rabbits) the exposure in humans at the RHD. Doravirine was transferred to the fetus through the placenta in embryo-fetal studies, with fetal plasma concentrations of up to 40% (rabbits) and 52% (rats) that of maternal concentrations observed on GD 20.
Islatravir
Islatravir was administered orally to pregnant rabbits (up to 10 mg/kg/day on GD 7 to 20) and rats (up to 50 mg/kg/day on GD 6 to 20 and separately up to 10 mg/kg from GD 6 to Lactation/Postpartum Day 20). No significant toxicological effects on embryo-fetal (rats and rabbits) or pre/post-natal (rats) development were observed at exposures (AUC) approximately 500 times (rats) and 1300 times (rabbits) the exposure in humans at the RHD. In rats, islatravir was transferred to the fetus through the placenta in rats, with fetal plasma concentrations of up to 88% that of maternal concentrations observed on GD 20.
8.2 Lactation
Risk Summary
It is unknown whether IDVYNSO or any of the components of IDVYNSO is present in human milk, affects human milk production, or has effects on the breastfed infant. Doravirine is present in the milk of lactating rats, while islatravir was detected in the plasma of nursing pups from lactating rats administered islatravir (see Data). Potential risks of breastfeeding include (1) HIV-1 transmission (in infants without HIV-1), (2) developing viral resistance (in infants with HIV-1), and (3) serious adverse reactions in a breastfed infant similar to those seen in adults.
Doravirine: Doravirine was excreted into the milk of lactating rats following oral administration (450 mg/kg/day) from GD 6 to Lactation Day (LD) 14, with milk concentrations approximately 1.5 times that of maternal plasma concentrations observed 2 hours post dose on LD 14.
Islatravir: Islatravir was detected in the plasma of nursing pups (LD 10) from lactating rats following oral administration (10 mg/kg) from GD 6 to LD 10, with concentrations 0.1% and 1.5% the maternal plasma concentrations observed 1 and 3 hours post dose on LD 10, respectively [see Nonclinical Toxicology (13.1)].
8.4 Pediatric Use
The safety and effectiveness of IDVYNSO have not been established in pediatric patients less than 18 years of age [see Clinical Pharmacology (12.3)].
8.5 Geriatric Use
Clinical trials in virologically-suppressed participants who received IDVYNSO (Trial 051 and Trial 052) included 81 (11%) participants aged 65 years and older, including 10 (1%) aged 75 years and older [see Clinical Studies (14)]. No overall differences in safety or effectiveness were observed between these participants and younger participants, but greater sensitivity of some older individuals cannot be ruled out.
8.6 Renal Impairment
No dosage adjustment of IDVYNSO is required in patients with eGFR ≥30 mL/min/1.73 m2. IDVYNSO is not recommended in patients with eGFR <30 mL/min/1.73 m2. IDVYNSO has not been studied in participants undergoing dialysis [see Clinical Pharmacology (12.3)].
8.7 Hepatic Impairment
No dosage adjustment of IDVYNSO is recommended in patients with mild or moderate hepatic impairment (Child-Pugh Class A or B). IDVYNSO has not been studied in patients with severe hepatic impairment (Child-Pugh Class C) and therefore is not recommended in these patients [see Clinical Pharmacology (12.3)].
8.8 HBV Co-Infection
IDVYNSO does not have activity against hepatitis B virus (HBV). Patients with HBV coinfection who switch to IDVYNSO from an antiretroviral regimen with activity against HBV, and patients on IDVYNSO who are newly diagnosed with HBV coinfection, should be closely monitored and specific anti-HBV therapy should be considered, as clinically appropriate.
- 10 OVERDOSAGE
-
11 DESCRIPTION
IDVYNSO is a fixed-dose combination tablet for oral administration containing doravirine, an HIV-1 non-nucleoside reverse transcriptase inhibitor (NNRTI), and islatravir, an HIV-1 nucleoside analog reverse transcriptase inhibitor (NRTI).
Each film-coated tablet contains 100 mg of doravirine and 0.25 mg islatravir as active ingredients. The tablets include the following inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, hypromellose acetate succinate, lactose monohydrate, magnesium stearate, and microcrystalline cellulose. The tablets are film coated with a coating material containing the following inactive ingredients: calcium carbonate, ferric oxide, ferrosoferric oxide, hypromellose, lactose monohydrate, and triacetin. The coated tablets are polished with carnauba wax.
Doravirine
The chemical name for doravirine is 3-chloro-5-[[1-[(4,5-dihydro-4-methyl-5-oxo-1H-1,2,4-triazol-3-yl)methyl]-1,2-dihydro-2-oxo-4-(trifluoromethyl)-3-pyridinyl]oxy]benzonitrile. The molecular formula is C17H11ClF3N5O3 and the molecular weight is 425.75. Doravirine is practically insoluble in water and has the following structural formula:
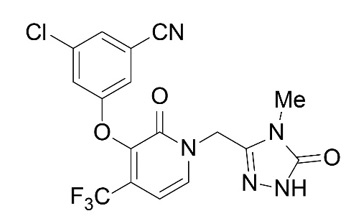
Islatravir
Islatravir is a crystalline monohydrate. The chemical name for islatravir is 2′-deoxy-4′-C-ethynyl-2-fluoroadenosine hydrate (1:1). The molecular formula is C12H12FN5O3.H2O and the molecular weight is 311.27. Islatravir monohydrate is very slightly soluble in water and has the following structural formula:
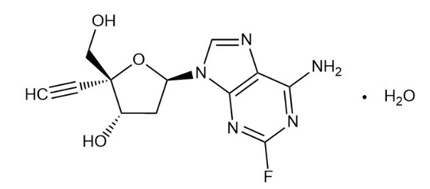
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
IDVYNSO is a fixed-dose combination of the antiretroviral drugs doravirine and islatravir [see Microbiology (12.4)].
