XTORO- finafloxacin suspension
Drug Labeling and Warnings
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use XTORO safely and effectively. See full prescribing information for XTORO.
XTORO (finafloxacin otic suspension) 0.3%,
for topical otic use
Initial U.S. Approval: 2014INDICATIONS AND USAGE
XTORO® is a quinolone antimicrobial indicated for the treatment of acute otitis externa (AOE) caused by susceptible strains of Pseudomonas aeruginosa and Staphylococcus aureus. (1)
DOSAGE AND ADMINISTRATION
Instill four drops in the affected ear(s) twice daily for seven days. For patients requiring use of an otowick, the initial dose can be doubled (to 8 drops), followed by 4 drops instilled into the affected ear twice daily for seven days. (2)
DOSAGE FORMS AND STRENGTHS
Otic Suspension: finafloxacin 0.3% (3)
CONTRAINDICATIONS
None. (4)
WARNINGS AND PRECAUTIONS
- Prolonged use of this product may lead to overgrowth of nonsusceptible organisms. Discontinue use if this occurs. (5.1)
- Allergic reactions may occur in patients with a history of hypersensitivity to finafloxacin, to other quinolones, or to any of the components in this medication. Discontinue use if this occurs. (5.2)
ADVERSE REACTIONS
The most common adverse reactions occurring in 1% of patients with XTORO were ear pruritus and nausea. (6)
To report SUSPECTED ADVERSE REACTIONS, contact Fonseca Biosciences, LLC at 1-877-436-6732 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 3/2026
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Growth of Resistant Organisms with Prolonged Use
5.2 Allergic Reactions
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.3 Pharmacokinetics
12.4 Microbiology
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
Instill four drops into the affected ear(s) twice daily for seven days. For patients requiring use of an otowick, the initial dose can be doubled (to 8 drops), followed by 4 drops instilled into the affected ear twice daily for seven days.
Important administration instructions include:
- Warm the suspension by holding the bottle in the hand for one or two minutes prior to dosing in order to avoid dizziness which may result from the instillation of a cold suspension. Shake bottle well before use.
- Lie with the affected ear upward, instill the drops, and maintain the position for 60 seconds to facilitate penetration of the drops into the ear canal.
- Repeat if necessary for the opposite ear.
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
- 5 WARNINGS AND PRECAUTIONS
-
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
A total of 618 patients were treated with XTORO in two Phase 3 clinical trials. The most frequently reported adverse reactions of those exposed to XTORO occurring at an incidence of 1% included ear pruritus and nausea.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no adequate or well-controlled studies with XTORO in pregnant women. Finafloxacin was shown to be teratogenic in rabbits and rats following oral administration. Neural tube defects and skeletal anomalies in both species, and limb anomalies in rabbits, were observed at exposures estimated to be at least 1300 times the maximum human systemic exposure following topical otic administration of 0.3% finafloxacin. Because animal studies are not always predictive of human responses, XTORO should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Data
Animal Data
In rabbit embryofetal studies, maternal toxicity was not observed at oral doses up to 9 mg/kg/day (estimated 8000 times the maximum human systemic exposure [0.234 ng/mL] following topical otic administration with 0.3% finafloxacin). Fetal toxicity was observed at the lowest dose tested, 1 mg/kg/day (estimated 1300 times the maximum human systemic exposure following topical otic administration with 0.3% finafloxacin), and included exencephaly, enlarged fontanel, spina bifida, phocomelia, paw hyperflexure, missing lumbar vertebra, missing lumbar arch, and sternebra fusion.
In a rat embryofetal study, no adverse maternal toxicity was observed at oral doses up to 100 mg/kg/day (estimated 60,000 times the maximum human systemic exposure following topical otic administration with 0.3% finafloxacin). The developmental no observed adverse effect level (NOAEL) was 30 mg/kg (estimated 22,000 times the maximum human systemic exposure following topical otic administration with 0.3% finafloxacin). Exencephaly was observed in one fetus at 100 mg/kg. At 500 mg/kg, additional developmental toxicities were observed including increased preimplantation loss, decreased fetal weight, decreased placental weight, increased incidence of non-ossified sternebrae, and delayed ossifications in the sternebrae, xiphisternum, sacral arches and metacarpals.
