KRESLADI- marnetegragene autotemcel injection, suspension
Drug Labeling and Warnings
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use KRESLADI safely and effectively. See full prescribing information for KRESLADI.
KRESLADI (marnetegragene autotemcel) suspension for intravenous infusion
Initial U.S. Approval: 2026INDICATIONS AND USAGE
KRESLADI is an autologous hematopoietic stem cell-based gene therapy indicated for the treatment of pediatric patients with severe leukocyte adhesion deficiency-I (LAD-I) due to biallelic variants in ITGB2 without an available human leukocyte antigen (HLA)-matched sibling donor for allogeneic hematopoietic stem cell transplant. (1)
This indication is approved under accelerated approval based on increase in neutrophil CD18 and CD11a surface expression [see Clinical Studies (14)]. Continued approval may be contingent upon verification and description of clinical benefit in a confirmatory trial(s).
DOSAGE AND ADMINISTRATION
For autologous use only. For intravenous use only.
- Patients must undergo hematopoietic stem cell (HSC) mobilization followed by apheresis to obtain CD34+ cells for KRESLADI manufacturing. (2.2)
- Dosing of KRESLADI is based on the number of CD34+ cells in the infusion bag(s) per kg of body weight. (2.1)
- The minimum recommended dose is 2.8 × 106 CD34+ cells/kg. (2.1)
- Full myeloablative conditioning must be administered before infusion of KRESLADI. (2.2)
- Verify the patient's identity matches the unique patient identification information on the KRESLADI infusion bag(s) prior to infusion. (2.2)
- Do not sample, alter, or irradiate KRESLADI. (2.2)
- Do not use an in-line blood filter or infusion pump. (2.3)
DOSAGE FORMS AND STRENGTHS
- KRESLADI is a cell suspension for intravenous infusion. (3)
- KRESLADI is composed of one or two infusion bags which contain 0.34 to 6.1 × 106 cells/mL (including 0.32 to 6.1 × 106 CD34+ cells/mL) suspended in a cryopreservation solution. (3)
- Each infusion bag contains approximately 30 mL of KRESLADI. (3)
CONTRAINDICATIONS
None. (4)
WARNINGS AND PRECAUTIONS
- Serious Infections: Monitor patients for signs and symptoms of infection before and after KRESLADI infusion and treat appropriately. Administer prophylactic antimicrobials according to institutional guidelines. (5.1)
- Veno-Occlusive Disease: Monitor patients for signs and symptoms of veno-occlusive disease including assessment of liver function tests during the first month following KRESLADI infusion. (5.2)
- Neutrophil Engraftment Failure: Monitor absolute neutrophil counts (ANC) after KRESLADI infusion. If neutrophil engraftment does not occur administer rescue cells. (5.3)
- Delayed Platelet Engraftment: Monitor patients for thrombocytopenia and bleeding until platelet engraftment and count recovery. (5.4)
- LVV-mediated Insertional Oncogenesis: Monitor patients at least annually for hematologic malignancies for at least 15 years after KRESLADI infusion. (5.5)
- Hypersensitivity Reactions: Monitor for hypersensitivity reactions during the infusion. (5.6)
ADVERSE REACTIONS
- The most common non-laboratory adverse reactions (≥ 30%): mucositis, upper respiratory tract infection, viral infection, febrile neutropenia, skin lesion, nausea/vomiting, rash/dermatitis, pyrexia, device related infection, and skin infection. (6.1)
- The most common laboratory adverse reactions (≥ 30%): hemoglobin decreased, platelet count decreased, neutrophil count decreased, leukocyte count decreased, aspartate aminotransferase increased, and alanine aminotransferase increased. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Rocket Pharmaceuticals at 1-800-982-2410 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- Anti-retrovirals: Patients should not take anti-retroviral medications for one month prior to mobilization, or for the expected duration required for elimination of the anti-retroviral medications, and until all cycles of apheresis are completed. (7.2)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 3/2026
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Dose
2.2 Preparation Before KRESLADI Infusion
2.3 Administration
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Serious Infections
5.2 Veno-Occlusive Disease
5.3 Neutrophil Engraftment Failure
5.4 Delayed Platelet Engraftment
5.5 LVV-Mediated Insertional Oncogenesis
5.6 Hypersensitivity Reactions
5.7 Anti-retroviral Use
5.8 Interference with Serology Testing
5.9 Blood, organ, tissue and cell donation
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
7 DRUG INTERACTIONS
7.1 Vaccines
7.2 Anti-retroviral Use
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Patients Seropositive for Human Immunodeficiency Virus (HIV)
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
KRESLADI is indicated for the treatment of pediatric patients with severe leukocyte adhesion deficiency-I (LAD-I) due to biallelic variants in ITGB2 without an available human leukocyte antigen (HLA)-matched sibling donor for allogeneic hematopoietic stem cell transplant.