12.2 Pharmacodynamics
In a Phase 2 trial evaluating doravirine over a dose range of 0.25 to 2 times the recommended dose of doravirine (in combination with FTC and TDF), in participants with no antiretroviral treatment history, no exposure-response relationship for efficacy was identified for doravirine.
In a Phase 2 trial evaluating islatravir over a dose range of 1 to 9 times the recommended dose of islatravir (in combination with doravirine and 3TC), in participants with no antiretroviral treatment history, no exposure-response relationship for efficacy was identified for islatravir.
Exposure-response modeling of data from Phase 2 and Phase 3 studies, including studies evaluating islatravir at daily doses from 0.25 mg to 2.25 mg (9 times the recommended daily dosage), demonstrated a relationship between islatravir exposure and decreases in lymphocytes and CD4+ T-cells. No clinically meaningful reduction in lymphocytes or CD4+ T-cells was observed at exposures associated with the recommended daily islatravir dose of 0.25 mg.
Cardiac Electrophysiology
At 12 times the maximum recommended doravirine dose, clinically significant QTc interval prolongation was not observed.
At 960 times the maximum recommended islatravir dose, clinically significant QTc interval prolongation was not observed.
12.3 Pharmacokinetics
Single-dose administration of one IDVYNSO tablet to healthy adult participants under fasted conditions provided comparable exposures of doravirine and islatravir to administration of a doravirine tablet (100 mg) plus an islatravir capsule (0.25 mg). No clinically significant difference in pharmacokinetics was observed between healthy participants and participants living with HIV-1. Plasma pharmacokinetic properties of the components of IDVYNSO are provided in Table 4.
Table 4: Pharmacokinetic Properties of the Components of IDVYNSO Parameter Doravirine Islatravir Abbreviations: AUC=area under the time concentration curve; Cmax=maximum concentration; C24=concentration at 24 hours; Tmax=time to Cmax; Vd/F=apparent volume of distribution; t1/2=half-life; CL/F=apparent clearance; ADA= adenosine deaminase - * Reported as geometric mean (%CV: geometric coefficient of variation)
- † Doravirine 100 mg once daily to participants living with HIV
- ‡ IDVYNSO once daily to participants living with HIV
- § Under fasted conditions
- ¶ Geometric mean ratio [high-fat meal/fasting] and (90% confidence interval) for pharmacokinetic parameters. High fat meal is approximately 1000 kcal, 50% fat. The effect of food is not clinically relevant.
- # Based on population pharmacokinetic modeling and reported as the population mean (%CV)
- Þ Reported as the effective half-life of islatravir. The terminal half-life of islatravir is approximately 230 hours. The terminal half-life of intracellular ISL-TP is approximately 186 hours.
General Steady State Exposure AUC0-24* 37.8 (29) μMhr† 31.4 (15.3) nMhr‡ Cmax* 2.26 (19) μM† 3.45 (5.5) nM‡ C24* 0.930 (63) μM† 0.779 (22.3) nM‡ Time to Steady State (d) 2 7 Accumulation Ratio 1.2 to 1.4 1.8 Absorption Absolute Bioavailability 64% unknown Tmax (hr)§ 4 1 Effect of Food¶ AUC Ratio 1.17 (1.10, 1.24) 1.13 (1.07, 1.19) Cmax Ratio 1.18 (1.11, 1.25) 0.80 (0.71, 0.91) Distribution Vd/F (L)# 162 (32.6) 264 Plasma protein binding 76% 3% Elimination t1/2 (h) 15 21Þ CL/F (L/hr)# 6.34 (35.2) 27.7 (15.1) Metabolism Primary Pathway(s) CYP3A Oxidative deamination by ADA Excretion Major route of elimination Metabolism Metabolism Urine (unchanged) 6% 32% Biliary/Fecal (unchanged) Minor Minor Specific Populations
No clinically significant differences in the pharmacokinetics of doravirine were observed based on age (18 to 78 years), sex, race/ethnicity (White, Black or African American, Asian, and other), mild to severe renal impairment (creatinine clearance (CLcr) > 15 mL/min), and mild (Child-Pugh Class A) or moderate (Child-Pugh Class B) hepatic impairment. The pharmacokinetics of doravirine in patients with end-stage renal disease or undergoing dialysis, or severe hepatic impairment (Child-Pugh C) is unknown.
No clinically significant differences in the pharmacokinetics of islatravir were observed based on age (18 to 83 years), weight (36.6 to 205.3 kg), sex, race/ethnicity (White, Black or African American, Asian, and other), mild to moderate renal impairment (eGFR ≥30 mL/min/1.73 m2), and mild (Child-Pugh Class A) or moderate (Child-Pugh Class B) hepatic impairment. The pharmacokinetics of islatravir in patients undergoing dialysis, patients with severe hepatic impairment (Child-Pugh Class C), or people <18 years of age is unknown.
Patients with Renal Impairment
Islatravir: In a study comparing 6 participants with severe renal impairment (eGFR < 30 mL/min/1.73 m2, not on dialysis) to 6 participants without renal impairment, the single dose exposure of islatravir was approximately 2-fold higher in participants with severe renal impairment [see Use in Specific Populations (8.6)].
Drug Interaction Studies
As IDVYNSO is a complete regimen for the treatment of HIV-1 infection, it is not recommended to be administered with other HIV-1 antiretroviral medications.
