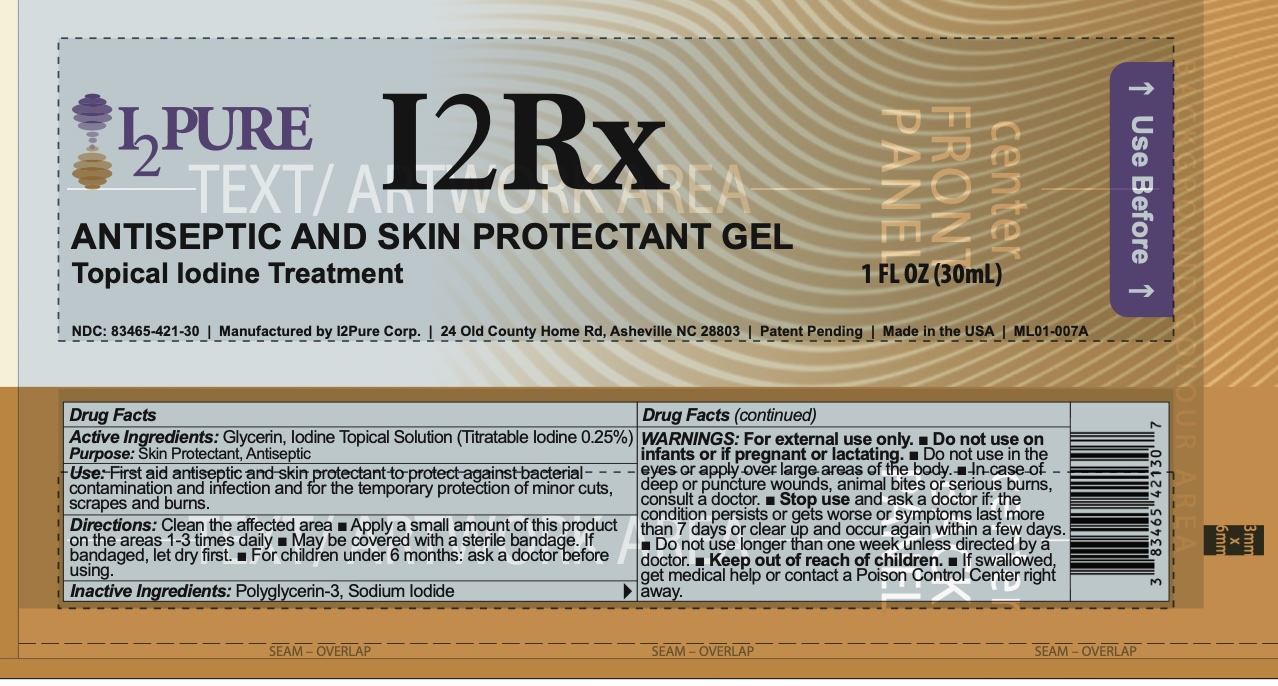
I2PURE I2RX- antiseptic and skin protectant gel gel
Drug Labeling and Warnings
Drug Details [pdf]
- DOSAGE & ADMINISTRATION
- INACTIVE INGREDIENT
- INDICATIONS & USAGE
- ACTIVE INGREDIENT
- ASK DOCTOR
- KEEP OUT OF REACH OF CHILDREN
- PURPOSE
- STOP USE
- WARNINGS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
I2PURE I2RX
antiseptic and skin protectant gel gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 83465-421 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GLYCERIN (UNII: PDC6A3C0OX) (GLYCERIN - UNII:PDC6A3C0OX) GLYCERIN 25.9 g in 30 mL IODINE (UNII: 9679TC07X4) (IODINE - UNII:9679TC07X4) IODINE 95 mg in 30 mL Inactive Ingredients Ingredient Name Strength SODIUM IODIDE (UNII: F5WR8N145C) 95 mg in 30 mL POLYGLYCERIN-3 (UNII: 4A0NCJ6RD6) 11.1 g in 30 mL Product Characteristics Color brown Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 83465-421-30 30 mL in 1 TUBE; Type 0: Not a Combination Product 05/11/2026 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M003 05/11/2026 Labeler - I2Pure Corp (117480499) Establishment Name Address ID/FEI Business Operations I2Pure Corp 145009177 manufacture(83465-421)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.