This indication is approved under accelerated approval based on increase in neutrophil CD18 and CD11a surface expression [see Clinical Studies (14)]. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial(s).
-
2 DOSAGE AND ADMINISTRATION
2.1 Dose
For autologous use only. For one-time single-dose intravenous use only.
The recommended minimum dose of KRESLADI is a single intravenous infusion of 2.8 × 106 CD34+ cells/kg.
The dose is calculated based on the patient's weight prior to first apheresis.
See the Lot Information Sheet provided with the product shipment for additional information pertaining to dose. Up to two drug product lots may be administered to meet the minimum dose.
2.2 Preparation Before KRESLADI Infusion
Before mobilization, apheresis, and conditioning are initiated, confirm that autologous hematopoietic stem cell (HSC) transplantation is appropriate for the patient.
Consider administering ustekinumab prior to mobilization and apheresis and/or prior to KRESLADI infusion.
Mobilization and Apheresis
Administer granulocyte-colony-stimulating factor (G-CSF) and plerixafor for HSC mobilization followed by apheresis to obtain CD34+ cells for product manufacturing.
Weigh the patient prior to the first apheresis collection. Collect a recommended target number of CD34+ cells of 20 × 106 CD34+ cells/kg.
Repeat additional cycles of mobilization and apheresis after at least 14 days to obtain more cells for KRESLADI manufacture if the minimum dose of 2.8 × 106 CD34+ cells/kg is not met.
Back-Up CD34+ Cell Collection
Obtain and cryopreserve an additional collection of CD34+ cells of ≥ 2 × 106 CD34+ cells/kg at the treatment center prior to initiating myeloablative conditioning and infusion with KRESLADI. Administer the back-up collection for rescue treatment if there is: 1) compromise of KRESLADI after initiation of conditioning and before KRESLADI infusion, 2) primary engraftment failure, or 3) loss of engraftment after infusion with KRESLADI.
Myeloablative Conditioning
Do not begin conditioning until KRESLADI has been received and stored at the treatment center and the availability of the back-up collection of CD34+ cells is confirmed.
Administer myeloablative conditioning before infusion of KRESLADI based on institutional guidelines. Consult the prescribing information for the conditioning agents prior to treatment.
Infuse KRESLADI 24-48 hours after completion of myeloablative conditioning.
Receipt and Storage of KRESLADI
- Ensure the availability of vapor phase of liquid nitrogen storage at less than or equal to -150°C (-238°F) at the treatment center.
- KRESLADI is shipped to the treatment center in the vapor phase of liquid nitrogen shipper.
- Confirm the patient's identity with the patient identifiers on the shipper and labels.
- If there are any concerns about the product or packaging upon receipt, contact Rocket Pharmaceuticals at 1-800-982-2410.
- Keep the infusion bag(s) in the metal cassette(s) and transfer KRESLADI from the vapor phase of liquid nitrogen shipper to the onsite vapor phase of liquid nitrogen storage (at ≤ -150°C (≤ -238°F) until ready for thaw and administration).
Preparation of KRESLADI for infusion
Confirm the infusion time in advance and adjust the start time of KRESLADI thaw such that it will be available for infusion when the patient and the healthcare provider are ready. Complete infusion of each bag of KRESLADI within 30 minutes of the start of thaw.
- 1. Confirm patient identity: Prior to KRESLADI preparation, match the patient's identity with the patient identifiers on the KRESLADI cassette(s). Do not remove the KRESLADI infusion bag from the cassette if the information on the patient-specific label does not match the intended patient and contact Rocket Pharmaceuticals at 1-800-982-2410.
- 2. Once patient identification is confirmed, remove the KRESLADI infusion bag(s) from the cassette(s) and check that the patient information on the cassette label(s) matches the bag label(s).