Doravirine
Doravirine is primarily metabolized by CYP3A, and drugs that induce or inhibit CYP3A may affect the clearance of doravirine. Co-administration of doravirine and drugs that induce CYP3A may result in decreased plasma concentrations of doravirine. Co-administration of doravirine and drugs that inhibit CYP3A may result in increased plasma concentrations of doravirine.
Doravirine is not likely to have a clinically relevant effect on the exposure of medicinal products metabolized by CYP enzymes. Doravirine did not inhibit major drug metabolizing enzymes in vitro, including CYPs 1A2, 2B6, 2C8, 2C9, 2C19, 2D6, 3A4, and UGT1A1 and is not likely to be an inducer of CYP1A2, 2B6, or 3A4. Based on in vitro assays, doravirine is not likely to be an inhibitor of Organic Anion Transporter Polypeptide (OATP)1B1, OATP1B3, P-glycoprotein (P-gp), Bile Salt Export Pump (BSEP), Organic Anion Transporter (OAT)1, OAT3, OCT2, Multidrug and Toxin Extrusion Transporter (MATE)1, and MATE2K. Drug interaction studies were performed with doravirine and other drugs likely to be co-administered or commonly used as probes for pharmacokinetic interactions. The effects of co-administration with other drugs on the exposure (Cmax, AUC, and C24) of doravirine are summarized in Table 5. A single doravirine 100 mg dose was administered in these studies unless otherwise noted [see Contraindications (4), Warnings and Precautions (5.2), and Drug Interactions (7.2)].
Table 5: Drug Interactions: Changes in Pharmacokinetic Parameter Values of Doravirine in the Presence of Co-administered Drug Co-administered Drug Regimen of Co-administered Drug N Geometric Mean Ratio (90% CI) of Doravirine Pharmacokinetics with/without Co-administered Drug (No Effect=1.00) AUC* Cmax C24 CI = confidence interval; QD = once daily; BID = twice daily
All studies were conducted with doravirine as a single entity- * AUC0-∞ for single-dose, AUC0-24 for once daily
- † Changes in doravirine pharmacokinetic values are not clinically relevant.
- ‡ Doravirine 100 mg BID resulted in similar pharmacokinetic values when compared to 100 mg QD without rifabutin.
- § A single doravirine 50 mg dose (0.5 times the recommended approved dose) was administered.
- ¶ The first day following the cessation of efavirenz therapy and initiation of doravirine 100 mg QD
- # 14 days following the cessation of efavirenz therapy and initiation of doravirine 100 mg QD
Azole Antifungal Agents ketoconazole† 400 mg QD 10 3.06
(2.85, 3.29)1.25
(1.05, 1.49)2.75
(2.54, 2.98)Antimycobacterials rifampin 600 mg QD 10 0.12
(0.10, 0.15)0.43
(0.35, 0.52)0.03
(0.02, 0.04)rifabutin 300 mg QD 12 0.50
(0.45, 0.55)0.99
(0.85, 1.15)0.32
(0.28, 0.35)300 mg QD‡ 15 1.03
(0.94, 1.14)0.97
(0.87, 1.08)0.98
(0.88, 1.10)HIV Antiviral Agents ritonavir†, § 100 mg BID 8 3.54
(3.04, 4.11)1.31
(1.17, 1.46)2.91
(2.33, 3.62)efavirenz 600 mg QD¶ 17 0.38
(0.33, 0.45)0.65
(0.58, 0.73)0.15
(0.10, 0.23)600 mg QD# 17 0.68
(0.58, 0.80)0.86
(0.77, 0.97)0.50
(0.39, 0.64)islatravir 2.25 mg QD 9 1.13
(1.01, 1.28)1.11
(0.99, 1.25)1.12
(0.95, 1.32)Based on drug interaction studies conducted with doravirine, no clinically significant drug interactions have been observed following the co-administration of doravirine and the following drugs: islatravir, dolutegravir, ritonavir, TDF, lamivudine, elbasvir and grazoprevir, ledipasvir and sofosbuvir, ketoconazole, aluminum hydroxide/magnesium hydroxide/simethicone containing antacid, pantoprazole, atorvastatin, an oral contraceptive containing ethinyl estradiol and levonorgestrel, metformin, methadone, and midazolam.
Islatravir
Islatravir is primarily metabolized by ADA to form an inactive deoxyinosine metabolite. Islatravir depends on dCK for phosphorylation and activation. Islatravir is not significantly metabolized by CYP enzymes; therefore, it is not expected to be an object of CYP-mediated interactions. Islatravir is not a substrate for major hepatic or renal transporters, such as OAT1, OAT3, OCT2, MATE1, MATE2K, or P-gp, and is not anticipated to be an object of drug interactions mediated by these transporters. Islatravir was determined to be a substrate of Breast Cancer Resistance Protein (BCRP); however, considering its high absorption and lack of significant biliary excretion observed in preclinical species, islatravir is not expected to be an object of BCRP-mediated drug interactions.
Islatravir is not likely to have a clinically relevant effect on the exposure of medicinal products metabolized by CYP enzymes as it does not inhibit any major drug metabolizing enzymes including CYPs 1A2, 2B6, 2C8, 2C9, 2C19, 2D6, 3A, UGT1A1, or UGT2B7. Islatravir did not inhibit OATP1B1, OATP1B3, OCT1, OAT1, MATE1, MATE2K, P-gp, BSEP, MRP2, MRP3, or MRP4 at concentrations ≥75 µM, a concentration >10,000-fold the Cmax at the therapeutic dose. Islatravir inhibited OCT2, OAT3, and BCRP by 15% to 35% at 100 µM, but not at clinically relevant concentrations (<1 μM). Islatravir does not induce CYP3A4, 1A2, or 2B6, in vitro.