8.2 Lactation
Finafloxacin has been identified in the milk of nursing rats following oral administration. The human systemic concentration of XTORO following topical otic treatment is low [see Clinical Pharmacology (12.3)]. It is not known whether topical otic administration could result in sufficient systemic absorption to produce detectable quantities in the human breast milk. Caution should be exercised when finafloxacin is administered to a nursing mother.
8.4 Pediatric Use
The safety and efficacy of XTORO in infants below one year of age have not been established. The safety and efficacy of XTORO in treating acute otitis externa in pediatric patients one year or older have been demonstrated in adequate and well controlled clinical trials [see Clinical Studies (14)].
-
11 DESCRIPTION
XTORO (finafloxacin otic suspension) 0.3% is a quinolone antimicrobial. Its chemical name is (-)-8-cyano-1-cyclopropyl-6-fluoro-7-[(4aS,7aS)-hexahydropyrrolo[3,4-b]-1,4-oxazin-6(2H)-yl]- 4-oxo-1,4-dihydroquinoline-3-carboxylic acid (CAS number 209342-40-5). Its structural formula is:
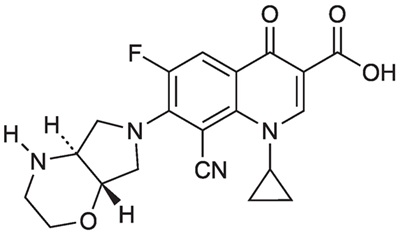
Finafloxacin has a molecular weight of 398.4. Finafloxacin is a white to yellow powder or crystals that is slightly soluble in water (0.125 mg/mL).
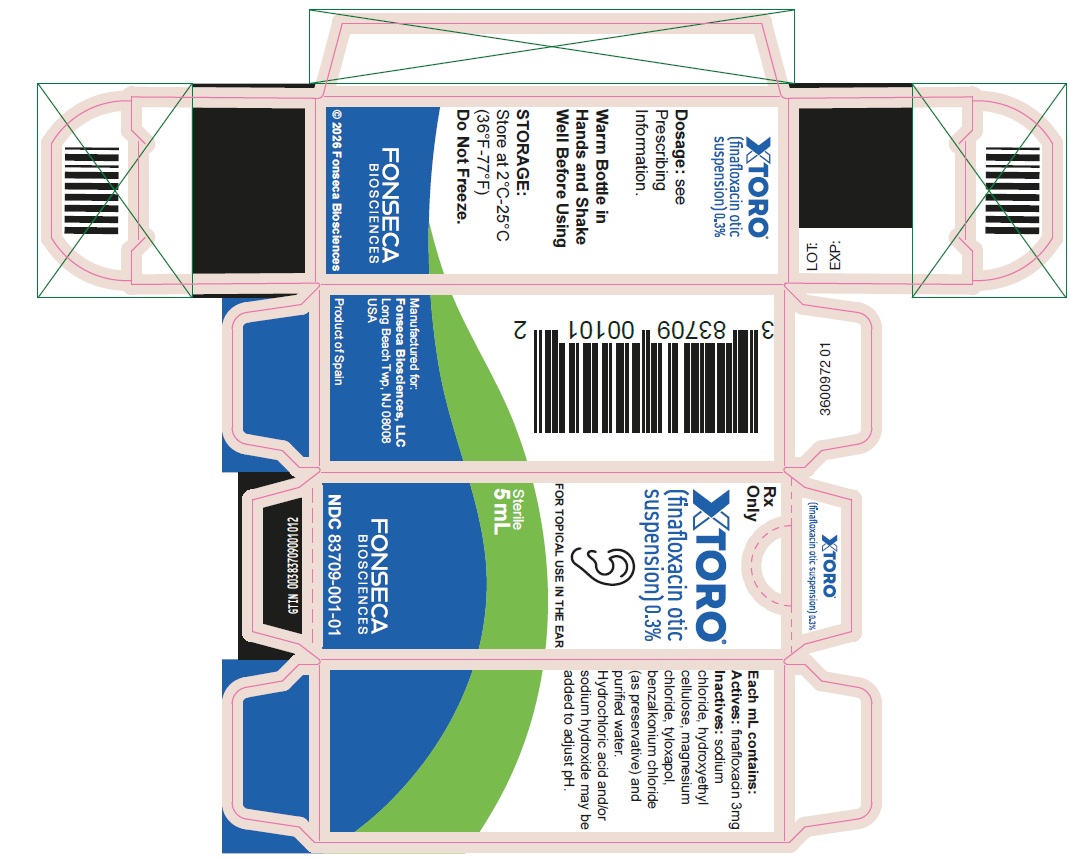
XTORO (finafloxacin otic suspension), 0.3% is supplied as a sterile, preserved, aqueous suspension for topical otic use. It has a pH of approximately 6.0 and an osmolality of approximately 290 mOsm/kg.