- 3. A KRESLADI dose may be contained in one or two patient-specific infusion bags. Two infusion bags could be from two different drug product lots. Verify the number of bags received for the indicated dose of KRESLADI. Use the accompanying Lot Information Sheet to confirm that each infusion bag is within the expiration date prior to preparation of KRESLADI for infusion. If two infusion bags have been received, thaw and administer each infusion bag one at a time. Wait 1 to 2 hours after administration of the first bag before thawing of the second KRESLADI bag. Maintain the second infusion bag, if applicable, within the vapor phase of liquid nitrogen storage to maintain temperature less than or equal to -150°C (-238°F) until time to thaw.
- 4. Inspect each infusion bag(s) for any breaches of integrity such as breaks or cracks before thawing. If the bag(s) is compromised, follow the local guidelines and call Rocket Pharmaceuticals at 1-800-982-2410.
- 5. Place the infusion bag inside a sterile plastic bag per local institutional guidelines.
- 6. Thaw KRESLADI at 37°C (98.6°F) in a water bath or dry-thaw method until there is no visible ice in the infusion bag. Thawing should take no longer than 5 minutes. Promptly remove the infusion bag once thawed and keep it at room temperature.
- Do not leave KRESLADI unattended and do not submerge the infusion ports if thawed in a water bath.
- 7. Gently mix the contents of the bag to disperse clumps of cellular material. If visible cell clumps remain, continue to gently mix the contents of the bag. Most small clumps of cellular material should disperse with gentle manual mixing.
- Do not shake the bag.
- Do not filter, wash, spin down and/or resuspend KRESLADI in new media prior to infusion.
- Do not sample, alter, irradiate, or refreeze KRESLADI.
2.3 Administration
- Complete KRESLADI infusion within 30 minutes of the start of thaw.
- Do not use an in-line blood filter or infusion pump.
- Use central venous access for the infusion of KRESLADI.
- Before infusion, confirm the patient's identity matches the patient identifiers on the KRESLADI infusion bag(s). Do not use KRESLADI if the information on the patient-specific label does not match the intended patient. Use the Lot Information Sheet to confirm the total number of infusion bags to be administered.
- Prime the tubing of the infusion set with normal saline prior to infusion.
- Expose the sterile port on the infusion bag by tearing off the protective wrap covering the port.
- Connect the port to the primed infusion tubing and then connect the tubing to the patient's central venous access aseptically per institutional standard operating procedures.
- Administer the entire contents of the KRESLADI bag by intravenous infusion. The time for infusion will vary but will usually be less than 10 minutes.
- At the end of the infusion, flush the infusion bag and tubing with normal saline solution to ensure as many cells as possible are delivered to the patient. Consider the infusion volume in relation to the age and weight of the patient and the priming volume of the infusion tubing set.
- If two infusion bags are provided, administer the contents of the first infusion bag completely before proceeding to thaw (following Section 2.2 steps 4 - 7) and infuse (following Section 2.3 steps 1 - 6) the contents of the second infusion bag. Wait 1 to 2 hours after administration of the first bag before thawing and infusion of the second bag.
KRESLADI contains human blood cells that are genetically modified with replication-incompetent, self-inactivating lentiviral vector (LVV). Follow universal precautions and local institutional biosafety guidelines for handling and disposal of KRESLADI to avoid potential transmission of infectious diseases.
After KRESLADI Administration
Follow standard procedures for patient monitoring and management after HSC transplantation after KRESLADI infusion.
- Irradiate any blood products required within the first 3 months after KRESLADI infusion.
- Patients treated with KRESLADI should not donate blood, organs, tissues, or cells at any time in the future.
-
3 DOSAGE FORMS AND STRENGTHS
KRESLADI is a cell suspension for intravenous infusion.
KRESLADI is composed of one or two infusion bags which contain 0.34 to 6.1 × 106 cells/mL (including 0.32 to 6.1 × 106 CD34+ cells/mL) suspended in a cryopreservation solution [see How Supplied/Storage and Handling (16)]. Each infusion bag contains approximately 30 mL of KRESLADI. Up to two drug product lots may be administered to meet the recommended dose.
See the Lot Information Sheet for actual strength and dose.
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Serious Infections
Serious infections have occurred with KRESLADI administration [see Adverse Reactions (6.1)]. Increased susceptibility to infections may occur due to administration of myeloablative conditioning prior to KRESLADI infusion.
Monitor patients for signs and symptoms of infection before and after KRESLADI infusion and treat appropriately. Administer prophylactic antimicrobials according to institutional guidelines.
Avoid administration of KRESLADI in patients with active bloodstream infections or other serious, untreated infections.
Any blood products required after KRESLADI infusion should be irradiated.