The effects of co-administration of other drugs on the Cmax, AUC, and C24 values of islatravir are summarized in Table 6 [see Drug Interactions (7.2)].
Table 6: Drug Interactions: Changes in Pharmacokinetic Parameter Values of Islatravir in the Presence of Co-administered Drug Co-administered Drug Regimen of Co-administered Drug Regimen of Islatravir N Geometric Mean Ratio (90% CI) of Islatravir Pharmacokinetics with/without Co-administered Drug (No Effect=1.00) AUC0-24 Cmax C24 All studies except pantoprazole were conducted with islatravir as a single entity. CI= confidence interval; QD= once daily; SD= single dose; TDF= tenofovir disoproxil fumarate - * Reported as AUC0-∞
- † Based on intracellular concentration of ISL-TP
HIV Antiviral Agents doravirine 100 mg QD 2.25 mg QD 9 1.06
(1.01, 1.12)1.08
(0.91, 1.27)-- dolutegravir + TDF 50 mg QD +300 mg QD 20 mg SD 12 1.28
(1.19, 1.37)*1.07
(0.93, 1.22)-- lamivudine† 300 mg QD 2 mg SD 20 0.13
(0.12, 0.15)*0.24
(0.20, 0.27)0.22
(0.18, 0.26)Proton Pump Inhibitors pantoprazole 40 mg QD 0.75 mg SD 6 1.05
(0.94, 1.16)0.99
(0.72, 1.35)-- Based on drug interaction studies conducted with islatravir, no clinically significant drug interactions have been observed following the co-administration of islatravir and the following drugs: doravirine, dolutegravir and TDF, an oral contraceptive containing ethinyl estradiol and levonorgestrel, methadone, atorvastatin, metformin.
In a clinical drug interaction study in healthy participants, co-administration of multiple daily doses of 300 mg lamivudine and a single dose of 2 mg islatravir (8 times the recommended dosage) decreased ISL-TP AUC0-∞ level by 87% and Cmax by 76% relative to islatravir alone. A similar effect is anticipated when islatravir is co-administered with FTC. The effect of other dCK substrates (including antineoplastic nucleoside metabolic inhibitors, e.g., cladribine, clofarabine, cytarabine, fludarabine, and gemcitabine) on the PK of ISL-TP is unknown; however, their co-administration with islatravir may result in a decrease in ISL-TP exposures [see Contraindications (4), Warnings and Precautions (5.2), and Drug Interactions (7.2)].
Drug interaction for the co-administration of IDVYNSO with ADA inhibitors has not been evaluated clinically. However, as islatravir is eliminated by renal excretion (approximately 32%) and ADA-mediated metabolism (approximately 53%), co-administration of ADA inhibitors (including pentostatin) with islatravir may result in an increase in plasma concentrations of islatravir [see Warnings and Precautions (5.2) and Drug Interactions (7.2)].
12.4 Microbiology
Mechanism of Action
Doravirine
Doravirine is a pyridinone non-nucleoside reverse transcriptase inhibitor of HIV-1 and inhibits HIV-1 replication by non-competitive inhibition of HIV-1 reverse transcriptase (RT). The inhibitory concentration at 50% (IC50) of doravirine for RNA-dependent DNA polymerization of recombinant wild-type HIV-1 RT in a biochemical assay was 12.2±2.0 nM (n=3). Doravirine does not inhibit the human cellular DNA polymerases α, β, and mitochondrial DNA polymerase γ.
Islatravir
Islatravir is a deoxyadenosine nucleoside analog reverse transcriptase inhibitor. Islatravir is phosphorylated by cellular kinases to form the pharmacologically active islatravir-triphosphate. Islatravir-triphosphate inhibits reverse transcriptase (RT) following incorporation into the nascent viral DNA by blocking translocation (immediate chain termination) and by inducing structural changes in viral DNA that prevent further nucleotide incorporation (delayed chain termination). Islatravir-triphosphate inhibited HIV-1 RT in a biochemical assay with an IC50 value of 346 ± 59 nM (n=17). Islatravir-triphosphate does not inhibit human cellular DNA polymerase β and mitochondrial DNA polymerase γ. Islatravir-triphosphate shows weak inhibition of human DNA polymerase α in a biochemical assay (IC50 value = 29.6 µM).
Antiviral Activity in Cell Culture
The combination of doravirine and islatravir was not antagonistic with respect to antiviral activity in cell culture.
Doravirine
Doravirine exhibited an EC50 value of 12.0±4.4 nM against wild-type laboratory strains of HIV-1 when tested in the presence of 100% normal human serum using MT4-GFP reporter cells and a median EC50 value for HIV-1 subtype B primary isolates (n=118) of 4.1 nM (range: 1.0 nM-16.0 nM). Doravirine demonstrated antiviral activity against a broad panel of primary HIV-1 isolates (subtypes A, A1, AE, AG, B, BF, C, D, G, H) with EC50 values ranging from 1.2 nM to 10.0 nM.
Islatravir
Islatravir exhibited EC50 values <1 nM against wild-type laboratory strains of HIV-1 in MT-4 cells, PBMCs and monocyte-derived macrophages. Islatravir demonstrated antiviral activity against a panel of 50 primary HIV-1 isolates (subtypes A, A1, AE, AG, B, BF, C, D, F1, G, H) with EC50 values ranging from 2.4 nM to 6.9 nM. The median EC50 value against the subtype B isolates was 4.3 nM (range 3.0 nM-5.3 nM) (n=6).
