GAMUNEX-C (immune globulin- human injection
Drug Labeling and Warnings
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use GAMUNEX-C FlexBag safely and effectively. See full prescribing information for GAMUNEX-C FlexBag.
GAMUNEX-C FlexBag, [immune globulin injection (human), 10%, caprylate/chromatography purified] for intravenous use.
Initial U.S. Approval: 2003WARNING: THROMBOSIS, RENAL DYSFUNCTION and ACUTE RENAL FAILURE
See full prescribing information for complete boxed warning.
- Thrombosis may occur with immune globulin products, including GAMUNEX-C FlexBag. Risk factors may include: advanced age, prolonged immobilization, hypercoagulable conditions, history of venous or arterial thrombosis, use of estrogens, indwelling vascular catheters, hyperviscosity, and cardiovascular risk factors.
- For patients at risk of thrombosis, administer GAMUNEX-C FlexBag at the minimum dose and infusion rate practicable. Ensure adequate hydration in patients before administration. Monitor for signs and symptoms of thrombosis and assess blood viscosity in patients at risk for hyperviscosity.
- Renal dysfunction, acute renal failure, osmotic nephrosis, and death may occur with immune globulin intravenous (IGIV) products in predisposed patients.
- Renal dysfunction and acute renal failure occur more commonly in patients receiving IGIV products containing sucrose. GAMUNEX-C FlexBag does not contain sucrose.
- For patients at risk of renal dysfunction or failure, administer GAMUNEX-C FlexBag at the minimum concentration available and the minimum infusion rate practicable. (5.2)
RECENT MAJOR CHANGES
INDICATIONS AND USAGE
GAMUNEX-C FlexBag is an immune globulin injection (human), 10% liquid indicated for treatment of:
DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHS
GAMUNEX-C FlexBag is a sterile solution for injection supplied in 5 g (50 mL), 10 g (100 mL), 20 g (200 mL), or 40 g (400 mL) single-dose flexible containers. (3)
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
- IgA deficient patients with antibodies against IgA are at greater risk of developing severe hypersensitivity and anaphylactic reactions. Have epinephrine available immediately to treat any acute severe hypersensitivity reactions. (5.1)
- Hyperproteinemia, with resultant changes in serum viscosity and electrolyte imbalances may occur in patients receiving IGIV therapy. (5.3)
- Aseptic Meningitis Syndrome (AMS) may occur, especially with high doses or rapid infusion. (5.5)
- Hemolysis, either intravascular or due to enhanced RBC sequestration, can develop subsequent to GAMUNEX-C FlexBag treatments. Risk factors include high doses and non-O blood group. Closely monitor patients for hemolysis and hemolytic anemia, especially in patients with pre-existing anemia and/or cardiovascular or pulmonary compromise. (5.6)
- Monitor patients for pulmonary adverse reactions (transfusion-related acute lung injury [TRALI]). (5.7)
- Volume overload. (5.8)
- GAMUNEX-C FlexBag is made from human plasma and may carry a risk of transmitting infectious agents, e.g., viruses, the variant Creutzfeldt-Jakob disease (vCJD) agent and, theoretically, the Creutzfeldt-Jakob disease (CJD) agent. (5.9)
- Passive transfer of antibodies may confound serologic testing. (5.11)
ADVERSE REACTIONS
The most common adverse reactions observed in ≥ 5% patients were: (6.1)
PI: Cough increased, rhinitis, pharyngitis, headache, asthma, nausea, fever, diarrhea, and sinusitis.
ITP: Headache, ecchymosis, vomiting, fever, nausea, rash, abdominal pain, back pain and dyspepsia.
CIDP: Headache, pyrexia, hypertension, chills, rash, nausea, arthralgia, and asthenia.
To report SUSPECTED ADVERSE REACTIONS, contact Grifols Therapeutics LLC at 1-800-520-2807 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- The passive transfer of antibodies may transiently interfere with the response to live virus vaccines, such as measles, mumps and rubella. (7)
USE IN SPECIFIC POPULATIONS
- Geriatric: In patients over 65 years of age do not exceed the recommended dose and infuse GAMUNEX-C FlexBag at the minimum infusion rate practicable. (8.5)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 4/2026
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: THROMBOSIS, RENAL DYSFUNCTION, and ACUTE RENAL FAILURE
1 INDICATIONS AND USAGE
1.1 Primary Humoral Immunodeficiency (PI)
1.2 Idiopathic Thrombocytopenic Purpura (ITP)
1.3 Chronic Inflammatory Demyelinating Polyneuropathy (CIDP)
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage
2.2 Preparation and Handling
2.3 Administration
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity
5.2 Renal Failure
5.3 Hyperproteinemia, Increased Serum Viscosity, and Hyponatremia
5.4 Thrombosis
5.5 Aseptic Meningitis Syndrome (AMS)
5.6 Hemolysis
5.7 Transfusion-related Acute Lung Injury (TRALI)
5.8 Volume Overload
5.9 Transmission of Infectious Agents
5.10 Monitoring: Laboratory Tests
5.11 Interference with Laboratory Tests
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.3 Pharmacokinetics
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: THROMBOSIS, RENAL DYSFUNCTION, and ACUTE RENAL FAILURE
- Thrombosis may occur with immune globulin products, including GAMUNEX®-C FlexBag. Risk factors may include: advanced age, prolonged immobilization, hypercoagulable conditions, history of venous or arterial thrombosis, use of estrogens, indwelling central vascular catheters, hyperviscosity, and cardiovascular risk factors. Thrombosis may occur in the absence of known risk factors. [see Warnings and Precautions (5.4), Patient Counseling Information (17)]
- For patients at risk of thrombosis, administer GAMUNEX-C FlexBag at the minimum dose and infusion rate practicable. Ensure adequate hydration in patients before administration. Monitor for signs and symptoms of thrombosis and assess blood viscosity in patients at risk for hyperviscosity. [see Dosage and Administration (2.3), Warnings and Precautions (5.4)]
- Renal dysfunction, acute renal failure, osmotic nephrosis, and death may occur with immune globulin intravenous (IGIV) products in predisposed patients. Patients predisposed to renal dysfunction include those with any degree of pre-existing renal insufficiency, diabetes mellitus, age greater than 65, volume depletion, sepsis, paraproteinemia, or patients receiving known nephrotoxic drugs.
- Renal dysfunction and acute renal failure occur more commonly in patients receiving IGIV products containing sucrose. GAMUNEX-C FlexBag does not contain sucrose.
- For patients at risk of renal dysfunction or failure, administer GAMUNEX-C FlexBag at the minimum concentration available and the minimum infusion rate practicable. [see Warnings and Precautions (5.2)]
-
1 INDICATIONS AND USAGE
GAMUNEX-C FlexBag is an immune globulin injection (human) 10% liquid that is indicated for the treatment of:
1.1 Primary Humoral Immunodeficiency (PI)
GAMUNEX-C FlexBag is indicated for treatment of primary humoral immunodeficiency in patients 2 years of age and older. This includes, but is not limited to, congenital agammaglobulinemia, common variable immunodeficiency, X-linked agammaglobulinemia, Wiskott-Aldrich syndrome, and severe combined immunodeficiencies.
-
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage
Primary Humoral Immunodeficiency (PI)
As there are significant differences in the half-life of IgG among patients with primary humoral immunodeficiencies, the ideal frequency and amount of immunoglobulin therapy may vary from patient to patient. The proper amount can be determined by monitoring clinical response.
The dose of GAMUNEX-C FlexBag for patients with PI is 300 mg/kg to 600 mg/kg body weight (3 mL/kg to 6 mL/kg) administered every 3 to 4 weeks. The dosage may be adjusted over time to achieve the desired trough levels and clinical responses.
The recommended initial infusion rate is 1 mg/kg/min (0.01 mL/kg/min). If the infusion is well-tolerated, the rate may be gradually increased to a maximum of 8 mg/kg/min (0.08 mL/kg/min). For patients judged to be at risk for renal dysfunction or thrombosis, administer GAMUNEX-C FlexBag at the minimum infusion rate practicable. [see Warnings and Precautions (5.2,5.4)]
If a patient routinely receives a dose of less than 400 mg/kg of GAMUNEX-C FlexBag every 3 to 4 weeks (less than 4 mL/kg), and is at risk of measles exposure (i.e., traveling to a measles endemic area), administer a dose of at least 400 mg/kg (4 mL/kg) just prior to the expected measles exposure. If a patient has been exposed to measles, a dose of 400 mg/kg (4 mL/kg) should be administered as soon as possible after exposure.
