REVYRA tablet
Drug Labeling and Warnings
Drug Details [pdf]
- Health Claim:
-
Description:
Revyra Multivitamin Tablets are tan, oblong tablets in plastic bottles of 30 ct.
85477-911-30
Reserved for Professional Recommendation
Revyra Tablets are orally administered prescription dietary supplement tablets intended to provide significant amounts of Vitamins A, B6, B12, C, D3, E, biotin, folate, calcium, magnesium, ferrous, and iodine to supplement the diet and help assure that nutritional deficiencies of these vitamins and minerals will not develop.
Revyra Multivitamin Tablets should be administered under the supervision of a licensed medical practitioner.
This listed product is not a National Drug Code, but instead has merely been formatted to comply with standard industry practice for pharmacy and insurance computer systems.
These statements have not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure or prevent any disease.
All prescriptions using this product shall be pursuant to state statutes as applicable. This is not an Orange Book product. This product may be administered only under a physician’s supervision. There are no implied or explicit claims on therapeutic equivalence.
Manufactured for:
Oncora Pharma LLC
Dallas TX 75228 -
Warnings:
This product is contraindicated in patients with a known hypersensitivity to any of the ingredients.
Revyra Multivitamin Tablets should only be used under the direction and supervision of a licensed medical practitioner. Use with caution in patients that may have a medical condition, are pregnant, lactating, trying to conceive, under the age of 18, or taking medications.
These statements have not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure, or prevent any disease.
- Dosage:
-
Precautions:
CONTRAINDICATIONS
This product is contraindicated in patients with known hypersensitivity to any of the ingredients.PRECAUTIONS
This product is contraindicated in patients with a known hypersensitivity to any of the ingredients.ADVERSE REACTIONS
Allergic sensitizations have been reported following oral administration of folic acid. Consult your physician immediately if adverse side effects occur.KEEP OUT OF THE REACH OF CHILDREN.
- HOW SUPPLIED HEALTH CLAIM:
- Label:
-
INGREDIENTS AND APPEARANCE
REVYRA
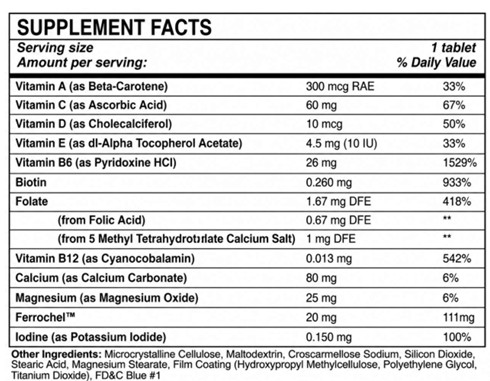
revyra tabletProduct Information Product Type DIETARY SUPPLEMENT Item Code (Source) NHRIC:85477-911 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BETA-CAROTENE (UNII: 01YAE03M7J) (BETA-CAROTENE - UNII:01YAE03M7J) BETA-CAROTENE .3 mg ASCORBIC ACID (UNII: PQ6CK8PD0R) (ASCORBIC ACID - UNII:PQ6CK8PD0R) ASCORBIC ACID 60 mg CHOLECALCIFEROL (UNII: 1C6V77QF41) (CHOLECALCIFEROL - UNII:1C6V77QF41) CHOLECALCIFEROL .01 mg .ALPHA.-TOCOPHEROL ACETATE, DL- (UNII: WR1WPI7EW8) (.ALPHA.-TOCOPHEROL, DL- - UNII:7QWA1RIO01) .ALPHA.-TOCOPHEROL, DL- 4.5 mg PYRIDOXINE HYDROCHLORIDE (UNII: 68Y4CF58BV) (PYRIDOXINE - UNII:KV2JZ1BI6Z) PYRIDOXINE HYDROCHLORIDE 26 mg BIOTIN (UNII: 6SO6U10H04) (BIOTIN - UNII:6SO6U10H04) BIOTIN .26 mg FOLIC ACID (UNII: 935E97BOY8) (FOLIC ACID - UNII:935E97BOY8) FOLIC ACID 1.67 mg CYANOCOBALAMIN (UNII: P6YC3EG204) (CYANOCOBALAMIN - UNII:P6YC3EG204) CYANOCOBALAMIN .013 mg CALCIUM CARBONATE (UNII: H0G9379FGK) (CALCIUM CATION - UNII:2M83C4R6ZB) CALCIUM CARBONATE 80 mg MAGNESIUM OXIDE (UNII: 3A3U0GI71G) (MAGNESIUM CATION - UNII:T6V3LHY838) MAGNESIUM OXIDE 25 mg FERROUS FUMARATE (UNII: R5L488RY0Q) (FERROUS CATION - UNII:GW89581OWR) FERROUS CATION 20 mg POTASSIUM IODIDE (UNII: 1C4QK22F9J) (IODIDE ION - UNII:09G4I6V86Q) POTASSIUM IODIDE .15 mg Inactive Ingredients Ingredient Name Strength MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) MALTODEXTRIN (UNII: 7CVR7L4A2D) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) STEARIC ACID (UNII: 4ELV7Z65AP) MAGNESIUM STEARATE (UNII: 70097M6I30) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NHRIC:85477-911-30 30 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date DIETARY SUPPLEMENT 05/01/2026 Supplement Facts Serving Size : Serving per Container : Amount Per Serving % Daily Value color scoring 1 shape size (solid drugs) 18 mm Labeler - Oncora Pharma, LLC (119482542)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.