Resistance
In Cell Culture
Doravirine
Doravirine-resistant strains were selected in cell culture starting from wild-type HIV-1 of different origins and subtypes, as well as NNRTI-resistant HIV-1. Observed emergent amino acid substitutions in RT included: V106A, V106I, V106M, V108I, H221Y, F227C, F227I, F227L, F227V, M230I, L234I, P236L, and Y318F. The V106A, V106M, V108I, H221Y, F227C, M230I, P236L, and Y318F substitutions conferred 3.4-fold to 70-fold reductions in susceptibility to doravirine. Y318F in combination with V106A, V106M, V108I, or F227C conferred greater decreases in susceptibility to doravirine than Y318F alone, which conferred a 10-fold reduction in susceptibility to doravirine.
Islatravir
In cell culture, M184I and M184V were the main amino acid substitutions selected in RT with islatravir in wild-type HIV-1 of different origins and subtypes. The M184I substitution conferred a reduction in islatravir susceptibility of 3- to 7-fold and the M184V substitution conferred a reduction in islatravir susceptibility of 4- to 10-fold. The following substitutions were also observed in islatravir selection experiments: M41L, L74I, V90I, A114S, I142V, A158T, C162Y, T165A or R, H221Y, and A400T. Although the A114S substitution alone conferred 2-fold decreased susceptibility to islatravir, when A114S was in combination with M184V and other substitutions (e.g., A114S/M184V, M41L/A114S/M184V, and M41L/A114S/M184V/A400T), islatravir had 38- to 65-fold decreased susceptibility. The other substitutions conferred a ≤2-fold decreased susceptibility to islatravir.
In Clinical Trials
In the IDVYNSO arm of Trial 051 (n=366), 5 participants had HIV-1 RNA ≥50 copies/mL at Week 48. Two participants had virologic failure with HIV-1 RNA >20,000 copies/mL at Week 4 and were ineligible for enrollment due to a prior history of virologic failure and/or doravirine resistance-associated substitutions. Both had NNRTI resistance-associated substitutions (A98A/G, K101A, K103K/N, V106V/I or M, Y188Y/L or L/W, F227L, and/or L228L/R), thymidine analog substitutions (many as mixtures with wild-type), and substitutions M184M/I/V or T at baseline. By Week 4, the baseline NNRTI resistance-associated substitutions and thymidine analog resistance substitutions had developed to full substitutions without mixtures with wild-type and were present with M184V or I/T. Phenotypic analysis showed resistance to doravirine with >74- to >93-fold reduced susceptibility and 8- to 25-fold reduced susceptibility to islatravir. Two other participants had no evidence of genotypic or phenotypic resistance emergence and one participant had all samples fail resistance testing.
There were no participants in the baseline ART group with HIV-1 RNA ≥50 copies/mL at Week 48 who had samples submitted for post-baseline resistance analysis.
In the IDVYNSO arm of Trial 052 (n=342), 5 participants had HIV-1 RNA ≥50 copies/mL at Week 48. One participant had resistance-associated substitutions T69T/S, K70K/R, K101K/E, K103K/N, and M184M/V at baseline and K103N, M184V, H221Y, M230L and L234I at Week 24. Phenotypic analysis at Week 24 showed resistance to doravirine with >104-fold reduced susceptibility and 7-fold reduced susceptibility to islatravir. Another participant, who had resistance-associated substitutions M41L, V106V/I, Y181Y/C, M184M/V, and T215E at baseline, had HIV-1 RNA <50 copies/mL at Week 60 and had no post-baseline resistance testing conducted. For the other 3 participants, there was no evidence of resistance post-baseline (one had no evidence of genotypic or phenotypic resistance, one had viral load below the assay cutoff for resistance testing, and one had failed resistance testing).
There were no participants in the BIC/FTC/TAF group with HIV-1 RNA ≥50 copies/mL at Week 48 who had samples submitted for post-baseline resistance analysis.
Baseline NNRTI and NRTI resistance-associated substitutions from virologic failure on previous ART regimens likely contributed to the treatment outcome (HIV-1 RNA ≥50 copies/mL) at Week 48 in 4 participants receiving IDVYNSO in Trial 051 and Trial 052, emphasizing the importance of having no prior virologic treatment failure before initiating IDVYNSO.
Presence of NNRTI Resistance-associated Substitutions and/or M184I, V, or T at Baseline
Of the 598 virologically-suppressed participants receiving IDVYNSO in Trial 051 and Trial 052 with resistance data at baseline and virologic data at Week 48, 152 (25%) had NNRTI resistance-associated substitutions and 41 (7%) had M184I, V or T at baseline. In the IDVYNSO groups, 4 participants who had NNRTI resistance-associated substitutions and M184I, V or T at baseline and 1 participant who had NNRTI resistance-associated substitutions only at baseline had HIV-1 RNA ≥50 copies/mL at Week 48. In the comparator groups in Trial 051 and Trial 052, none of the participants who had NNRTI resistance-associated substitutions and M184I, V or T at baseline and 2 participants who had NNRTI resistance-associated substitutions only at baseline had HIV-1 RNA ≥50 copies/mL at Week 48.
Cross-Resistance
Doravirine
Cross-resistance has been observed among NNRTIs. Treatment-emergent doravirine resistance-associated substitutions can confer cross resistance to efavirenz, etravirine, nevirapine, and rilpivirine. The treatment-emergent doravirine resistance-associated substitution Y318F alone did not confer reduced susceptibility to efavirenz, etravirine, or rilpivirine.
A panel of 96 diverse clinical isolates containing NNRTI resistance-associated substitutions was evaluated for susceptibility to doravirine. Clinical isolates containing the Y188L substitution alone or in combination with K103N or V106I, V106A in combination with G190A and F227L, or E138K in combination with Y181C and M230L showed greater than 100-fold reduced susceptibility to doravirine.
Doravirine maintained antiviral activity against variants containing NRTI resistance-associated substitutions M184I and M184V.