Idiopathic Thrombocytopenic Purpura (ITP)
GAMUNEX-C FlexBag may be administered at a total dose of 2 g/kg, divided in two doses of 1 g/kg (10 mL/kg) given on two consecutive days or into five doses of 0.4 g/kg (4 mL/kg) given on five consecutive days. If after administration of the first of two daily 1 g/kg (10 mL/kg) doses, an adequate increase in the platelet count is observed at 24 hours, the second dose of 1 g/kg (10 mL/kg) body weight may be withheld.
The high dose regimen (1 g/kg × 1-2 days) is not recommended for individuals with expanded fluid volumes or where fluid volume may be a concern. [see Warnings and Precautions (5.8), Clinical Studies (14)]
The recommended initial infusion rate is 1 mg/kg/min (0.01 mL/kg/min). If the infusion is well-tolerated, the rate may be gradually increased to a maximum of 8 mg/kg/min (0.08 mL/kg/min). For patients judged to be at risk for renal dysfunction or thrombosis, administer GAMUNEX-C FlexBag at the minimum infusion rate practicable. [see Warnings and Precautions (5.2,5.4)]
Chronic Inflammatory Demyelinating Polyneuropathy (CIDP)
GAMUNEX-C FlexBag may be initially administered as a total loading dose of 2 g/kg (20 mL/kg) given in divided doses over two to four consecutive days. GAMUNEX-C FlexBag may be administered as a maintenance infusion of 1 g/kg (10 mL/kg) administered over 1 day or divided into two doses of 0.5 g/kg (5 mL/kg) given on two consecutive days, every 3 weeks. Not all patients may require continued maintenance therapy beyond the initial 6 months of therapy in order to maintain their therapeutic response.
The recommended initial infusion rate is 2 mg/kg/min (0.02 mL/kg/min). If the infusion is well tolerated, the rate may be gradually increased to a maximum of 8 mg/kg/min (0.08 mL/kg/min). For patients judged to be at risk for renal dysfunction or thrombosis, administer GAMUNEX-C FlexBag at the minimum infusion rate practicable. [see Warnings and Precautions (5.2,5.4)]
2.2 Preparation and Handling
- Visually inspect GAMUNEX-C FlexBag for particulate matter and discoloration prior to administration, whenever solution and container permit. Do not use if turbid or if particulate matter is present
- Do not freeze. Do not use solutions that have been frozen.
- Prior to use, allow the solution to reach ambient room temperature.
- If the container or overwrap show any signs of damage or tampering, do not use the product and notify Grifols Therapeutics LLC immediately [1-800-520-2807].
- The GAMUNEX-C FlexBag is for single use only. GAMUNEX-C FlexBag contains no preservative. Use any bag that has been entered promptly. Discard partially used bags. Do not store after entry into bag.
- Infuse GAMUNEX-C FlexBag using a separate line by itself, without mixing with other intravenous fluids or medications the subject might be receiving. The GAMUNEX-C FlexBag infusion line can be flushed with 5% dextrose in water (D5/W) or 0.9% sodium chloride for injection.
- If dilution is required, GAMUNEX-C may be diluted with 5% dextrose in water (D5/W). Do not dilute with saline.
- Avoid simultaneous administration of GAMUNEX-C FlexBag and Heparin through a single lumen delivery device due to incompatibility of GAMUNEX-C FlexBag with heparin. Flush Heparin Lock (Hep-Lock) through which GAMUNEX-C FlexBag was administered with 5% dextrose in water (D5/W) or 0.9% sodium chloride for injection, and do not flush with Heparin. See table below.
Additional Solutions Dilution Line Flush Delivery Device Flush 5% Dextrose in water Yes Yes Yes 0.9% Sodium Chloride No Yes Yes Heparin No No No - Do not mix with immune globulin intravenous (IGIV) products from other manufacturers.
- Do not use after expiration date.
- Some moisture or condensation may be observed in the protective overwrap. This is normal and does not affect the quality or safety of the solution.
- Prior to administration, check the bag for any leaks by squeezing it firmly. If leaks are found, discard the bag as sterility may be compromised.
CAUTION: Do not use bags in series connections. Such use could result in air embolism due to residual air being drawn from the primary bag before the administration of the fluid from the secondary bag is complete.
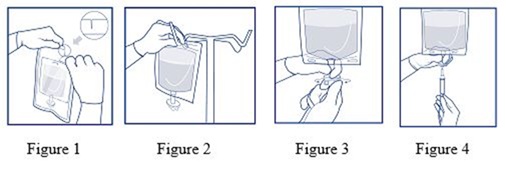
- After checking that protective overwrap is not damaged, remove it by tearing the slots at either end (refer to Figure 1).
- Suspend the inner bag from the eyelet support (refer to Figure 2).
- Holding the protective safety shield at the infusion port of the inner bag with one hand, use the free hand to exert light force to turn the twist-off opening about 90 degrees until it leaves the port (refer to Figure 3).
- Attach either a non-vented or vented administration set (refer to Figure 4). Adjust the infusion rate to the individual circumstances and the indication. Refer to the complete directions of administration set used.
2.3 Administration
Administer intravenously.
- Administer GAMUNEX-C FlexBag at room temperature.
- Inspect GAMUNEX-C FlexBag visually for particulate matter and discoloration prior to administration, whenever the solution and container permit.
- Do not use if turbid, if particulate matter is present, and/or if discoloration is observed.
- Promptly administer GAMUNEX-C after opening the Flexbag. Discard partially used bags.
- If dilution is required, GAMUNEX-C FlexBag may be diluted with 5% dextrose in water (D5/W). Do not dilute with saline. Infuse GAMUNEX-C FlexBag using a separate line by itself, without mixing with other intravenous fluids or medications the subject might be receiving. The GAMUNEX-C FlexBag infusion line can be flushed with 5% dextrose in water (D5/W) or 0.9% sodium chloride for injection.
Rate of Administration
Following initial infusion (see table below), the infusion rate may be gradually increased to a maximum of 0.08 mL/kg per minute (8 mg/kg per minute) as tolerated.
Indication Initial Infusion Rate
(first 30 minutes)Maximum Infusion Rate
(if tolerated)PI 1 mg/kg/min 8 mg/kg/min ITP 1 mg/kg/min 8 mg/kg/min CIDP 2 mg/kg/min 8 mg/kg/min Monitor patient vital signs throughout the infusion. Slow or stop infusion if adverse reactions occur. If symptoms subside promptly, the infusion may be resumed at a lower rate that is comfortable for the patient.
Certain severe adverse drug reactions may be related to the rate of infusion. Slowing or stopping the infusion usually allows the symptoms to disappear promptly.
Ensure that patients with pre-existing renal insufficiency are not volume depleted. For patients at risk of renal dysfunction or thrombosis, administer GAMUNEX-C FlexBag at the minimum infusion rate practicable and discontinue GAMUNEX-C FlexBag if renal function deteriorates.
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity
Severe hypersensitivity reactions may occur with IGIV products, including GAMUNEX-C FlexBag. In case of hypersensitivity, discontinue GAMUNEX-C FlexBag infusion immediately and institute appropriate treatment. Have medications such as epinephrine available for immediate treatment of acute hypersensitivity reaction.