5.2 Veno-Occlusive Disease
Veno-occlusive disease has occurred with KRESLADI treatment [see Adverse Reactions (6.1)]. Increased susceptibility to veno-occlusive disease may occur due to administration of myeloablative conditioning prior to KRESLADI infusion. Monitor patients for signs and symptoms of veno-occlusive disease including assessment of liver function tests during the first month following KRESLADI infusion.
5.3 Neutrophil Engraftment Failure
Neutrophil engraftment failure may occur after treatment with KRESLADI. Neutrophil engraftment failure is defined as failure to achieve three consecutive absolute neutrophil counts (ANC) ≥ 500 cells/microliter obtained on different days by Day 43 after infusion of KRESLADI. Monitor neutrophil counts until engraftment has been achieved. If neutrophil engraftment failure occurs in a patient treated with KRESLADI, provide rescue treatment with the back-up collection of CD34+ cells [see Preparation Before KRESLADI Infusion (2.2)].
5.4 Delayed Platelet Engraftment
Delayed platelet engraftment may occur after treatment with KRESLADI. Monitor platelet counts and bleeding until platelet engraftment and platelet recovery are achieved.
5.5 LVV-Mediated Insertional Oncogenesis
Lentiviral vector (LVV)-mediated insertional oncogenesis may occur after treatment with KRESLADI. Hematologic malignancy is a lifelong risk and patients treated with KRESLADI may develop hematologic malignancy at any time following treatment.
Monitor for hematologic malignancies clinically, and with a complete blood count (with differential) at least annually and integration site analysis as warranted for at least 15 years after treatment with KRESLADI and as clinically indicated. If malignancy is detected in any patient who received KRESLADI, contact Rocket Pharmaceuticals, Inc. at 1-800-982-2410 for reporting and to obtain instructions on collection of samples for testing.
5.6 Hypersensitivity Reactions
Hypersensitivity reactions including anaphylaxis may occur with the infusion of KRESLADI. The dimethyl sulfoxide (DMSO) in KRESLADI may cause hypersensitivity reactions which may occur in patients with and without prior exposure to DMSO.
Monitor patients for signs and symptoms of hypersensitivity reactions during and after KRESLADI infusion. If a hypersensitivity reaction occurs, pause infusion if ongoing and manage according to clinical practice.
5.7 Anti-retroviral Use
Anti-retroviral medications may interfere with manufacturing of KRESLADI [see Drug Interactions (7.2)]. If a patient requires anti-retrovirals for HIV prophylaxis, mobilization and apheresis of CD34+ cells for KRESLADI manufacturing should be delayed until HIV infection is adequately ruled out. Patients should not take anti-retroviral medications for at least one month prior to mobilization, or for the expected duration required for the elimination of the anti-retroviral medications, and until all cycles of apheresis are completed.
5.8 Interference with Serology Testing
Patients who have received KRESLADI are likely to test positive by polymerase chain reaction (PCR) assays for HIV due to LVV provirus insertion resulting in a false-positive test for HIV. Therefore, patients who have received KRESLADI should not be screened for HIV infection using a PCR-based assay.
-
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
As clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety data described in this section reflect exposure to KRESLADI in one clinical study (Study RP-L201-0318). A total of 9 pediatric patients with severe LAD-I received a single dose of intravenous KRESLADI with a median dose of 4.3 × 106 CD34+ cells/kg (range: 2.8 to 10 CD34+ cells/kg) [see Clinical Studies (14)]. The median duration of follow up after KRESLADI administration was 4.2 years (range: 3.6 to 5.7 years).
Serious adverse reactions were reported in 4 patients (44%) including serious infections (n=4), pulmonary arterial hypertension (n=1), sensorineural deafness (n=1), and veno-occlusive disease (n=1).
Table 1 presents the most common adverse reactions reported in Study RP-L201-0318.