XTORO contains Active ingredient: finafloxacin, 0.3%. Preservative: benzalkonium chloride (0.005%). Inactive ingredients include: sodium chloride, hydroxyethylcellulose, tyloxapol, magnesium chloride, and purified water. May contain hydrochloric acid and/or sodium hydroxide to adjust pH.
-
12 CLINICAL PHARMACOLOGY
12.3 Pharmacokinetics
Finafloxacin plasma concentrations were evaluated following single or repeated ototopical doses of XTORO (finafloxacin otic suspension) 0.3%. In healthy subjects administered 4 drops in each ear twice daily for seven days, quantifiable finafloxacin concentrations were observed in 2 of 14 subjects; and these concentrations were just above the quantitation limit (0.05 ng/mL). Similarly, in AOE patients administered a single dose of 4 or 8 drops in each ear, quantifiable finafloxacin concentrations of up to 0.234 ng/mL were observed in plasma samples from 2 of 36 AOE patients.
12.4 Microbiology
Finafloxacin belongs to the fluoroquinolone class of antibacterials which involves the inhibition of bacterial type II topoisomerase enzymes, DNA gyrase and topoisomerase IV, which are required for bacterial DNA replication, transcription, repair and recombination.
Finafloxacin has been shown to be active against most isolates of the following bacteria, both in vitro and clinical studies as described in the INDICATIONS and USAGE section of the package insert for XTORO
Pseudomonas aeruginosa
Staphylococcus aureusMechanism of Resistance
Resistance to fluoroquinolones occurs primarily by mutations in the chromosomal DNA that encode for DNA gyrase and DNA topoisomerase enzymes, decreased outer membrane permeability or drug efflux mechanisms. In vitro resistance to finafloxacin due to spontaneous mutation is rare.
Cross ResistanceCross-resistance has been observed between finafloxacin and other fluoroquinolones. No cross-resistance has been observed between finafloxacin and other classes of antibacterial agents.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity
Animal studies have not been conducted to determine the carcinogenic potential of finafloxacin.Mutagenesis
Finafloxacin was shown to be genotoxic and clastogenic in vitro, with and without metabolic activation, and in vivo. In a bacterial reverse mutation assay, finafloxacin was positive in only one strain (TA102). Finafloxacin was positive in mammalian cell culture assays: mouse lymphoma cell forward mutation assays, a mutagenicity assay in V79 Chinese hamster lung cells, and a micronucleus test in V79 cells. Finafloxacin was clastogenic in mouse micronucleus studies.Impairment of fertility
An oral rat fertility study detected a NOAEL for male and female fertility of 100 mg/kg/day (estimated 60,000 times the maximum human systemic exposure following topical otic administration with 0.3% finafloxacin). At 500 mg/kg/day, males were completely infertile, presumably due to low sperm count and sperm immobility.General toxicity studies in rats have confirmed sperm toxicity following oral and intravenous dosing. Following intravenous dosing, the NOAEL for sperm toxicity was 30 mg/kg/day (150,000 times the maximum human exposure following topical otic administration with 0.3% finafloxacin).
-
14 CLINICAL STUDIES
In two randomized multicenter, vehicle controlled clinical trials, XTORO dosed four drops twice daily for 7 days was superior to its vehicle for both clinical and microbiological outcomes as well as in time to cessation of ear pain in patients with acute otitis externa (AOE).