GAMUNEX-C FlexBag contains trace amounts of IgA (average 46 micrograms/mL). Patients with known antibodies to IgA may have a greater risk of developing potentially severe hypersensitivity and anaphylactic reactions. It is contraindicated in IgA deficient patients with antibodies against IgA and history of hypersensitivity reaction. [see Contraindications (4)]
5.2 Renal Failure
Acute renal dysfunction/failure, acute tubular necrosis, proximal tubular nephropathy, osmotic nephrosis and death may occur upon use of IGIV products, especially those containing sucrose. GAMUNEX-C FlexBag does not contain sucrose. Ensure that patients are not volume depleted prior to the initiation of the infusion of GAMUNEX-C FlexBag. Periodic monitoring of renal function and urine output is particularly important in patients judged to have a potential increased risk for developing acute renal failure. Assess renal function, including measurement of blood urea nitrogen (BUN)/serum creatinine, prior to the initial infusion of GAMUNEX-C FlexBag and again at appropriate intervals thereafter. If renal function deteriorates, consider discontinuation of GAMUNEX-C FlexBag. [see Patient Counseling Information (17)] For patients judged to be at risk for developing renal dysfunction, including patients with any degree of pre-existing renal insufficiency, diabetes mellitus, age greater than 65, volume depletion, sepsis, paraproteinemia, or patients receiving known nephrotoxic drugs, administer GAMUNEX-C FlexBag at the minimum infusion rate practicable [less than 8 mg/kg/min (0.08 mL/kg/min)]. [see Dosage and Administration (2.3)]
5.3 Hyperproteinemia, Increased Serum Viscosity, and Hyponatremia
Hyperproteinemia, increased serum viscosity and hyponatremia may occur in patients receiving IGIV treatment, including GAMUNEX-C FlexBag. It is clinically critical to distinguish true hyponatremia from a pseudohyponatremia that is associated with concomitant decreased calculated serum osmolality or elevated osmolar gap, because treatment aimed at decreasing serum free water in patients with pseudohyponatremia may lead to volume depletion, a further increase in serum viscosity and a possible predisposition to thrombosis.
5.4 Thrombosis
Thrombosis may occur following treatment with immune globulin products, including GAMUNEX-C FlexBag. Risk factors may include: advanced age, prolonged immobilization, hypercoagulable conditions, history of venous or arterial thrombosis, use of estrogens, indwelling central vascular catheters, hyperviscosity, and cardiovascular risk factors. Thrombosis may occur in the absence of known risk factors.
Consider baseline assessment of blood viscosity in patients at risk for hyperviscosity, including those with cryoglobulins, fasting chylomicronemia/markedly high triacylglycerols (triglycerides), or monoclonal gammopathies. For patients at risk of thrombosis, administer GAMUNEX-C FlexBag at the minimum dose and infusion rate practicable. Ensure adequate hydration in patients before administration. Monitor for signs and symptoms of thrombosis and assess blood viscosity in patients at risk for hyperviscosity. [see Boxed Warning, Dosage and Administration (2.3), Patient Counseling Information (17)]
5.5 Aseptic Meningitis Syndrome (AMS)
AMS may occur infrequently with IGIV treatment, including GAMUNEX-C FlexBag. Discontinuation of IGIV treatment has resulted in remission of AMS within several days without sequelae. The syndrome usually begins within several hours to two days following IGIV treatment. AMS is characterized by the following symptoms and signs: severe headache, nuchal rigidity, drowsiness, fever, photophobia, painful eye movements, nausea and vomiting. Cerebrospinal fluid (CSF) studies are frequently positive with pleocytosis up to several thousand cells per cu mm, predominantly from the granulocytic series, and with elevated protein levels up to several hundred mg/dL, but negative culture results. Conduct a thorough neurological examination on patients exhibiting such symptoms and signs including CSF studies, to rule out other causes of meningitis. AMS may occur more frequently in association with high doses (2 g/kg) and/or rapid infusion of IGIV.
5.6 Hemolysis
GAMUNEX-C FlexBag may contain blood group antibodies which may act as hemolysins and induce in vivo coating of red blood cells (RBCs) with immunoglobulin, causing a positive direct antiglobulin reaction and hemolysis. Delayed hemolytic anemia can develop subsequent to IGIV therapy due to enhanced RBC sequestration, and acute hemolysis consistent with intravascular hemolysis, has been reported. [see Adverse Reactions (6)]
The following risk factors may be related to the development of hemolysis: high doses (e.g., ≥ 2 grams/kg, single administration or divided over several days) and non-O blood group. Underlying inflammatory state in an individual patient may increase the risk of hemolysis, but its role is uncertain.
Closely monitor patients for clinical signs and symptoms of hemolysis [see Warnings and Precautions (5.11)], particularly patients with risk factors noted above and those with pre-existing anemia and/or cardiovascular or pulmonary compromise. Consider appropriate laboratory testing in higher risk patients, including measurement of hemoglobin or hematocrit prior to infusion and within approximately 36 hours and again 7 to 10 days post infusion. If clinical signs and symptoms of hemolysis or a significant drop in hemoglobin or hematocrit have been observed, perform additional confirmatory laboratory testing. If transfusion is indicated for patients who develop hemolysis with clinically compromising anemia after receiving IGIV, perform adequate cross-matching to avoid exacerbating on-going hemolysis.
5.7 Transfusion-related Acute Lung Injury (TRALI)
Noncardiogenic pulmonary edema may occur in patients following treatment with IGIV products, including GAMUNEX-C FlexBag. TRALI is characterized by severe respiratory distress, pulmonary edema, hypoxemia, normal left ventricular function, and fever. Symptoms typically occur within 1 to 6 hours after treatment.
Monitor patients for pulmonary adverse reactions. [see Patient Counseling Information (17)] If TRALI is suspected, perform appropriate tests for the presence of anti-neutrophil and anti-HLA antibodies in both the product and patient serum. TRALI may be managed using oxygen therapy with adequate ventilatory support.
5.8 Volume Overload
The high dose regimen (1 g/kg x 1-2 days) is not recommended for individuals with expanded fluid volumes or where fluid volume may be a concern.
5.9 Transmission of Infectious Agents
Because GAMUNEX-C FlexBag is made from human blood, it may carry a risk of transmitting infectious agents, e.g., viruses, the variant Creutzfeldt-Jakob disease (vCJD) agent and, theoretically, the Creutzfeldt-Jakob disease (CJD) agent. No cases of transmission of viral diseases, vCJD or CJD have ever been identified for GAMUNEX-C FlexBag. ALL infections suspected by a physician possibly to have been transmitted by this product should be reported by the physician or other healthcare provider to Grifols Therapeutics LLC [1-800-520-2807].
5.10 Monitoring: Laboratory Tests
- Periodic monitoring of renal function and urine output is particularly important in patients judged to be at increased risk of developing acute renal failure. Assess renal function, including measurement of BUN and serum creatinine, before the initial infusion of GAMUNEX-C FlexBag and at appropriate intervals thereafter.
- Consider baseline assessment of blood viscosity in patients at risk for hyperviscosity, including those with cryoglobulins, fasting chylomicronemia/markedly high triacylglycerols (triglycerides), or monoclonal gammopathies, because of the potentially increased risk of thrombosis.
- If signs and/or symptoms of hemolysis are present after an infusion of GAMUNEX-C FlexBag, perform appropriate laboratory testing for confirmation.
- If TRALI is suspected, perform appropriate tests for the presence of anti-neutrophil antibodies and anti-HLA antibodies in both the product and patient’s serum.
5.11 Interference with Laboratory Tests
After infusion of IgG, the transitory rise of the various passively transferred antibodies in the patient’s blood may yield positive serological testing results, with the potential for misleading interpretation. Passive transmission of antibodies to erythrocyte antigens (e.g., A, B, and D) may cause a positive direct or indirect antiglobulin (Coombs) test.
-
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of one drug cannot be directly compared to rates in other clinical trials of another drug and may not reflect the rates observed in clinical practice.
Primary Immunodeficiency (PI)
The most serious adverse event observed in clinical study subjects receiving GAMUNEX-C IV for PI was an exacerbation of autoimmune pure red cell aplasia in one subject.
In four different clinical trials to study PI, out of 157 subjects treated with GAMUNEX-C, 4 subjects discontinued due to the following adverse events: Coombs negative hypochromic anemia, autoimmune pure red cell aplasia, arthralgia/hyperhidrosis/fatigue/myalgia/nausea and migraine.
In a study of 87 subjects, 9 subjects in each treatment group were pretreated with non-steroidal medication prior to infusion, such as diphenhydramine and acetaminophen.
Table 1 lists the adverse reactions reported by at least 5% of subjects during the 9-month treatment.