Table 1: Non-Laboratory Adverse Reactions Reported in ≥ 20% of Patients in Study RP-L201-0318 (N=9) Adverse Reaction All Grades (%) Grade ≥3
(%)Gastrointestinal disorders Note: Adverse reactions are defined as adverse events that occurred from myeloablative conditioning administration through year 2 following KRESLADI administration. - * Includes multiple related terms
- † Device-related infection includes vascular device infection, device related bacteremia, and bacteremia that occurred with a central line in place
- ‡ Skin lesion includes pyoderma gangrenosum, skin lesion, aseptic pustule, lip erythema, hand erythema, skin erythema at port site, and skin hyperpigmentation
- § Rash includes rash, eczema, atopic dermatitis and diaper dermatitis
Nausea/Vomiting* 4 (44%) 0 Constipation 2 (22%) 0 General disorders and administration site conditions Mucositis* 8 (89%) 5 (56%) Febrile neutropenia 6 (67%) 6 (67%) Pyrexia 3 (33%) 0 Infections and infestations Upper respiratory tract infection* 8 (89%) 5 (56%) Viral infection* 7 (78%) 1 (11%) Skin infection* 3 (33%) 0 Device-related infection† 3 (33%) 2 (22%) Lower respiratory tract infection* 2 (22%) 1 (11%) Gastroenteritis 2 (22%) 1 (11%) Skin candidiasis* 2 (22%) 0 Urinary tract infection* 2 (22%) 0 Skin and subcutaneous tissue disorders Skin lesion‡ 6 (67%) 1 (11%) Rash§ 4 (44%) 0 Alopecia 2 (22%) 0 Table 2 presents laboratory abnormalities that worsened from baseline in ≥20% of patients in Study RP-L201-0318.
Table 2: Laboratory Abnormalities that Worsened from Baseline Reported in ≥ 20% of Patients in Study RP-L201-0318 (N=9)* Laboratory Abnormality† All Grades (%) Grade ≥ 3 (%) - * Baseline laboratory values were assessed prior to myeloablative conditioning
- † All events occurred within 30 days post-infusion except for liver enzymes increased, which occurred in 3 patients within 30 days and in one patient after 90 days post-infusion
Hemoglobin decreased 9 (100%) 9 (100%) Platelet count decreased 9 (100%) 9 (100%) Neutrophil count decreased 9 (100%) 9 (100%) Leukocyte count decreased 5 (56%) 5 (56%) Aspartate aminotransferase increased 4 (44%) 0 Alanine aminotransferase increased 3 (33%) 0 -
7 DRUG INTERACTIONS
No formal drug interaction studies have been performed. KRESLADI is not expected to interact with the hepatic cytochrome P-450 family of enzymes or drug transporters.
7.1 Vaccines
The safety and effectiveness of immunization with live viral vaccines during or following KRESLADI treatment has not been studied. Vaccination is not recommended during the 6 weeks preceding the start of myeloablative conditioning, and until hematological recovery following treatment with KRESLADI. Where feasible, administer childhood vaccinations prior to myeloablative conditioning for KRESLADI.
7.2 Anti-retroviral Use
Patients should not take anti-retroviral medications for at least one month prior to initiating medications for stem cell mobilization and for the expected duration for elimination of the medications, and until all cycles of apheresis are completed [see Warnings and Precautions (5.6)]. Anti-retroviral medications may interfere with manufacturing of KRESLADI.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no available data from KRESLADI administration in pregnant women.
No animal reproductive and developmental toxicity studies have been conducted to assess whether KRESLADI can cause fetal harm when administered to a pregnant woman.
No nonclinical germline transmission studies have been conducted with KRESLADI.
KRESLADI must not be administered during pregnancy because of the risk associated with conditioning.
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
8.2 Lactation
Risk Summary
There is no available data regarding the presence of KRESLADI in human milk, the effect on the breastfed infant, and the effects on milk production. Because of the potential risks associated with myeloablative conditioning, discontinue breastfeeding during conditioning. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for KRESLADI and any potential adverse effects on the breastfed child from KRESLADI or from the underlying maternal condition.
8.3 Females and Males of Reproductive Potential
Pregnancy Testing
A negative serum pregnancy test must be confirmed prior to the start of mobilization and re-confirmed prior to conditioning procedures and before administration of KRESLADI in females of childbearing potential.
Contraception
There are insufficient exposure data to provide a precise recommendation on duration of contraception following treatment with KRESLADI. Consult the Prescribing Information of the mobilization and conditioning agents for information on the need for effective contraception.
Infertility
There are no data on the effects of KRESLADI on fertility. Data are available on the risk of infertility with myeloablative conditioning. Advise patients of the option to cryopreserve semen or ova before treatment, if appropriate.
8.4 Pediatric Use
The safety and efficacy of KRESLADI have been established in pediatric patients with severe LAD-I. The use of KRESLADI in the pediatric population was supported by one clinical study which treated 9 patients 9.8 months to 9.8 years of age [see Adverse Reactions (6) and Clinical Studies (14)].
8.5 Geriatric Use
The safety and effectiveness of KRESLADI has not been established in geriatric patients. Clinical studies of KRESLADI for this indication did not include patients aged 65 years and over.