Among 560 patients (161 with an otowick) that were pathogen positive (baseline microbiological specimen that contained Staphylococcus aureus and/or Pseudomonas aeruginosa), clinical cure on Day 11 was 71% in XTORO versus 37% in Vehicle. Among 1234 patients who received study treatment (Intent to Treat population (ITT)), aged 6 months to 85 years, clinical cures were 71% for XTORO and 50% in Vehicle.
Clinical Curesa at Day 11 (Pathogen Positive Subset and ITT) Study 1 Study 2 a A clinical cure was attained if the sum of the numerical scores of the 3 signs and symptoms of AOE (tenderness, erythema, and edema) was 0 at Day 11 (TOC). XTORO Vehicle XTORO vs.
Vehicle
Difference (95%
CI)XTORO Vehicle XTORO vs.
Vehicle
Difference (95%
CI)Pathogen
+ Subset104/145
(71.7%)46/138
(33.3%)38.4%
(27.6%, 49.1%)101/147
(68.7%)52/130
(40.0%)28.7%
(17.4%, 40.0%)ITT 245/344
(71.2%)173/342
(50.6%)20.6%
(13.5%, 27.8%)194/274
(70.8%)134/274
(48.9%)21.9%
(13.9%, 29.9%)The median time to cessation of ear pain in pathogen positive patients treated with XTORO was 3.5 days compared to 6.8 days in Vehicle. The median time to cessation of ear pain in ITT patients treated with XTORO was 3.5 days compared to 5.3 days in Vehicle.
Median Time (in Days) to Cessation of Ear Pain (Pathogen Positive Subset and ITT) Study 1 Study 2 XTORO Vehicle XTORO vs.
Vehicle
Difference (95%
CI)XTORO Vehicle XTORO vs.
Vehicle
Difference (95%
CI)Pathogen
+ Subset4.0 7.0 -3.0
(-5.0, -0.8)3.0 6.5 -3.6
(-5.0, -2.0)ITT 4.0 5.0 1.0
(-2.0, -0.5)3.0 6.5 -2.2
(-3.0, -1.0)Among the pathogen positive patients, microbiological success (eradication of all baseline organisms) was achieved on Day 11 in 67% in XTORO versus 13% in the Vehicle treated patients.
Microbiological Successb at Day 11 (Pathogen Positive Subset) Study 1 Study 2 b Microbiological success was attained if all pre-therapy bacteria were absent from the exit otic specimen. The presence of fungi and/or yeast was not considered in the determination of microbiological success. XTORO Vehicle XTORO vs.
Vehicle
Difference
(95% CI)XTORO Vehicle XTORO vs.
Vehicle
Difference
(95% CI)Pathogen + Subset 97/145
(66.9%)18/138
(13.0%)53.9%
(44.4%, 63.4%)97/147
(66.0%)15/130
(11.5%)54.4% (45.0%, 63.9%) In clinically cured pathogen positive patients, XTORO demonstrated eradication rates of 89% in both Staphylococcus aureus and Pseudomonas aeruginosa. Vehicle eradication rates were 33% for Staphylococcus aureus and 20% for Pseudomonas aeruginosa.
-
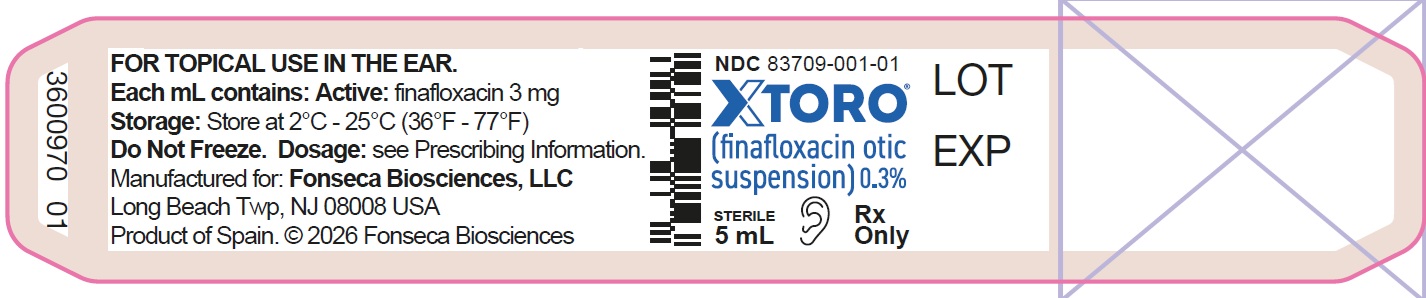
16 HOW SUPPLIED/STORAGE AND HANDLING
XTORO (finafloxacin otic suspension) 0.3% is a sterile, preserved, aqueous, otic suspension supplied in an opaque plastic bottle with a controlled drop tip and a white cap:
5 mL fill in a 5 mL bottle (NDC: 83709-001-01)
Storage and HandlingStore at 2°C to 25°C (36°F to 77°F). Do not freeze.