Table 1: Adverse Reactions Occurring in ≥ 5% of Subjects * An adverse reaction is an adverse event that meets any of the following 3 criteria: (a) that began during or within 72 hours of the end of product infusion, (b) that were considered at least possibly related by either the investigator or the applicant, and/or (c) whose causality assessment by the investigator was missing or indeterminate. Adverse Reactions GAMUNEX®-C GAMIMUNE® N, 10% No. of subjects: 87 No. of subjects: 85 No. of subjects with adverse reaction (percentage of all subjects) No. of subjects with adverse reaction (percentage of all subjects) Cough increased 27 (31.0%) 25 (29.4%) Rhinitis 21 (24.1%) 24 (28.2%) Headache 13 (14.9%) 17 (20.0%) Pharyngitis 14 (16.1%) 16 (18.8%) Asthma 13 (14.9%) 10 (11.8%) Fever 6 (6.9%) 10 (11.8%) Nausea 10 (11.5%) 9 (10.6%) Diarrhea 6 (6.9%) 9 (10.6%) Sinusitis 5 (5.7%) 6 (7.1%) Table 2 lists the frequency of adverse reactions (as defined for Table 1), which were reported by at least 5% of subjects.
Table 2: Adverse Reactions Frequency Adverse Reactions GAMUNEX®-C
No. of infusions: 825
Number (percentage of all infusions)GAMIMUNE® N, 10%
No. of infusions: 865
Number (percentage of all infusions)Cough increased 40 (4.8%) 47 (5.4%) Rhinitis 34 (4.1%) 44 (5.1%) Headache 17 (2.1%) 24 (2.8%) Pharyngitis 20 (2.4%) 24 (2.8%) Fever 8 (1.0%) 20 (2.3%) Asthma 17 (2.1%) 12 (1.4%) Diarrhea 10 (1.2%) 10 (1.2%) Nausea 10 (1.2%) 10 (1.2%) Sinusitis 6 (0.7%) 7 (0.8%) The mean number of adverse reactions per infusion that occurred during or within 72 hours of the end of product infusion was 0.33 for the GAMUNEX-C and 0.39 for the GAMIMUNE® N, 10% [Immune Globulin Intravenous (Human), 10%] treatment group.
In all three trials in primary humoral immunodeficiencies, the maximum infusion rate was 0.08 mL/kg/min (8 mg/kg/min). The infusion rate was reduced for 11 of 222 exposed subjects (7 GAMUNEX-C, 4 GAMIMUNE N, 10%) at 17 occasions. In most instances, mild to moderate hives/urticaria, itching, pain or reaction at infusion site, anxiety or headache was the main reason. There was one case of severe chills. There were no anaphylactic or anaphylactoid reactions to GAMUNEX-C or GAMIMUNE N, 10% in clinical trials.
In the IV efficacy and safety study, serum samples were drawn to monitor the virus safety at baseline and one week after the first infusion of IGIV (for parvovirus B19), eight weeks after first and fifth infusion of IGIV (for hepatitis C, hepatitis B, and HIV-1), 16 weeks after the first and fifth infusion of IGIV (for hepatitis C) and at any time of premature discontinuation of the study (for hepatitis C, hepatitis B, HIV-1, and parvovirus B19). Viral markers of hepatitis C, hepatitis B, HIV-1, and parvovirus B19 were monitored by nucleic acid testing (NAT, Polymerase Chain Reaction [PCR]) and serological testing. There were no treatment related emergent findings of virus transmission for either GAMUNEX-C or GAMIMUNE N, 10%.
Idiopathic Thrombocytopenic Purpura (ITP)
In two different clinical trials to study ITP, out of 76 subjects treated with GAMUNEX-C, 2 subjects discontinued due to the following adverse reactions: Hives and Headache/Fever/Vomiting.
One subject, a 10-year-old boy, died suddenly from myocarditis 50 days after his second infusion of GAMUNEX-C. The death was judged to be unrelated to GAMUNEX-C.
No pre-medication with corticosteroids was permitted by the protocol. Twelve ITP subjects treated in each treatment group were pretreated with medication prior to infusion. Generally, diphenhydramine and/or acetaminophen were used. More than 90% of the observed drug related adverse events were of mild to moderate severity and of transient nature.
The infusion rate was reduced for 4 of the 97 exposed subjects (1 GAMUNEX-C, 3 GAMIMUNE N, 10%) on 4 occasions. Mild to moderate headache, nausea, and fever were the reported reasons.
Table 3 lists the adverse reactions (as defined for Table 1) reported by at least 5% of subjects during the 3-month efficacy and safety study.
Table 3: Adverse Reactions Occurring in ≥ 5% of Subjects Adverse Reaction GAMUNEX®-C
No. of subjects: 48
Number (percentage of all subjects)GAMIMUNE® N, 10%
No. of subjects: 49
Number (percentage of all subjects)Headache 25 (52.1%) 26 (53.1%) Vomiting 6 (12.5%) 8 (16.3%) Ecchymosis 7 (14.6%) 2 (4.1%) Fever 6 (12.5%) 6 (12.2%) Nausea 6 (12.5%) 5 (10.2%) Rash 4 (8.3%) 0 Abdominal pain 3 (6.3%) 3 (6.1%) Back pain 3 (6.3%) 2 (4.1%) Dyspepsia 3 (6.3%) 0 Asthenia 2 (4.2%) 3 (6.1%) Dizziness 2 (4.2%) 3 (6.1%) Serum samples were drawn to monitor the virus safety of the ITP subjects at baseline, nine days after the first infusion (for parvovirus B19), and 3 months after the first infusion of IGIV and at any time of premature discontinuation of the study. Viral markers of hepatitis C, hepatitis B, HIV-1, and parvovirus B19 were monitored by nucleic acid testing (NAT, PCR), and serological testing. There were no treatment related emergent findings of virus transmission for either GAMUNEX-C or GAMIMUNE N, 10%.
Chronic Inflammatory Demyelinating Polyneuropathy (CIDP)
In the CIDP efficacy and safety study, 113 subjects were exposed to GAMUNEX-C and 95 were exposed to Placebo. [see Clinical Studies (14)] As a result of the study design, the drug exposure with GAMUNEX-C was almost twice that of Placebo, with 1096 GAMUNEX-C infusions versus 575 Placebo infusions. Therefore, adverse reactions are reported per infusion (represented as frequency) to correct for differences in drug exposure between the 2 groups. The majority of loading-doses were administered over 2 days. The majority of maintenance-doses were administered over 1 day. Infusions were administered in the mean over 2.7 hours.
Table 4 shows the numbers of subjects per treatment group in the CIDP clinical trial, and the reason for discontinuation due to adverse events.
Table 4: Reasons for Discontinuation Due to Adverse Events Number of Subjects Number of Subjects Discontinued
due to Adverse EventsAdverse Event GAMUNEX®-C 113 3 (2.7%) Urticaria, Dyspnea,
BronchopneumoniaPlacebo 95 2 (2.1%) Cerebrovascular Accident,
Deep Vein ThrombosisThe most common adverse reactions with GAMUNEX-C were headache and pyrexia. Table 5 lists adverse reactions (as defined for Table 1) reported by at least 5% of subjects in any treatment group.
Table 5: Adverse Reactions Occurring in ≥ 5% of Subjects - * Reported in ≥ 5% of subjects in any treatment group.
- † Calculated by the total number of adverse reactions divided by the number of infusions received (1096 for GAMUNEX-C and 575 for Placebo).
GAMUNEX®-C
No. of subjects: 113Placebo
No. of subjects: 95MedDRA
Preferred Term*No. of
Subjects
(%)No. of
Adverse
ReactionsIncidence
density†No. of
Subjects
(%)No. of
Adverse
ReactionsIncidence
density†Headache 35 (31.0%) 50 0.046 7 (7.4%) 9 0.016 Pyrexia 15 (13.3%) 27 0.025 0 0 Hypertension 10 (8.8%) 19 0.017 3 (3.2%) 3 0.005 Chills 9 (8.0%) 10 0.009 0 (0%) 0 Nausea 7 (6.2%) 9 0.008 3 (3.2%) 3 0.005 Rash 7 (6.2%) 10 0.009 1 (1.1%) 1 0.002 Arthralgia 6 (5.3%) 7 0.006 0 0 Asthenia 6 (5.3%) 6 0.005 1 (1.1%) 2 0.003 The most serious adverse reaction observed in clinical study subjects receiving GAMUNEX-C for CIDP was pulmonary embolism (PE) in one subject with a history of PE.