Islatravir
Islatravir was evaluated in cell culture against a panel of 94 diverse clinical isolates containing substitutions in HIV-1 RT for antiviral activity. The M184I and M184V substitutions conferred a 5-fold reduction in susceptibility to islatravir. Thymidine analog substitutions conferred 3- to 4-fold reductions in susceptibility to islatravir; variants with insertions at RT position 69 (69 Ins) conferred a 10-fold reduction in susceptibility to islatravir. The addition of M184I or V substitutions in variants containing thymidine analog substitutions or 69 ins reduced islatravir susceptibility by 11- to 18-fold and 21-fold, respectively.
Islatravir maintained antiviral activity against variants containing NNRTI resistance-associated substitutions.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Doravirine
Doravirine was not carcinogenic in a 6-month transgenic mouse study at doses of up to 300 mg/kg/day or in a 2-year rat oral carcinogenicity study at exposures up to 7 times the human exposures at the RHD. A statistically significant incidence of thyroid parafollicular cell adenoma and carcinoma seen only in female rats at the high dose was within the range observed in historical controls.
Islatravir
Islatravir was not carcinogenic in a 6-month transgenic mouse study at doses of up to 13 mg/kg/day or in a 2-year rat oral carcinogenicity study at exposures approximately 600 times the human exposures at the RHD.
Mutagenesis
Doravirine
Doravirine was not genotoxic in a battery of in vitro or in vivo assays, including microbial mutagenesis, chromosomal aberration in Chinese hamster ovary cells, and in in vivo rat micronucleus assays.
Islatravir
Islatravir was not mutagenic in the in vitro microbial mutagenicity assay. Islatravir was positive in an in vitro chromosomal aberration assay in Chinese hamster ovary cells likely due to nucleotide pool imbalances; however, it was not genotoxic in an in vivo rat micronucleus assay at systemic exposures (AUC) approximately 2700 times the human exposure at the RHD. Islatravir is not genotoxic at clinically relevant exposures.
Impairment of Fertility
Doravirine
There were no effects on fertility, mating performance, or early embryonic development when doravirine was administered to rats at systemic exposures (AUC) approximately 7 times the exposure in humans at the RHD.
Islatravir
There were no effects on fertility, mating performance, or early embryonic development when islatravir was administered to rats at systemic exposures (AUC) approximately 1000 times the exposure in humans at the RHD.
-
14 CLINICAL STUDIES
The efficacy of IDVYNSO is supported by data from 2 randomized, active-controlled, non-inferiority trials (Trial 051 [NCT05631093] and Trial 052 [NCT05630755]) in virologically-suppressed (HIV-1 RNA less than 50 copies per mL) participants living with HIV. Participants must have been stably suppressed on their baseline regimen for at least 3 months prior to trial entry and had no history of treatment failure. Participants with active HBV infection [hepatitis B surface antigen (HBsAg) positive or HBV DNA positive] were excluded from the trials.
In open-labeled Trial 051, participants switched from an oral ART regimen to IDVYNSO. A total of 551 participants were randomized (2:1) and switched to once-daily IDVYNSO (N=366) or remained on their baseline ART (N=185). Randomization was stratified by baseline ART. At baseline, participants had a mean age of 50 years (range: 18 to 83), 60% of participants were male, 39% were White, 45% were Black/African American, 5% were Asian, and 11% were other or unknown. A total of 15% identified as Hispanic/Latino. The mean baseline CD4+ T-cell count was 748 cells/mm3 and 80% of participants had baseline CD4+ T-cell count >500 cells/mm3. At enrollment, 64% of the participants were receiving InSTI-based regimens, 5% PI-based regimens (including combinations with InSTI), and 30% other regimens. Approximately 29% of participants in both study arms had evidence of past HBV infection [hepatitis B core antibody (HBcAb) positive] at enrollment. These characteristics were similar between treatment groups.
In the double-blind Trial 052, participants switched from bictegravir/emtricitabine/tenofovir alafenamide (BIC/FTC/TAF) to IDVYNSO. A total of 513 participants were randomized (2:1) and switched to once-daily IDVYNSO (N=342) or remained on BIC/FTC/TAF (N=171). At baseline, participants had a mean age of 48 years (range: 19 to 77), 79% of participants were male, 61% were White, 31% were Black/African American, 6% were Asian, and 3% were other or unknown. A total of 23% identified as Hispanic/Latino. The mean baseline CD4+ T-cell count was 717 cells/mm3 and 75% of participants had baseline CD4+ T-cell count >500 cells/mm3. Approximately 26% of participants in both study arms were HBcAb positive at enrollment. These characteristics were similar between treatment groups.
The primary endpoint of Trial 051 and Trial 052 was the proportion of participants with HIV-1 RNA ≥50 copies/mL at Week 48. Treatment outcomes are shown in Table 7.
Table 7: Virologic Outcomes in Trial 051 and Trial 052 at Week 48 in Virologically-Suppressed Adults Who Switched to IDVYNSO Outcomes Trial 051 Trial 052 IDVYNSO
N=366Baseline ART
N=185IDVYNSO
N=342BIC/FTC/TAF
N=171- * CIs were calculated based on the stratified Miettinen and Nurminen method with Cochran-Mantel-Haenszel weights for Trial 051 (with baseline ART as stratification factors), and unstratified Miettinen and Nurminen method for Trial 052; non-inferiority (NI) was assessed using a NI margin of 4%
- † Includes participants who discontinued because of AE or death if this resulted in no virologic data on treatment during the Week 48 window
- ‡ Other reasons include: lost to follow-up, physician decision, and withdrawal by participant
HIV-1 RNA ≥50 copies/mL 1% 5% 1% 1% Treatment Difference (95% CI)* -3.6% (-7.8%, -0.8%) 0.9% (-1.9%, 2.9%) HIV-1 RNA <50 copies/mL 96% 92% 92% 94% No Virologic Data at Week 48 Window 3% 3% 7% 5% Discontinued study due to AE or Death† 1% 1% 2% 2% Discontinued study for Other Reasons‡ 2% 2% 5% 4% On study drug but missing data in window 1% 1% - - In Trial 051, treatment outcomes between treatment groups were similar across subgroups by age, sex, race and baseline ART regimens. In Trial 052, treatment outcomes between treatment groups were similar across subgroups by age, sex and race.