8.6 Patients Seropositive for Human Immunodeficiency Virus (HIV)
KRESLADI has not been studied in patients with HIV-1, HIV-2, HTLV-1, or HTLV-2. A negative serology test for HIV is necessary to ensure acceptance of apheresis material for KRESLADI manufacturing. Apheresis material from patients with a positive test for HIV will not be accepted for KRESLADI manufacturing.
-
11 DESCRIPTION
KRESLADI (marnetegragene autotemcel) is an autologous hematopoietic stem cell (HSC)-based gene therapy prepared from the patient's HSCs, which are collected via apheresis procedure(s). The autologous cells are enriched for CD34+ cells, then transduced ex vivo with LV-RP-201 overnight in the presence of defined growth factors before harvest and suspended in a cryopreservation solution. LV-RP-201 is a replication-incompetent, self-inactivating lentivirus vector encoding for the CD18 β-subunit of human β2 integrins (ITGB2). The ITGB2 gene is under the control of a human chimeric promoter (a fusion of the FES and CTSG minimal 5'-flanking regions).
The transduced CD34+ cells are washed, formulated into a suspension, and then cryopreserved. KRESLADI is frozen in a patient-specific infusion bag(s) and is thawed prior to intravenous administration [see Dosage and Administration (2.1), How Supplied/Storage and Handling (16)]. The thawed product is a colorless to slightly white suspension of cells. Due to the presence of cells, the solution may be clear to slightly cloudy and may contain visible cell aggregates.
The formulation contains 5% (v/v) dimethyl sulfoxide (DMSO) and dextran.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
KRESLADI adds functional copies of the ITGB2 gene into patients' hematopoietic stem cells (HSCs) through transduction of autologous CD34+ cells with LV-RP-201. After KRESLADI infusion, transduced CD34+ HSCs engraft in the bone marrow and differentiate into various cell types, including leukocytes capable of expressing functional CD18 protein. Functional CD18 protein enables formation of the CD18/CD11a heterodimer (Leukocyte Function-Associated Antigen-1, LFA-1) which facilitates leukocyte adhesion to endothelial surfaces and extravasation to infectious and inflammatory sites.
12.2 Pharmacodynamics
In Study RP-L201-0318, Neutrophil CD18 and CD11a surface expression after KRESLADI administration were evaluated [see Clinical Studies (14)].
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No carcinogenicity studies have been performed with KRESLADI.
Intravenous administration of KRESLADI in a mouse model of LAD-I and intravenous administration of KRESLADI manufactured from healthy donors in immunodeficient mice showed no evidence of toxicity, genotoxicity, or oncogenesis (tumorigenicity).
No studies have been conducted to evaluate the effects of KRESLADI on fertility.
-
14 CLINICAL STUDIES
The efficacy of KRESLADI was evaluated in one open-label, single-arm, multicenter study (RP-L201-0318; NCT03812263) in 9 pediatric patients with molecularly confirmed ITGB2-associated severe leukocyte adhesion deficiency-I (LAD-I). Severe LAD-I was defined as having neutrophil CD18 expression <2% or CD11a and/or CD11b expression <2% (if neutrophil CD18 expression ≥2%), documented biallelic ITGB2 mutations, and clinical history consistent with severe LAD-I or a known family history. Patients with an available HLA-identical sibling donor for allogeneic hematopoietic stem cell transplant (HSCT) were excluded.
All patients underwent mobilization with G-CSF and plerixafor followed by apheresis. Three patients underwent a third day of apheresis collection, and one patient required a second mobilization and apheresis cycle to obtain sufficient cells for manufacturing. Eight patients received a subcutaneous dose of ustekinumab 0.75 mg/kg approximately 2 weeks prior to mobilization and at least one dose between mobilization and KRESLADI infusion. Busulfan was administered every 12 hours for 8 doses over 4 days, with pharmacokinetic-guided dosing to achieve a cumulative target AUC of 65,000 ng/mL per hour in the first 3 patients, and 75,000 ng/mL per hour in the remaining patients. Patients received infection prophylaxis and anti-seizure, anti-emetic, and analgesic treatment per institutional guidelines. KRESLADI was administered 25 to 69 hours after the final busulfan dose.
All patients received KRESLADI as a single intravenous infusion with a median dose of 4.3 × 106 CD34+ cells/kg (range: 2.8 to 10 × 106 CD34+ cells/kg). One patient required two cycles of mobilization and apheresis and received two KRESLADI infusions.
