DIGENYX- hyoscyamine sulfate 0.125 mg tablet
Drug Labeling and Warnings
Drug Details [pdf]
- Full Prescribing Information:
-
DESCRIPTION:
Digenyx™ (Hyoscyamine Sulfate USP, 0.125 mg) tablets are off-white to light beige round tablet containing 0.125 mg hyoscyamine sulfate formulated for oral administration. Each tablet also contains inactive ingredients: Lactose Monohydrate, Magnesium Stearate, Microcrystalline Cellulose, Stearic Acid, Sodium Starch Glycolate.
Hyoscyamine sulfate is one of the principal anticholinergic/antispasmodic components of belladonna alkaloids. The empirical formula is (C 17H 23NO 3) 2 H 2SO 4 2H 2O and the molecular weight is 712.85. Chemically, it is benzeneacetic acid,α-(hydroxymethyl)-, 8-methyl-8-azabicyclo [3.2.1] oct-3-yl ester, [3(S)-endo]-, sulfate (2:1), dihydrate with the following structure: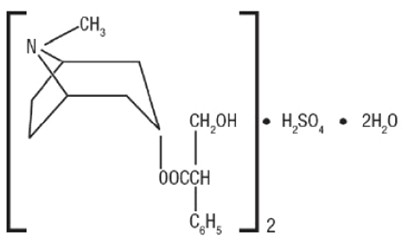
-
CLINICAL PHARMACOLOGY:
Hyoscyamine sulfate is an anticholinergic agent that works by inhibiting the action of acetylcholine on muscarinic receptors in smooth muscle. By blocking acetylcholine, Hyoscyamine reduces gastrointestinal motility and secretions, alleviating symptoms associated with irritable bowel syndrome (IBS) and other gastrointestinal disorders such as spasms, cramping, and bloating.
Hyoscyamine also decreases gastric acid secretion, resulting in a reduction in digestive tract activity. It relaxes the muscles in the gut, improving the passage of food and preventing spasms. In addition, it reduces the secretion of fluids, including saliva, sweat, and digestive juices, which further aids in the management of conditions involving excess gastrointestinal secretions.
Pharmacokinetically, hyoscyamine is rapidly absorbed after oral administration, with a peak plasma concentration occurring within 1 to 2 hours. Its effects are generally noticeable within 30 to 60 minutes, and its half-life is about 2 to 3.5 hours. Hyoscyamine sulfate is partially hydrolyzed to tropic acid and tropine; however, the majority of the drug is excreted unchanged in the urine within the first 12 hours. It is metabolized in the placenta and liver and eliminated primarily via the kidneys, largely in unchanged form. Hyoscyamine crosses the blood–brain barrier and may produce central nervous system effects, including drowsiness or dizziness. -
INDICATIONS AND USAGE:
Digenyx™ (Hyoscyamine Sulfate USP, 0.125 mg) tablets are an anticholinergic agent used as adjunctive therapy for a range of gastrointestinal and genitourinary conditions. Hyoscyamine reduces gastrointestinal motility and secretions and relieves smooth muscle spasm.
Digenyx™ (Hyoscyamine Sulfate USP, 0.125 mg) tablets may be used as adjunctive therapy in conditions such as:
-Peptic ulcer disease
-Irritable bowel syndrome and functional gastrointestinal disorders (e.g., spastic colon, mucous colitis)
-Acute enterocolitis
-Neurogenic bladder and bladder spasms
-Biliary and renal colic (in combination with narcotic analgesics) - CONTRAINDICATIONS:
-
WARNINGS:
Digenyx™ (Hyoscyamine Sulfate USP, 0.125 mg) tablets may cause drowsiness, dizziness, or blurred vision, so patients should avoid activities that require mental alertness, such as driving or operating machinery. It should be used with caution in individuals with heart disease, high blood pressure, hyperthyroidism, or enlarged prostate, and is not recommended for patients with narrow-angle glaucoma.
In hot environments, the use of this drug may lead to heat prostration (fever, heat stroke due to decreased sweating).
Diarrhea could be an early sign of incomplete intestinal obstruction, especially for patients with an ileostomy or colostomy, in which case using this product may be harmful.
Psychosis has been reported in sensitive individuals given anticholinergic drugs including hyoscyamine sulfate.
CNS signs and symptoms include confusion, disorientation, short-term memory loss, hallucinations, dysarthria, ataxia, coma, euphoria, anxiety, decreased anxiety, fatigue, insomnia, agitation and mannerisms and inappropriate affect. These CNS signs and symptoms usually resolve within 12 to 48 hours after discontinuation of the drug. -
PRECAUTIONS:
General:
Use with caution in patients with autonomic neuropathy, hyperthyroidism, heart disease, heart failure, arrhythmias, high blood pressure, or kidney disease. Hyoscyamine may increase heart rate, so check for tachycardia before using anticholinergics. Patients with hiatal hernia and reflux esophagitis should also be monitored closely. Prolonged use may decrease saliva, leading to dental issues.