In Trial 051, the mean increase from baseline at Week 48 in CD4+ T-cell counts in the IDVYNSO and baseline ART groups were 5 and 18 cells/mm3, respectively. In Trial 052, the mean increase from baseline at Week 48 in CD4+ T-cell counts in the IDVYNSO and BIC/FTC/TAF groups was 30 and 28 cells/mm3, respectively.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
Each IDVYNSO tablet contains 100 mg of doravirine and 0.25 mg of islatravir, is pink, oval-shaped and film-coated, and is debossed with 772 on one side and plain on the other side. Each bottle contains 30 tablets (NDC: 0006-5092-01) with desiccant and is closed with a child-resistant closure.
Store IDVYNSO in the original bottle. Keep the bottle tightly closed to protect from moisture. Do not remove the desiccant.
Store IDVYNSO at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
-
17 PATIENT COUNSELING INFORMATION
Advise patients to read the FDA-approved patient labeling (Patient Information).
Skin and Hypersensitivity Reactions
Inform patients that severe skin reactions including Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN) and Drug Rash with Eosinophilia and Systemic Symptoms (DRESS) have been reported with doravirine-containing regimens. Advise patients to immediately contact their healthcare provider if they develop a rash. Instruct patients to immediately stop taking IDVYNSO and seek medical attention if a painful rash with mucosal involvement or a rash with constitutional symptoms develops [see Warnings and Precautions (5.1)].
Drug Interactions
Inform patients that IDVYNSO may interact with certain other drugs; therefore, advise patients to report to their healthcare provider the use of any other prescription or nonprescription medication or herbal products, including St. John’s wort [see Contraindications (4), Warnings and Precautions (5.2), and Drug Interactions (7.2)].
For patients concomitantly receiving rifabutin, take one tablet of doravirine (PIFELTRO) 100 mg approximately 12 hours after the dose of IDVYNSO [see Dosage and Administration (2.2)].
Dosing Instructions
Advise patients to take IDVYNSO every day at a regularly scheduled time with or without food. Inform patients that it is important not to miss or skip doses as it can result in development of resistance. If patients forget to take IDVYNSO, tell patients to take the missed dose right away, unless it is almost time for the next dose [see Dosage and Administration (2.1)].
Pregnancy Registry
Inform patients that there is an antiretroviral pregnancy registry to monitor fetal outcomes in pregnant individuals exposed to IDVYNSO [see Use in Specific Populations (8.1)].
Lactation
Inform patients that the potential risks of breastfeeding include: (1) HIV-1 transmission (in infants without HIV-1), (2) developing viral resistance (in infants with HIV-1), and (3) serious adverse reactions in a breastfed infant similar to those seen in adults [see Use in Specific Populations (8.2)].
-
SPL UNCLASSIFIED SECTION
Manufactured for: Merck Sharp & Dohme LLC, Rahway, NJ 07065, USA
For patent information: www.msd.com/research/patent. The trademarks depicted herein are owned by Merck & Co., Inc.
Copyright © 2026 Merck & Co., Inc., Rahway, NJ, USA, and its affiliates.
All rights reserved.uspi-mk8591a-t-2604r000
-
PATIENT PACKAGE INSERT
Patient Information
IDVYNSO™ (ihd‑VIHN‑soh)
(doravirine and islatravir)
tabletsThis Patient Information has been approved by the U.S. Food and Drug Administration Issued: 04/2026 What is IDVYNSO?
IDVYNSO is a prescription medicine that is used without other Human Immunodeficiency Virus-1 (HIV-1) medicines to treat HIV-1 infection in adults to replace their current HIV-1 medicines for people whose healthcare provider determines that they meet certain requirements.
HIV-1 is the virus that causes Acquired Immune Deficiency Syndrome (AIDS).
IDVYNSO contains the prescription medicines doravirine and islatravir.
It is not known if IDVYNSO is safe and effective in children under 18 years of age.Do not take IDVYNSO if you take: - certain medicines called strong CYP3A inducers which increase the enzyme CYP3A in your liver.
- lamivudine or emtricitabine.
Before taking IDVYNSO, tell your healthcare provider about all of your medical conditions, including if you: - have liver problems
- have ever had hepatitis B virus infection
- have kidney problems
- are pregnant or plan to become pregnant. It is not known if IDVYNSO can harm your unborn baby. Tell your healthcare provider if you become pregnant during treatment with IDVYNSO.
Pregnancy Registry: There is a pregnancy registry for people who take IDVYNSO during pregnancy. The purpose of this registry is to collect information about the health of you and your baby. Talk to your healthcare provider about how you can take part in this registry. - are breastfeeding or plan to breastfeed. It is not known if IDVYNSO passes into your breast milk. Talk with your healthcare provider about the following risks to your baby from breastfeeding during treatment with IDVYNSO:
- the HIV-1 virus may pass to your baby if your baby does not have HIV-1.
- the HIV-1 virus may become harder to treat if your baby has HIV-1.
- your baby may get side effects from IDVYNSO.