The population characteristics were as follows: median age 42 months (range: 9.8 to 117 months), 5 patients (56%) were female, 6 patients (67%) were white, 2 patients (22%) were Asian, and race was not reported for 1 patient (11%). Three of the 9 patients were siblings carrying the same pathogenic ITGB2 variant. No patients had a history of prior allogeneic HSCT. Seven out of 9 patients had baseline CD18 expression <2% and all patients had baseline neutrophil CD11a expression <2%.
The surrogate endpoints for efficacy included improvement in CD18 and CD11a surface expression in neutrophils at Month 12 and sustained expression through Month 24 post-infusion.
Seven out of nine patients had baseline neutrophil CD18 expression <2% and thus were evaluable for post-treatment CD18 assessment. CD18 expression increased in all seven patients after KRESLADI infusion with median CD18 surface expression at Month 12 and Month 24 post-infusion of 54% (range: 20% to 87%) and 50% (range: 16% to 82%), respectively. Neutrophil CD18 expression was sustained through at least Month 42 post-infusion in all seven patients.
Neutrophil CD11a surface expression increased after KRESLADI infusion in all 9 patients. Median CD11a surface expression at Month 12 and Month 24 post-infusion were 45% (range: 18% to 75%) and 39% (range: 17% to 65%), respectively. Neutrophil CD11a surface expression was sustained through at least Month 42 post-infusion in all 9 patients.
None of the 9 treated patients received allogeneic HSCT after product administration. The median duration of follow-up after KRESLADI administration was 4.2 years (range: 3.6 to 5.7 years).
-
16 HOW SUPPLIED/STORAGE AND HANDLING
KRESLADI is supplied in one or two infusion bags containing a frozen suspension of genetically modified autologous cells enriched for CD34+ cells in a cryo-preservative medium containing 5% DMSO and dextran 40. Each infusion bag contains approximately 30 mL and is individually packed within an overwrap in a metal cassette for protection. KRESLADI is shipped from the manufacturing facility to the infusion center in a cryoshipper, which may contain one or two cassettes intended for treatment of a single patient. A Lot Information Sheet is affixed inside the shipper.
- 50 mL infusion bag and metal cassette (NDC: 83537-034-01)
- Match the identity of the patient with the patient identifiers on the cassette(s) and Lot Information Sheet upon receipt.
- Keep the infusion bag(s) in the cassette(s) and store KRESLADI frozen in the vapor phase of liquid nitrogen at less than or equal to -150°C (≤ -238°F) until ready for thaw and administration.
- Thaw KRESLADI prior to infusion [see Dosage and Administration (2)].
- Do not re-freeze after thawing.
- Do not irradiate KRESLADI, as this could lead to inactivation.
-
17 PATIENT COUNSELING INFORMATION
Manufacturing Failure
Ensure that the patient and/or caregiver understand the risk of manufacturing failure. In case of manufacturing failure or the need for additional cells, additional cell collection and manufacturing of KRESLADI would be needed [see Dosage and Administration (2.2)].
Serious Infections
Inform the patient and/or caregiver that patients are at increased risk of developing a variety of infections, which may be due to administration of myeloablative conditioning prior to treatment with KRESLADI. Advise the patient and/or caregiver to contact their healthcare provider if the patient develops any symptoms of infection such as fever, sweats or chills, cough or flu-like symptoms, muscle aches, or warm, red, painful areas on the skin [see Warnings and Precautions (5.1)].
Veno-Occlusive Disease
Inform the patient and/or caregiver of the risk of veno-occlusive disease, which may be due to administration of myeloablative conditioning prior to treatment with KRESLADI. Advise the patient and/or caregiver of the plan to monitor liver function following treatment with KRESLADI [see Warnings and Precautions (5.2)].
Neutrophil Engraftment Failure
Inform the patient and/or caregiver of the potential risk of neutrophil engraftment failure and the need for rescue treatment with their back-up collection of CD34+ cells, if engraftment failure occurs [see Warnings and Precautions (5.3)].
Delayed Platelet Engraftment
Inform the patient that a risk of bleeding or bruising exists after myeloablative conditioning and before platelet engraftment occurs [see Warnings and Precautions (5.4)].
