KETOROLAC TROMETHAMINE tablet, film coated
Drug Labeling and Warnings
Drug Details [pdf]
- Dosage Form and Strength
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
KETOROLAC TROMETHAMINE
ketorolac tromethamine tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 87234-7695(NDC:69452-275) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength KETOROLAC TROMETHAMINE (UNII: 4EVE5946BQ) (KETOROLAC - UNII:YZI5105V0L) KETOROLAC TROMETHAMINE 10 mg Product Characteristics Color white Score no score Shape ROUND Size 8mm Flavor Imprint Code KT Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 87234-7695-1 10 in 1 BOTTLE; Type 0: Not a Combination Product 05/08/2026 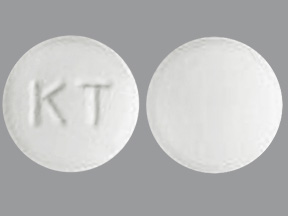
Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA216651 05/08/2026 Labeler - Injecta Medcial LLC (141762915)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.
 Ketorolac 10mg Tab 10CT
Ketorolac 10mg Tab 10CT