Information for Patients:
Digenyx™ (Hyoscyamine Sulfate USP, 0.125 mg) tablets can cause drowsiness, dizziness, and blurred vision. Avoid driving or operating machinery until you know how the medication affects you. During hot weather or exercise, be cautious as this medication can reduce sweating, increasing the risk of heat stroke. Patients should observe caution before driving, using machinery or performing other tasks requiring mental alertness. Take the medication 30 minutes to 1 hour before meals and swallow the tablets whole.
Drug Interactions:
Hyoscyamine may interact with anticholinergics, antihistamines, antidepressants, pain medications, and antacids, which can lead to increased side effects like constipation or confusion. Inform your healthcare provider of all medications and supplements you're taking.
Antacids may interfere with the absorption of hyoscyamine sulfate. Administer hyoscyamine sulfate before meals; antacids after meals.
Pregnancy:
Hyoscyamine is classified as Pregnancy Category C. There are no data on its effects during pregnancy. It should only be used if clearly needed and prescribed by a licensed healthcare practitioner, as it crosses the placenta.
Nursing Mothers:
Hyoscyamine passes into breast milk. It should not be used while breastfeeding unless clearly necessary.
Pediatric Use:
Infants and young children are especially sensitive to its effects, including increased body temperature in hot environments. Close supervision is required for children with brain conditions as they may react more strongly to the medication.
Geriatric Use:
Reported clinical experience has not identified differences in safety between patients aged 65 and over and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function and of concomitant disease or other drug therapy.
This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function. -
ADVERSE REACTIONS:
The following adverse reactions have been reported for hyoscyamine sulfate and for pharmacologically similar drugs with anticholinergic/antispasmodic action. Adverse reactions may include dryness of the mouth; urinary hesitancy and retention; blurred vision; tachycardia; palpitations; mydriasis; cycloplegia; increased ocular tension; loss of taste; headache; nervousness; drowsiness; weakness; fatigue; dizziness; insomnia; nausea; vomiting; impotence; suppression of lactation; constipation; bloated feeling; abdominal pain; diarrhea; allergic reactions or drug idiosyncrasies; urticaria and other dermal manifestations; ataxia; speech disturbance; some degree of mental confusion and/or excitement (especially in elderly persons); short-term memory loss; hallucinations; and decreased sweating.
To report SUSPECTED ADVERSE REACTIONS, contact PureTek Corporation at 1-877-921-7873 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. -
OVERDOSAGE:
The signs and symptoms of overdose are headache, nausea, vomiting, blurred vision, dilated pupils, hot dry skin, dizziness, dryness of the mouth, difficulty in swallowing and CNS stimulation.
Measures to be taken are immediate lavage of the stomach and injection of physostigmine 0.5 to 2 mg intravenously and repeated as necessary up to a total of 5 mg. Fever may be treated symptomatically (tepid water sponge baths, hypothermic blanket). Excitement to a degree which demands attention may be managed with sodium thiopental 2% solution given slowly intravenously or chloral hydrate (100-200 mL of a 2% solution) by rectal infusion. In the event of progression of the curare-like effect to paralysis of the respiratory muscles, artificial respiration should be instituted and maintained until effective respiratory action returns.
In rats, the LD50 for hyoscyamine is 375 mg/kg. Hyoscyamine sulfate is dialyzable. -
DOSAGE AND ADMINISTRATION:
Dosage should be individualized according to the condition and severity of symptoms. Administer orally 30 to 60 minutes before meals and at bedtime, or as directed by licensed healthcare practitioner.
Adults and pediatric patients 12 years of age and older: 1 to 2 tablets every four hours or as needed. Do not exceed 12 tablets in 24 hours.
Pediatric patients 2 to under 12 years of age: 1/2 to 1 tablet every four hours or as needed. Do not exceed 6 tablets in 24 hours.
Note: Geriatric patients may be more sensitive to the effects of the usual adult dose. -
HOW SUPPLIED:
Digenyx™ (Hyoscyamine Sulfate USP, 0.125 mg) are off-white to light beige round tablets. Each bottle contains 30 tablets (NDC: 59088-150-54). Dispense in a tight, light-resistant container as defined in the USP/NF with a child-resistant closure.
- STORAGE AND HANDLING
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
DIGENYX
hyoscyamine sulfate 0.125 mg tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 59088-150 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HYOSCYAMINE SULFATE (UNII: F2R8V82B84) (HYOSCYAMINE - UNII:PX44XO846X) HYOSCYAMINE SULFATE 0.125 mg Inactive Ingredients Ingredient Name Strength MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) STEARIC ACID (UNII: 4ELV7Z65AP) Product Characteristics Color white (off-white to light beige) Score no score Shape ROUND Size 4mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 59088-150-54 30 in 1 BOTTLE; Type 0: Not a Combination Product 05/13/2026 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 05/13/2026 Labeler - PureTek Corporation (785961046)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.