Laboratory Abnormalities
During the course of the clinical program, ALT and AST elevations were identified in some subjects.
- For ALT, in the IV PI study treatment emergent elevations above the upper limit of normal were transient and observed among 14/80 (18%) of subjects in the GAMUNEX-C group versus 5/88 (6%) of subjects in the GAMIMUNE N, 10% group (p = 0.026).
- In the ITP study which employed a higher dose per infusion, but a maximum of only two infusions, the reverse finding for elevation of ALT was observed among 3/44 (7%) of subjects in the GAMUNEX-C group versus 8/43 (19%) of subjects in the GAMIMUNE N, 10% group (p = 0.118).
- In the CIDP study, 15/113 (13%) of subjects in the GAMUNEX-C group and 7/95 (7%) in the Placebo group (p=0.168) had a treatment emergent transient elevation of ALT.
Elevations of ALT were generally mild (< 3 times upper limit of normal), transient, and were not associated with obvious symptoms of liver dysfunction.
GAMUNEX-C may contain low levels of anti-Blood Group A and B antibodies primarily of the IgG4 class. Direct antiglobulin tests (DAT or direct Coombs tests), which are carried out in some centers as a safety check prior to red blood cell transfusions, may become positive temporarily. There were 2 cases of hemolytic anemia across these clinical trials. One hemolytic event not associated with positive DAT findings was observed in the IV PI study in a woman with common variable immune deficiency and B12 deficiency (pernicious anemia) at a dose of (450 mg/kg). The other hemolytic event occurred in the CIDP study in a subject with positive DAT at a dose of 1g/kg.
6.2 Postmarketing Experience
Because adverse reactions are voluntarily reported post-approval from a population of uncertain size, it is not always possible to reliably estimate their frequencies or establish a causal relationship to product exposure.
The following adverse reactions have been identified during post-approval use of IGIV products, including GAMUNEX-C:
- Infusion Reactions: Hypersensitivity (e.g., anaphylaxis), tachycardia, malaise, flushing, or other skin reactions, chest discomfort, rigors, and changes in blood pressure
- Renal: Acute renal dysfunction/failure, osmotic nephropathy
- Respiratory: Apnea, Acute Respiratory Distress Syndrome (ARDS), TRALI, cyanosis, hypoxemia, pulmonary edema, bronchospasm
- Cardiovascular: Cardiac arrest, thromboembolism, vascular collapse, hypotension
- Neurological: Coma, loss of consciousness, seizures/convulsions, tremor, aseptic meningitis
- Integumentary: Stevens-Johnson syndrome, epidermolysis, erythema multiforme, dermatitis (e.g., bullous dermatitis)
- Hematologic: Pancytopenia, leukopenia, hemolysis, hemolytic anemia, positive direct antiglobulin (Coombs test)
- General/Body as a Whole: Rigors
- Gastrointestinal: Hepatic dysfunction
-
7 DRUG INTERACTIONS
GAMUNEX-C FlexBag may be diluted with 5% dextrose in water (D5/W). Do not dilute with saline. Admixtures of GAMUNEX-C FlexBag with other drugs and intravenous solutions have not been evaluated. It is recommended that GAMUNEX-C FlexBag be administered separately from other drugs or medications which the patient may be receiving. The product should not be mixed with IGIVs from other manufacturers.
The infusion line may be flushed before and after administration of GAMUNEX-C FlexBag with 5% dextrose in water (D5/W) or 0.9% sodium chloride for injection.
Avoid simultaneous administration of GAMUNEX-C FlexBag and Heparin through a single lumen delivery device due to GAMUNEX-C FlexBag, Heparin incompatibilities. Flush Heparin Lock (Hep-Lock) through which GAMUNEX-C FlexBag was administered with 5% dextrose in water (D5/W) or 0.9% sodium chloride for injection, and do not flush with Heparin.
Various passively transferred antibodies in immunoglobulin preparations can confound the results of serological testing.
Passive transfer of antibodies may transiently interfere with the immune response to live virus vaccines such as measles, mumps, rubella and varicella. Inform the immunizing physician of recent therapy with GAMUNEX-C FlexBag so that appropriate measures may be taken. [see Patient Counseling Information (17)]
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no data with GAMUNEX-C FlexBag use in pregnant women to inform a drug-associated risk. Animal reproduction studies have not been conducted with GAMUNEX-C FlexBag. It is not known whether GAMUNEX-C FlexBag can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. GAMUNEX-C FlexBag should be given to a pregnant woman only if clearly needed. In the U.S. general population, the estimated background risk of major birth defect and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
8.2 Lactation
Risk Summary
There is no information regarding the presence of GAMUNEX-C FlexBag in human milk, the effect on the breastfed infant, and the effects on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for GAMUNEX-C FlexBag and any potential adverse effects on the breastfed infant from GAMUNEX-C FlexBag or from the underlying maternal condition.
8.4 Pediatric Use
Primary Humoral Immunodeficiency (PI)
The safety and effectiveness of GAMUNEX-C have been established in pediatric patients 2 years of age and older for primary humoral immunodeficiency.
GAMUNEX-C was evaluated in 18 pediatric subjects (age range 0-16 years). Twenty-one percent of PI subjects exposed to GAMUNEX-C were children. Pharmacokinetics, safety and efficacy were similar to those in adults with the exception that vomiting was more frequently reported in pediatrics (3 of 18 subjects). No pediatric-specific dose requirements were necessary to achieve serum IgG levels.
Idiopathic Thrombocytopenic Purpura (ITP)
The safety and effectiveness of GAMUNEX-C have been established in pediatric patients for idiopathic thrombocytopenic purpura. GAMUNEX-C was evaluated in 12 pediatric subjects with acute ITP. Twenty-five percent of the acute ITP subjects exposed to GAMUNEX-C were children. Pharmacokinetics, safety and efficacy were similar to those in adults with the exception that fever was more frequently reported in pediatrics (6 of 12 subjects). No pediatric-specific dose requirements were necessary to achieve serum IgG levels. One subject, a 10-year-old boy, died suddenly from myocarditis 50 days after his second infusion of GAMUNEX-C. The death was judged to be unrelated to GAMUNEX-C.
Chronic Inflammatory Demyelinating Polyneuropathy (CIDP)
The safety and effectiveness of GAMUNEX-C FlexBag have not been established in pediatric subjects with CIDP.
8.5 Geriatric Use
Use caution when administering GAMUNEX-C FlexBag to patients age 65 and over who are at increased risk for thrombosis or renal insufficiency. [see Boxed Warning, Warnings and Precautions (5.2, 5.4)] Do not exceed recommended doses, and administer GAMUNEX-C FlexBag at the minimum infusion rate practicable. Clinical studies of GAMUNEX-C did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects.
- 10 OVERDOSAGE
-
11 DESCRIPTION
GAMUNEX-C FlexBag is a ready-to-use sterile, non-pyrogenic solution of human immune globulin protein for intravenous administration. GAMUNEX-C FlexBag is clear to opalescent, and colorless to pale yellow. GAMUNEX-C FlexBag consists of 9%–11% protein in 0.16–0.24 M glycine. Not less than 98% of the protein has the electrophoretic mobility of gamma globulin. The main component of GAMUNEX-C FlexBag is IgG (≥ 98%) with a sub-class distribution of IgG1, IgG2, IgG3 and IgG4 of approximately 62.8%, 29.7%, 4.8% and 2.7% respectively. The distribution of IgG subclasses is similar to that found in normal serum.