-
17 PATIENT COUNSELING INFORMATION
Allergic Reactions
Advise patients that if a rash or allergic reaction occurs, they should discontinue the use of the product immediately and contact their physician.Warm the Bottle in Hands Before Use
Advise patients or caregivers that prior to administration of XTORO, they should warm the bottle by holding it in their hands for one or two minutes to avoid dizziness which may result from the instillation of a cold solution.For Use with an Otowick
Advise patients that following instillation of 8 drops at the time of otowick insertion, they should continue with the lower dose of 4 drops administered twice daily for seven days.U.S. Patents Nos: 8,536,167; 9,119,859; 9,504,691; 9,993,483
©2026 Fonseca Biosciences, LLCManufactured For:
Fonseca Biosciences, LLC
2807 Long Beach Blvd Ste 1A
Long Beach Township, New Jersey 08008 USA
1-877-436-6732
medinfo@fonsecabio.com -
PATIENT PACKAGE INSERT
PATIENT INFORMATION
XTORO (ex tore’ oh)
(finafloxacin otic suspension) 0.3%
for topical otic useWhat is XTORO?
XTORO is a prescription medicine that is used to treat bacterial infections of the external ear canal, with or without an otowick in people 1 year of age and older.It is not known if XTORO is safe and effective in children under 1 year of age.
Before using XTORO, tell your healthcare provider about all of your medical conditions, including if you:
- allergic to any antibacterial including fluoroquinolones or to any of the ingredients in XTORO. See the end of this Patient Information leaflet for a complete list of the ingredients in XTORO.
- are pregnant or plan to become pregnant. It is not known if XTORO will harm your unborn baby.
- are breastfeeding or plan to breastfeed. It is not known if XTORO can pass into your breast milk. Talk to your healthcare provider about the best way to feed your baby during treatment with XTORO.
How should I use XTORO?
- XTORO is for use in the ear only. Do not use XTORO in the eyes or mouth.
- Use XTORO exactly as your healthcare provider tells you to take it.
- Read the Instructions for Use that comes with XTORO.
What are the possible side effects of XTORO? XTORO may cause serious side effects, including:
- Growth of yeast or fungi. XTORO may cause growth of yeast or fungi during long term use. Stop using XTORO if you develop a growth of yeast or fungi.
- Allergic reactions. Stop using XTORO and call your healthcare provider or go to the nearest emergency room right away if you have any of the following signs or symptoms of an allergic:
- rash
- hives
- swelling of your face, lips, mouth, or tongue
- itching
- trouble breathing
- dizziness, fast heartbeat, or pounding in your chest.
The most common side effects of XTORO include:
- itching in the ear
- nausea
Tell your healthcare provider if you have any side effects that bothers you or does not go away.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.How should I store XTORO?
- Store XTORO between 36°F to 77°F (2°C to 25°C).
- Do not freeze XTORO.
Keep XTORO and all medicines out of the reach of children.
General Information about the safe and effective use of XTORO.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use XTORO for a condition for which it was not prescribed. Do not give XTORO to other people, even if they have the same symptoms that you have. It may harm them. This Patient Information leaflet summarizes the most important information about XTORO. You can ask your pharmacist or healthcare provider for information about XTORO that is written for healthcare professionals.What are the ingredients in XTORO?