- Some medicines interact with IDVYNSO. Keep a list of your medicines to show your healthcare provider and pharmacist.
- Tell your healthcare provider if you have taken rifabutin in the past 4 weeks.
- You can ask your healthcare provider or pharmacist for a list of medicines that interact with IDVYNSO.
- Do not start taking a new medicine without telling your healthcare provider. Your healthcare provider can tell you if it is safe to take IDVYNSO with other medicines.
How should I take IDVYNSO? - Take IDVYNSO every day exactly as your healthcare provider tells you to take it.
- Take IDVYNSO 1 time each day, at about the same time every day.
- Take IDVYNSO with or without food.
- IDVYNSO is taken by itself (without other HIV-1 medicines).
- If you take the medicine rifabutin during treatment with IDVYNSO, your healthcare provider will also prescribe an additional dose of doravirine for you. You may not have enough doravirine in your blood if you take rifabutin during treatment with IDVYNSO. Carefully follow your healthcare provider’s instructions about when to take doravirine and how much to take. This is usually 1 tablet of doravirine about 12 hours after your dose of IDVYNSO.
- Do not change your dose or stop taking IDVYNSO without talking to your healthcare provider. Stay under a healthcare provider’s care when taking IDVYNSO.
- It is important that you do not miss or skip doses of IDVYNSO.
- If you miss a dose of IDVYNSO, take it as soon as you remember. If it is almost time for your next dose, skip the missed dose, and take the next dose at your regular time. Do not take 2 doses of IDVYNSO at the same time.
- If you have any questions, call your healthcare provider or pharmacist.
- If you take too much IDVYNSO, call your healthcare provider or go to the nearest hospital emergency room right away.
- When your supply of IDVYNSO starts to run low, get more from your healthcare provider or pharmacy. This is very important because the amount of virus in your blood may increase if the medicine is stopped for even a short time. The virus may develop resistance to IDVYNSO and become harder to treat.
What are the possible side effects of IDVYNSO?
IDVYNSO may cause serious side effects including:- Severe skin reactions have happened in people treated with IDVYNSO. Call your healthcare provider right away if you develop a rash during treatment with IDVYNSO. Stop taking IDVYNSO and get medical help right away if you develop a painful rash with any of the following symptoms:
- fever
- blisters or peeling of the skin
- feeling tired or generally ill
- blisters or sores in the mouth
- swelling of the face or lymph nodes
- redness or swelling of the eyes (conjunctivitis)
The most common side effects of IDVYNSO include: - diarrhea
- dizziness
- tiredness
- stomach (abdominal) bloating
- headache
- weight gain
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.How should I store IDVYNSO? - Store IDVYNSO tablets at room temperature between 68°F and 77°F (20°C to 25°C).
- Keep IDVYNSO in the original bottle.
- Do not take the tablets out of the bottle to store in another container, such as a pill box.
- Keep the bottle tightly closed to protect IDVYNSO from moisture.
- The IDVYNSO bottle contains desiccants to help keep your medicine dry (protect it from moisture). Keep the desiccants in the bottle. Do not eat the desiccants.
General information about the safe and effective use of IDVYNSO.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use IDVYNSO for a condition for which it was not prescribed. Do not give IDVYNSO to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about IDVYNSO that is written for healthcare professionals.What are the ingredients in IDVYNSO?
Active ingredients: doravirine and islatravir
Inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, hypromellose acetate succinate, lactose monohydrate, magnesium stearate, microcrystalline cellulose.
The tablet film coating contains calcium carbonate, ferric oxide, ferrosoferric oxide, hypromellose, lactose monohydrate, and triacetin. The coated tablets are polished with carnauba wax.
Manufactured for Merck Sharp & Dohme LLC, Rahway, NJ 07065, USA
For patent information: www.msd.com/research/patent. The trademark depicted herein is owned by Merck & Co., Inc.
Copyright © 2026 Merck & Co., Inc., Rahway, NJ, USA, and its affiliates. All rights reserved.
usppi-mk8591a-t-2604r000 -
PRINCIPAL DISPLAY PANEL - 30 Tablet Bottle Label
NDC: 0006-5092-01
Idvynso™
(doravirine and islatravir)
tablets100 mg/0.25 mg
ALERT: Find out about medicines that
should NOT be taken with Idvynso™.Rx only
30 Tablets
-
INGREDIENTS AND APPEARANCE
IDVYNSO
doravirine, islatravir tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0006-5092 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DORAVIRINE (UNII: 913P6LK81M) (DORAVIRINE - UNII:913P6LK81M) DORAVIRINE 100 mg ISLATRAVIR ANHYDROUS (UNII: QPQ082R25D) (ISLATRAVIR ANHYDROUS - UNII:QPQ082R25D) ISLATRAVIR ANHYDROUS 0.25 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) HYPROMELLOSE ACETATE SUCCINATE, UNSPECIFIED (UNII: A7ZHS2RJ34) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) CALCIUM CARBONATE (UNII: H0G9379FGK) FERRIC OXIDE RED (UNII: 1K09F3G675) FERROSOFERRIC OXIDE (UNII: XM0M87F357) HYPROMELLOSE 2910 (15 MPA.S) (UNII: 36SFW2JZ0W) TRIACETIN (UNII: XHX3C3X673) CARNAUBA WAX (UNII: R12CBM0EIZ) HYPROMELLOSE 2910 (6 MPA.S) (UNII: 0WZ8WG20P6) Product Characteristics Color PINK Score no score Shape OVAL Size 17mm Flavor Imprint Code 772;plain Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0006-5092-01 30 in 1 BOTTLE; Type 0: Not a Combination Product 04/23/2026 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA216964 04/23/2026 Labeler - Merck Sharp & Dohme LLC (118446553)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.