GAMUNEX-C FlexBag contains trace levels of fragments, IgA (average 0.046 mg/mL), and IgM. GAMUNEX-C FlexBag doses of 1 g/kg correspond to a glycine dose of 0.15 g/kg. While toxic effects of glycine administration have been reported, the doses and rates of administration were 3–4 fold greater than those for GAMUNEX-C FlexBag. In another study it was demonstrated that intravenous bolus doses of 0.44 g/kg glycine were not associated with serious adverse effects. Caprylate is a saturated medium-chain (C8) fatty acid of plant origin. Medium chain fatty acids are considered to be essentially non-toxic. Human subjects receiving medium chain fatty acids parenterally have tolerated doses of 3.0 to 9.0 g/kg/day for periods of several months without adverse effects. Residual caprylate concentrations in the final container are no more than 0.216 g/L (1.3 mmol/L). The measured buffer capacity is 35 mEq/L (0.35 mEq/g protein) and the osmolality is approximately 258 mOsmol/kg solvent, which is close to physiological osmolality (285-295 mOsmol/kg). A dose of 1 g/kg body weight therefore represents an acid load of 0.35 mEq/kg body weight. The total buffering capacity of whole blood in a normal individual is 45–50 mEq/L of blood, or 3.6 mEq/kg body weight. Thus, the acid load delivered with a dose of 1 g/kg of GAMUNEX-C FlexBag would be neutralized by the buffering capacity of whole blood alone, even if the dose was infused instantaneously. The pH of GAMUNEX-C FlexBag is 4.0–4.5. GAMUNEX-C FlexBag contains no preservative. GAMUNEX-C FlexBag is not made with natural rubber latex.
GAMUNEX-C FlexBag is made from large pools of human plasma by a combination of cold ethanol fractionation, caprylate precipitation and filtration, and anion-exchange chromatography. Isotonicity is achieved by the addition of glycine. GAMUNEX-C FlexBag is incubated in the final container (at the low pH of 4.0–4.3). The product is intended for intravenous administration.
The capacity of the manufacturing process to remove and/or inactivate enveloped and non-enveloped viruses has been validated by laboratory spiking studies on a scaled down process model, using the following enveloped and non-enveloped viruses: human immunodeficiency virus, type I (HIV-1) as the relevant virus for HIV-1 and HIV–2; bovine viral diarrhea virus (BVDV) as a model for hepatitis C virus; pseudorabies virus (PRV) as a model for large enveloped DNA viruses (e.g., herpes viruses); Reovirus type 3 (Reo) as a model for non-enveloped viruses and for its resistance to physical and chemical inactivation; hepatitis A virus (HAV) as relevant non-enveloped virus, and porcine parvovirus (PPV) as a model for human parvovirus B19.
Overall virus reduction was calculated only from steps that were mechanistically independent from each other and truly additive. In addition, each step was verified to provide robust virus reduction across the production range for key operating parameters.
Table 6: Log10 Virus Reduction - * C/I - Interference by caprylate precluded determination of virus reduction for this step. Although removal of viruses is likely to occur at the caprylate precipitation/depth filtration step, BVDV is the only enveloped virus for which reduction is claimed. The presence of caprylate prevents detection of other, less resistant enveloped viruses and therefore their removal cannot be assessed.
- † NA - Not Applicable: This step has no effect on non-enveloped viruses.
- ‡ Some mechanistic overlap occurs between depth filtration and other steps. Therefore, Grifols Therapeutics LLC has chosen to exclude this step from the global virus reduction calculations.
- § CAP - The presence of caprylate in the process at this step prevents detection of enveloped viruses, and their removal cannot be assessed.
- ¶ M/I - Interference by the process intermediate matrix precluded determination of virus removal capacity for this step.
- # Sum of reduction factors greater than or equal to 1 log10.
Process Step Log10 Virus Reduction Enveloped Viruses Non-enveloped Viruses HIV PRV BVDV Reo HAV PPV Caprylate Precipitation/Depth Filtration C/I* C/I* 2.7 ≥ 3.5 ≥ 3.6 4.0 Caprylate Incubation ≥ 4.5 ≥ 4.6 ≥ 4.5 NA† NA† NA† Depth Filtration‡ CAP§ CAP§ CAP§ ≥ 4.3 ≥ 2.0 3.3 Column Chromatography ≥ 3.0 ≥ 3.3 4.0 ≥ 4.0 ≥ 1.4 4.2 Nanofiltration ≥ 3.7 M/I¶ ≥ 4.1 ≥ 1.8 M/I¶ < 1.0 Low pH Incubation ≥ 6.5 ≥ 4.3 ≥ 5.1 NA† NA† NA† Global Reduction# ≥ 17.7 ≥ 12.2 ≥ 20.4 ≥ 9.3 ≥ 5.0 8.2 Additionally, the manufacturing process was investigated for its capacity to decrease the infectivity of an experimental agent of transmissible spongiform encephalopathy (TSE), considered as a model for the vCJD and CJD agents.
Several of the individual production steps in the GAMUNEX-C FlexBag manufacturing process have been shown to decrease TSE infectivity of that experimental model agent. TSE reduction steps include two depth filtrations (in sequence, a total of ≥ 6.6 log10). These studies provide reasonable assurance that low levels of CJD/vCJD agent infectivity, if present in the starting material, would be removed.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Primary Humoral Immunodeficiency (PI)
GAMUNEX-C FlexBag supplies a broad spectrum of opsonic and neutralizing IgG antibodies against bacterial, viral, parasitic, and mycoplasmal agents, and their toxins. The mechanism of action in PI has not been fully elucidated.
Idiopathic Thrombocytopenic Purpura (ITP)
The mechanism of action of high doses of immunoglobulins in the treatment of ITP has not been fully elucidated.
Chronic Inflammatory Demyelinating Polyneuropathy (CIDP)
The precise mechanism of action in CIDP has not been fully elucidated.
12.3 Pharmacokinetics
Two pharmacokinetic crossover trials were carried out with GAMUNEX-C in 44 subjects with Primary Humoral Immunodeficiency to assess intravenous vs subcutaneous administration. In the first study, a single sequence, open-label, crossover trial in adults and adolescents, the pharmacokinetics, safety, and tolerability of subcutaneously (SC) administered GAMUNEX-C in subjects with PI were evaluated. A total of 32 and 26 subjects received GAMUNEX-C as IV or SC for PK study, respectively, of whom 3 were adolescents. Subjects received GAMUNEX-C 200-600 mg/kg IV every 3-4 weeks for at least 3 months, at which time they entered the IV phase of the study. Subjects were crossed over to weekly SC infusions. The weekly SC dose was determined by multiplying the total IV dose by 1.37 and dividing the resultant new total dose by 3 or 4 depending on the previous IV interval.
In the second study, a single sequence, open-label, crossover trial, the pharmacokinetics, safety, and tolerability of SC administered GAMUNEX-C were evaluated in children and adolescents. The design of the study was essentially the same as above. A total of 11 subjects received GAMUNEX-C as IV and 10 received GAMUNEX-C as SC for PK analysis. Age groups were as follows: age 2 to 5 years, [N = 1 both phases]; 6 to 11 years, [N = 5 completing IV phase, N = 4 evaluated in SC phase]; 12-16 years: [N = 5 both phases].
The pharmacokinetic parameters of GAMUNEX-C, measured as total IgG for intravenous administration are shown in Table 7.
Table 7: PK Parameters Following IV Administration of GAMUNEX®-C by Age - * SD - standard deviation; NA - not applicable.
Source: studies 060001, T5004-401Age Group Statistics t1/2
(hr)AUC(0-t)
(hr*mg/mL)AUC(0-tau)
(hr*mg/mL)CL(0-t)
(mL/hr/kg)Vss
(mL/kg)2 – 5 years
N = 1Mean 1038.5 7479.0 7499.0 0.05 82.0 SD* NA* NA* NA* NA* NA* 6 – 11 years
N = 5Mean 758.5 5953.6 6052.6 0.09 94.8 SD* 138.0 1573.8 1333.6 0.03 17.7 12 – 16 years
N = 8Mean 717.9 8131.9 8009.5 0.07 73.3 SD* 170.1 1173.4 1358.8 0.02 17.2 ≥ 17 years
N = 29Mean 720.6 7564.9 7524.8 0.06 65.5 SD* 130.9 1190.7 1183.1 0.02 18.7 The mean trough concentrations (mean Ctrough) of total IgG following IV administration are presented in Table 8 for both studies.
- * SD - standard deviation; NA - not applicable.