Active ingredients: finafloxacin, 0.3%
Inactive ingredients: sodium chloride, hydroxyethylcellulose, tyloxapol, magnesium chloride, and purified water. Hydrochloric acid and sodium hydroxide may be added to adjust pH.
U.S. Patent Nos: 8,536,167; 9,119,859; 9,504,691; 9,993,483
©2026 Fonseca Biosciences, LLC
Manufactured For: Fonseca Biosciences, LLC 2807 Long Beach Blvd Ste 1A Long Beach Township, New Jersey 08008 USA
1-877-436-6732
medinfo@fonsecabio.comThis Patient Information has been approved by the U.S. Food and Drug Administration Revised: 3/2026
-
INSTRUCTIONS FOR USE
PATIENT INSTRUCTIONS FOR USE
XTORO (ex tore’ oh)(finafloxacin otic suspension) 0.3%
for topical otic use
Important information you need to know before using XTORO.
- XTORO is for use in the ear only. Do not use XTORO in the eyes or mouth.
- Shake XTORO well before use.
- Do not touch the ear, fingers or any other surfaces with the tip of the bottle. This could contaminate the drops.
How should I use XTORO?
1. Wash your handsWash your hands with soap and water.
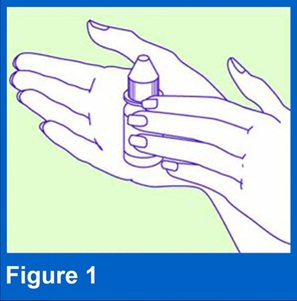
2. Warm and shake the bottleHold the bottle in your hands for 1 to 2 minutes to warm XTORO (see Figure 1). Using cold drops may cause dizziness. Shake the bottle of XTORO well before use.
3. Add the drops
Lie down on your side with the affected ear facing up (see Figure 2).
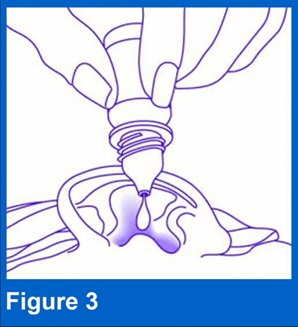
Squeeze the bottle and put 4 drops of XTORO into the affected ear (see Figure 3). If otowick is in place, put 4 drops of XTORO into the affected ear at the surface of the otowick. Do not touch the ear, fingers or any other surfaces with the tip of the bottle as it could contaminate the drops.
4. Pull ear lobe
Gently pull the outer ear lobe upward and backward (see Figure 4). This will allow the ear drops to flow down into the ear canal.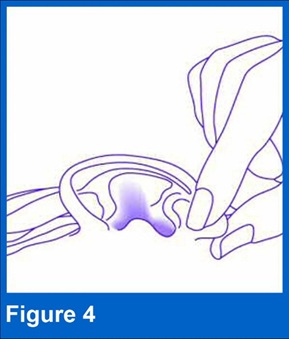
5. Stay on your side
Stay on your side with the affected ear facing up for at least 60 seconds. Repeat Steps 3-5 for the other ear if both ears are infected.How should I store XTORO?
- Store XTORO between 36°F to 77°F (2°C to 25°C).
- Do not freeze XTORO.
Keep XTORO and all medicines out of the reach of children.