LVV-mediated insertional oncogenesis
Inform the patient and/or caregiver that the risk for insertional oncogenesis is lifelong and the patient will need monitoring for hematologic malignancies via blood tests at least annually for at least 15 years after treatment with KRESLADI. Advise patients that they may require more frequent blood tests and invasive bone marrow biopsies if routine blood tests are concerning for the development of malignancy. Advise patients to have their treating physician contact Rocket Pharmaceuticals, Inc. at 1-800-982-2410 if they are diagnosed with a malignancy [see Warnings and Precautions (5.5)].
Hypersensitivity Reactions
Inform patients to seek immediate medical attention for signs or symptoms associated with hypersensitivity reactions [see Warnings and Precautions (5.6)].
Laboratory Test Interference
Advise patients that they may test positive for HIV if tested using a PCR assay after being treated with KRESLADI [see Warnings and Precautions (5.8)]. Advise the patient and/or caregiver that they should notify any healthcare provider about this possibility prior to being tested for HIV.
Inform patients that they should not donate blood, organs, tissues, or cells at any time in the future [see Dosage and Administration (2.3), Warnings and Precautions (5.9)].
- SPL UNCLASSIFIED SECTION
-
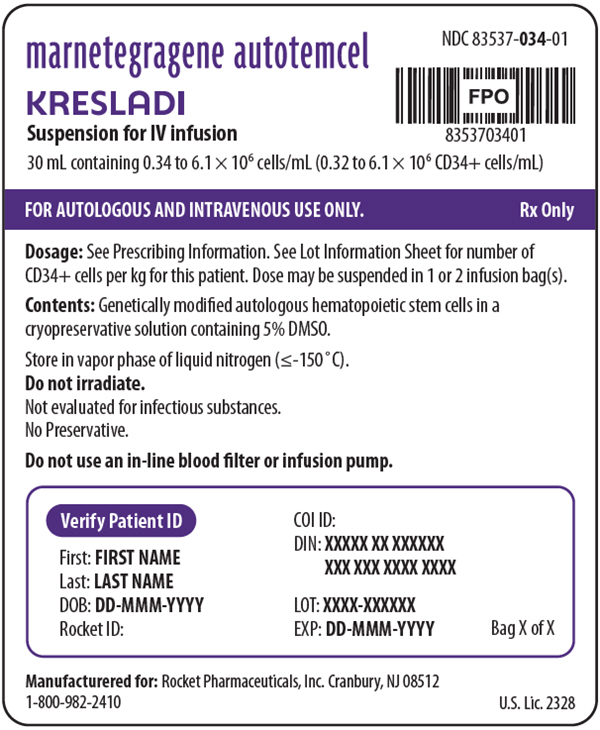
PRINCIPAL DISPLAY PANEL - 30 mL Bag Label
NDC: 83537-034-01
marnetegragene autotemcel
KRESLADI
Suspension for IV infusion30 mL containing 0.34 to 6.1 × 106 cells/mL (0.32 to 6.1 × 106 CD34+ cells/mL)
FOR AUTOLOGOUS AND INTRAVENOUS USE ONLY.
Rx OnlyDosage: See Prescribing Information. See Lot Information Sheet for number of
CD34+ cells per kg for this patient. Dose may be suspended in 1 or 2 infusion bag(s).Contents: Genetically modified autologous hematopoietic stem cells in a
cryopreservative solution containing 5% DMSO.Store in vapor phase of liquid nitrogen (≤-150°C).
Do not irradiate.
Not evaluated for infectious substances.
No Preservative.Do not use an in-line blood filter or infusion pump.
Verify Patient ID
First: FIRST NAME
Last: LAST NAME
DOB: DD-MMM-YYYY
Rocket ID:COI ID:
DIN: XXXXX XX XXXXXX
XXX XXX XXXX XXXX
LOT: XXXX-XXXXXX
EXP: DD-MMM-YYYYBag X of X
Manufacturered for: Rocket Pharmaceuticals, Inc. Cranbury, NJ 08512
1-800-982-2410U.S. Lic. 2328
-
INGREDIENTS AND APPEARANCE
KRESLADI
marnetegragene autotemcel injection, suspensionProduct Information Product Type CELLULAR THERAPY Item Code (Source) NDC: 83537-034 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MARNETEGRAGENE AUTOTEMCEL (UNII: RD86XHH46N) (MARNETEGRAGENE AUTOTEMCEL - UNII:RD86XHH46N) MARNETEGRAGENE AUTOTEMCEL 6100000 in 1 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 83537-034-01 30 mL in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125806 03/26/2026 Labeler - Rocket Pharmaceuticals, Inc. (090253571)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.