-
14 CLINICAL STUDIES
Primary Humoral Immunodeficiency (PI)
In a randomized, double-blind, parallel group clinical trial with 172 subjects with primary humoral immunodeficiencies GAMUNEX-C was demonstrated to be at least as efficacious as GAMIMUNE N, 10% in the prevention of any infection, i.e., validated plus clinically defined, non-validated infections of any organ system, during a nine month treatment period. Twenty-six subjects were excluded from the Per Protocol analysis (2 due to non-compliance and 24 due to protocol violations). The analysis for efficacy was based on the annual rate of bacterial infections, pneumonia, acute sinusitis and acute exacerbations of chronic sinusitis.
Table 9: Efficacy Results Per Protocol Analysis - * Validated infections plus clinically defined, non-validated infections.
No. of Subjects with at Least One Infection (%) GAMUNEX®-C
(n=73)GAMIMUNE® N, 10%
(n=73)Mean Difference
(90% Confidence Interval)p-Value Validated Infections 9 (12%) 17 (23%) -0.117
(-0.220, -0.015)0.06 Acute Sinusitis 4 (5%) 10 (14%) Exacerbation of
Chronic Sinusitis5 (7%) 6 (8%) Pneumonia 0 (0%) 2 (3%) Any Infection* 56 (77%) 57 (78%) -0.020
(-0.135, 0.096)0.78 The annual rate of validated infections (Number of Infections/year/subject) was 0.18 in the group treated with GAMUNEX-C and 0.43 in the group treated with GAMIMUNE N, 10% (p=0.023). The annual rates for any infection (validated plus clinically defined, non-validated infections of any organ system) were 2.88 and 3.38, respectively (p=0.300).
Primary Humoral Immunodeficiency (ITP)
A double-blind, randomized, parallel group clinical trial with 97 ITP subjects was carried out to test the hypothesis that GAMUNEX-C was at least as effective as GAMIMUNE N, 10% in raising platelet counts from less than or equal to 20 x109/L to more than 50 x109/L within 7 days after treatment with 2 g/kg IGIV. Twenty-four percent of the subjects were less than or equal to 16 years of age.
GAMUNEX-C was demonstrated to be at least as effective as GAMIMUNE N, 10% in the treatment of adults and children with acute or chronic ITP.
Table 10: Platelet Response of Per Protocol Analysis Number of Responders (percent of all subjects) GAMUNEX®-C
(n=39)GAMIMUNE® N, 10%
(n=42)Mean Difference
(90% Confidence Interval)By Day 7 35 (90%) 35 (83%) 0.075
(-0.037, 0.186)By Day 23 35 (90%) 36 (86%) 0.051
(-0.058, 0.160)Sustained for 7 days 29 (74%) 25 (60%) 0.164
(0.003, 0.330)A trial was conducted to evaluate the clinical response to rapid infusion of GAMUNEX-C in patients with ITP. The study involved 28 chronic ITP subjects, wherein the subjects received 1 g/kg GAMUNEX-C on three occasions for treatment of relapses. The infusion rate was randomly assigned to 0.08, 0.11, or 0.14 mL/kg/min (8, 11 or 14 mg/kg/min). Pre-medication with corticosteroids to alleviate infusion-related intolerability was not permitted. Pre-treatment with antihistamines, anti-pyretics and analgesics was permitted. The average dose was approximately 1 g/kg body weight at all three prescribed rates of infusion (0.08, 0.11 and 0.14 mL/kg/min). All patients were administered each of the three planned infusions except seven subjects. Based on 21 patients per treatment group, the a posteriori power to detect twice as many drug-related adverse events between groups was 23%. Of the seven subjects that did not complete the study, five did not require additional treatment, one withdrew because he refused to participate without concomitant medication (prednisone) and one experienced an adverse event (hives); however, this was at the lowest dose rate level (0.08 mL/kg/min).
Chronic Inflammatory Demyelinating Polyneuropathy (CIDP)
A multi-center, randomized, double-blind, Placebo-controlled trial (The Immune Globulin Intravenous (Human), 10% Caprylate/Chromatography Purified CIDP Efficacy or ICE study) was conducted with GAMUNEX-C. This study included two separately randomized periods to assess whether GAMUNEX-C was more effective than Placebo for the treatment of CIDP (assessed in the Efficacy Period for up to 24 weeks) and whether long-term administration of GAMUNEX-C could maintain long-term benefit (assessed in the 24 week Randomized Withdrawal Period).
In the Efficacy Period, there was a requirement for Rescue (crossover) to the alternate study drug if the subject did not improve and maintain this improvement until the end of the 24 week treatment period. Subjects entering the Rescue phase followed the same dosing and schedule as in the Efficacy period. Any subject who was rescued (crossed over) and did not improve and maintain this improvement was withdrawn from the study.
Subjects who completed 24 weeks treatment in the Efficacy period or Rescue phase and responded to therapy were eligible for entry into a double-blind Randomized Withdrawal Period. Eligible subjects were re-randomized to GAMUNEX-C or Placebo. Any subject who relapsed was withdrawn from the study.
The Efficacy Period and the Rescue treatment started with a loading dose of 2 g/kg body weight of GAMUNEX-C or equal volume of Placebo given over 2-4 consecutive days. All other infusions (including the first infusion of the Randomized Withdrawal Period) were given as maintenance doses of 1 g/kg body weight (or equivalent volume of Placebo) every three weeks.
The Responder rates of the GAMUNEX-C and Placebo treatment groups were measured by the INCAT score. The INCAT (Inflammatory Neuropathy Cause and Treatment) scale is used to assess functional disability of both upper and lower extremities in demyelinating polyneuropathy. The INCAT scale has upper and lower extremity components (maximum of 5 points for upper (arm disability) and maximum of 5 points for lower (leg disability)) that add up to a maximum of 10-points (0 is normal and 10 is severely incapacitated). At the start of the efficacy portion of the study, the INCAT scores were as follows: Upper Extremity mean was 2.2 ± 1.0, and median was 2.0 with a range of 0 to 5; Lower Extremity mean was 1.9 ± 0.9, and median was 2.0 with a range of 1 to 5; Total Overall Score mean was 4.2 ± 1.4, and median was 4.0 with a range of 2 to 9. A Responder was defined as a subject with at least 1-point improvement from baseline in the adjusted INCAT score that was maintained through 24 weeks.
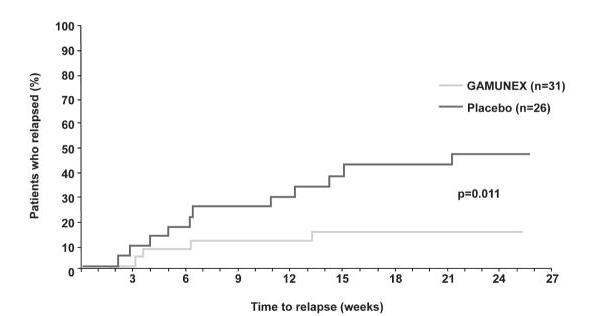
More subjects with CIDP responded to GAMUNEX-C: 28 of 59 subjects (47.5%) responded to GAMUNEX-C compared with 13 of 58 subjects (22.4%) administered Placebo (25% difference; 95% CI 7%-43%; p=0.006). The study included both subjects who were IGIV naïve and subjects who had previous IGIV experience. The outcome was influenced by the group of subjects who experienced prior therapy with IGIV, as shown by the outcomes table, below.
Time to relapse for the subset of 57 subjects who previously responded to GAMUNEX-C was evaluated: 31 were randomly reassigned to continue to receive GAMUNEX-C and 26 subjects were randomly reassigned to Placebo in the Randomized Withdrawal Period. Subjects who continued to receive GAMUNEX-C experienced a longer time to relapse versus subjects treated with Placebo (p=0.011). The probability of relapse was 13% with GAMUNEX-C versus 45% with Placebo (hazard ratio, 0.19; 95% confidence interval, 0.05, 0.70).