Fonseca Biosciences, LLC
Long Beach Township, New Jersey 08008This Instructions for Use has been approved by the U.S. Food and Drug Administration Revised: 3/2026
- PRINCIPAL DISPLAY PANEL - 5 mL
-
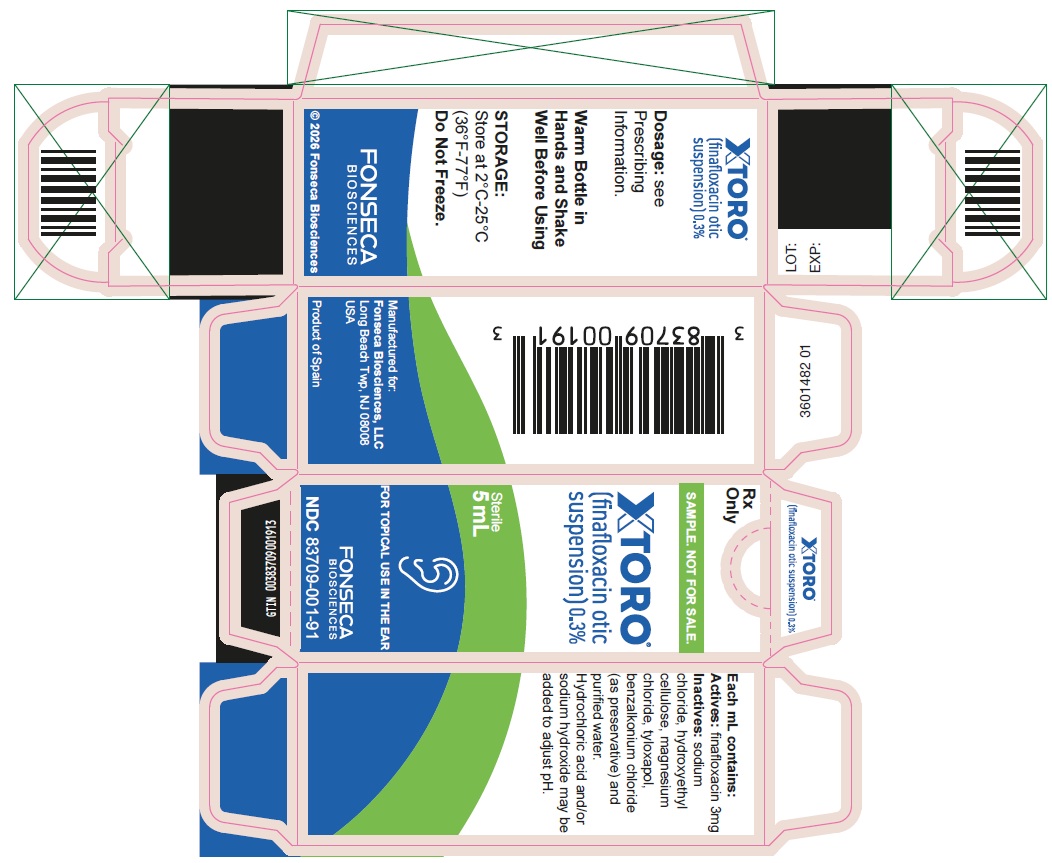
PRINCIPAL DISPLAY PANEL- 1.5 mL SAMPLE
NDC: 83709-001-90
Xtoro® (finafloxacin otic suspension) 0.3%
®Fonseca Biosciences, LLC
STERILE
1.5 mL


- PRINCIPAL DISPLAY PANEL - 5 mL SAMPLE
-
INGREDIENTS AND APPEARANCE
XTORO
finafloxacin suspensionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 83709-001 Route of Administration AURICULAR (OTIC) Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Finafloxacin (UNII: D26OSN9Q4R) (Finafloxacin - UNII:D26OSN9Q4R) Finafloxacin 3 mg in 1 mL Inactive Ingredients Ingredient Name Strength Benzalkonium Chloride (UNII: F5UM2KM3W7) Sodium Chloride (UNII: 451W47IQ8X) Hydroxyethyl Cellulose (1500 Mpa.s At 1%) (UNII: L605B5892V) Tyloxapol (UNII: Y27PUL9H56) Magnesium Chloride (UNII: 02F3473H9O) Water (UNII: 059QF0KO0R) Hydrochloric Acid (UNII: QTT17582CB) Sodium Hydroxide (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 83709-001-01 1 in 1 CARTON 05/01/2026 1 5 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product 2 NDC: 83709-001-91 1 in 1 CARTON 05/01/2026 2 5 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product 3 NDC: 83709-001-90 1 in 1 CARTON 05/01/2026 3 1 in 1 POUCH 3 1.5 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA206307 05/01/2026 Labeler - Fonseca Biosciences, LLC (118929241) Registrant - Fonseca Biosciences, LLC (118929241) Establishment Name Address ID/FEI Business Operations Siegfried El Masnou, S.A. 462029349 manufacture(83709-001)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.