Table 11: Outcomes in Intent-to-Treat Population Efficacy Period - * p-value based on Fisher's exact method
Efficacy Period GAMUNEX®-C Placebo p-value* Responder Non-Responder Responder Non-Responder All Subjects 28/59 (47.5%) 31/59 (52.5%) 13/58 (22.4%) 45/58 (77.6%) 0.006 IGIV -
Naïve Subjects
17/39 (43.6%)
22/39 (56.4%)
13/46 (28.3%)
33/46 (71.7%)
0.174IGIV -
Experienced Subjects
11/20 (55.0%)
9/20 (45.0%)
0/12 (0%)
12/12 (100%)
0.002The following table shows outcomes for the Rescue Phase (which are supportive data):
Table 12: Outcomes in Rescue Phase - * p-value based on Fisher's exact method
Rescue Phase GAMUNEX®-C Placebo p-value* Success Failure Success Failure All Subjects 25/45
(55.6%)20/45
(44.4%)6/23
(26.1%)17/23
(73.9%)0.038 IGIV - Naïve Subjects 19/33
(57.6%)14/33
(42.4%)6/18
(33.3%)12/18
(66.7%)0.144 IGIV - Experienced Subjects 6/12
(50%)6/12
(50%)0/5
(0%)5/5
(100%)0.102 The following Kaplan-Meier curves show the outcomes for the Randomized Withdrawal Period:
-
16 HOW SUPPLIED/STORAGE AND HANDLING
GAMUNEX-C FlexBag is supplied in single-dose flexible containers with the labeled amount of functionally active IgG. GAMUNEX-C FlexBag, the flexible container, and the protective overwrap are not made with natural rubber latex. The flexible container is free of polyvinyl chloride (PVC), diethylhexyl phthalate (DEHP), or other plasticizers. The protective overwrap on each container contains no chloride, PVC or plasticizers. Each container is individually laser-etched with at least the lot number and expiration date.
GAMUNEX-C FlexBag is supplied in the following sizes:
Carton NDC Bags/Carton Size Grams Protein 13533-805-01 1 50 mL 5 13533-805-11 1 100 mL 10 13533-805-31 1 200 mL 20 13533-805-45 1 400 mL 40 - DO NOT FREEZE
- Keep the flexible container in the carton to protect from light. Keep the overwrap intact.
- GAMUNEX-C FlexBag may be stored for 36 months at 2-8°C (36-46°F) from the date of manufacture, AND product may be stored at temperatures not to exceed 25°C (77°F) for up to 6 months anytime during the 36 month shelf life, after which the product must be immediately used or discarded.
- Do not use after expiration date.
-
17 PATIENT COUNSELING INFORMATION
[see Boxed Warning and Warnings and Precautions]
Instruct patients to immediately report the following signs and symptoms to their healthcare provider:
- Decreased urine output, sudden weight gain, fluid retention/edema, and/or shortness of breath [see Warnings and Precautions (5.2)]
- Symptoms of thrombosis which may include: pain and/or swelling of an arm or leg with warmth over the affected area, discoloration of an arm or leg, unexplained shortness of breath, chest pain or discomfort that worsens on deep breathing, unexplained rapid pulse, numbness or weakness on one side of the body [see Warnings and Precautions (5.4)]
- Severe headache, neck stiffness, drowsiness, fever, sensitivity to light, painful eye movements, nausea, and vomiting [see Warnings and Precautions (5.5)]
- Increased heart rate, fatigue, yellowing of the skin or eyes, and dark-colored urine [see Warnings and Precautions (5.6)]
- Trouble breathing, chest pain, blue lips or extremities, and fever [see Warnings and Precautions (5.7)]
Inform patients that GAMUNEX-C FlexBag is made from human plasma and may contain infectious agents that can cause disease. While the risk GAMUNEX-C FlexBag can transmit an infectious agent has been reduced by screening plasma donors for prior exposure, testing donated plasma, and by inactivating or removing pathogens during manufacturing, patients should report any symptoms that concern them. [see Warnings and Precautions (5.9)]
Inform patients that GAMUNEX-C FlexBag can interfere with their immune response to live virus vaccines such as measles, mumps and rubella. Inform patients to notify their healthcare professional of this potential interaction when they are receiving vaccinations. [see Drug Interactions (7)]
Manufactured by:

Grifols Therapeutics LLC
Clayton, NC 27520 USA 3068256
U.S. License No. 1871
-
PACKAGE LABEL
10 g / 100 mL
NDC: 13533-805-11
Immune Globulin Injection (Human), 10%,
Caprylate/Chromatography Purified
gamunex-c flexbag
Solution for Infusion
Rx Only
For intravenous administration
GRIFOLS
The patient and physician should discuss the risks and benefits of this product.
Refer to package insert for dosage and administration and compatibility with other solutions.
Single-dose Flexible Container
Discard unused portion. Do not store after entry into container.
Contains no preservative.
Each milliliter (mL) contains approximately 100 mg of protein, not less than 98% of which is
immunoglobulin, and approximately 15 mg glycine.
Inspect the protective overwrap and do not use
if it shows any signs of damage or tampering.
Not Returnable for Credit or Exchange.
Grifols Therapeutics LLC
Clayton, NC 27520 USA
U.S. License No. 1871
GTIN 00313533805111
LOT B01K043422
EXP 19FEB2028
SN 1234567890123456
Contents: One single-dose flexible container of Gamunex-C
Sterile, Non-pyrogenic
Keep the overwrapped bag in the carton to protect from light
Prior to administration, check bag for leaks by squeezing firmly. If leaks are found, discard bag as sterility may be compromised.
Keep the overwrap intact. Do not use if turbid.
Gamunex-C may be stored for 36 months at 2°C to 8°C (36°F to 46°F) AND product may be stored at temperatures not to exceed 25°C (77°F) for up to 6 months anytime during the 36 month shelf life, after which the product must be immediately used or discarded. Do not freeze.
Date removed from refrigeration:

NDC: 13533-805-10
10 g / 100 mL
Immune Globulin Injection (Human), 10%,
Caprylate/Chromatography Purified
gamunex-c flexbag Rx only
Solution for Infusion Single-dose flexible container
For intravenous administration
The patient and physician should discuss the risks and benefits of this
product. Refer to the package insert for dosage and administration and
compatibility with other solutions.
Prior to administration, check bag for leaks by squeezing firmly. If leaks
are found, discard bag as sterility may be compromised.
Gamunex-C may be stored for 36 months at 2°C to 8°C (36°F to 46°F),
AND product may be stored at temperatures not to exceed 25°C (77°F)
for up to 6 months anytime during the 36 month shelf life, after which
the product must be immediately used or discarded. Do not freeze.
Keep the overwrap intact. Do not use if turbid.
Grifols Therapeutics LLC GRIFOLS
Clayton, NC 27520 USA
U.S. License No. 1871
4001022
(01)003 13533 805104
-
INGREDIENTS AND APPEARANCE
GAMUNEX-C
immune globulin (human) injectionProduct Information Product Type PLASMA DERIVATIVE Item Code (Source) NDC: 13533-805 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Human Immunoglobulin G (UNII: 66Y330CJHS) (Human Immunoglobulin G - UNII:66Y330CJHS) Human Immunoglobulin G 10 g in 100 mL Inactive Ingredients Ingredient Name Strength Glycine (UNII: TE7660XO1C) Water (UNII: 059QF0KO0R) Product Characteristics Color WHITE (Clear to opalescent liquid, colorless to pale yellow) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 13533-805-01 1 in 1 CARTON 1 NDC: 13533-805-00 50 mL in 1 BAG; Type 0: Not a Combination Product 2 NDC: 13533-805-11 1 in 1 CARTON 2 NDC: 13533-805-10 100 mL in 1 BAG; Type 0: Not a Combination Product 3 NDC: 13533-805-31 1 in 1 CARTON 3 NDC: 13533-805-30 200 mL in 1 BAG; Type 0: Not a Combination Product 4 NDC: 13533-805-45 1 in 1 CARTON 4 NDC: 13533-805-44 400 mL in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125046 10/13/2010 Labeler - GRIFOLS USA, LLC (048987452) Establishment Name Address ID/FEI Business Operations GRIFOLS THERAPEUTICS LLC 611019113 manufacture(13533-805) Establishment Name Address ID/FEI Business Operations GRIFOLS BIOLOGICALS LLC 092694538 manufacture(13533-805) Establishment Name Address ID/FEI Business Operations GRIFOLS BIOLOGICALS LLC 121076871 manufacture(13533-805)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.