OTARMENI- lunsotogene parvec injection, solution
Drug Labeling and Warnings
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use OTARMENI safely and effectively. See full prescribing information for OTARMENI.
OTARMENI™ (lunsotogene parvec-cwha) suspension, for intracochlear infusion
Initial U.S. Approval: 2026INDICATIONS AND USAGE
OTARMENI is an adeno-associated virus vector-based gene therapy indicated for the treatment of pediatric and adult patients with severe-to-profound and profound sensorineural hearing loss (any frequency >90 dB HL) associated with molecularly confirmed biallelic variants in the OTOF gene, preserved outer hair cell function, and no prior cochlear implant in the same ear. (1)
This indication is approved under accelerated approval based on the improvement of hearing sensitivity assessed by average pure tone audiometry (PTA) at Week 24. Continued approval for this indication may be contingent upon verification and description of clinical benefit in the confirmatory clinical trial. (1, 14)
Limitations of Use
OTARMENI is not recommended in patients in whom preoperative imaging demonstrates that access to the inner ear is not feasible including those with abnormal mastoid pneumatization or clinically significant anatomic variations of the middle ear and inner ear. (1)
DOSAGE AND ADMINISTRATION
For intracochlear infusion only.
- Recommended dose of OTARMENI for each ear is 7.2 × 1012 vector genomes (vg) in a total volume of 0.24 mL, administered by a single-dose intracochlear infusion. (2.2)
DOSAGE FORMS AND STRENGTHS
CONTRAINDICATIONS
- None. (4)
WARNINGS AND PRECAUTIONS
- Procedure-Related Risks: Monitor patients for procedure-related adverse reactions with OTARMENI administration. (5.1)
ADVERSE REACTIONS
The most common adverse reactions (incidence ≥5%) include otitis media, vomiting, nausea, dizziness, procedural pain, gait disturbance, and nystagmus. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Regeneron at 1-866-500-GENE or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 4/2026
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Critical Dosing and Administration Information
2.2 Recommended Dosage
2.3 Preparation
2.4 Administration
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Procedure-Related Risks
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.5 Geriatric Use
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
12.6 Immunogenicity
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
13.2 Animal Toxicology and/or Pharmacology
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
OTARMENI is indicated for the treatment of pediatric and adult patients with severe-to-profound and profound sensorineural hearing loss (any frequency >90 dB HL) associated with molecularly confirmed biallelic variants in the OTOF gene, preserved outer hair cell function, and no prior cochlear implant in the same ear.
This indication is approved under accelerated approval based on the improvement of hearing sensitivity assessed by average pure tone audiometry (PTA) at Week 24 [see Clinical Studies (14)]. Continued approval for this indication may be contingent upon verification and description of clinical benefit in the confirmatory clinical trial.
Limitations of Use
OTARMENI is not recommended in patients in whom preoperative imaging demonstrates that access to the inner ear is not feasible including those with abnormal mastoid pneumatization or clinically significant anatomic variations of the middle ear and inner ear.
-
2 DOSAGE AND ADMINISTRATION
2.1 Critical Dosing and Administration Information
- Confirm patient has biallelic, likely pathogenic or pathogenic OTOF variants.
- OTARMENI should be administered by a surgeon experienced in intracochlear surgery and trained in the administration procedure.
- Administer bilateral OTARMENI, if applicable, in a single surgical session.
- For prophylaxis against inflammatory and immunological responses, administer systemic oral corticosteroids equivalent in strength to prednisone at 1 mg/kg/day the day of the OTARMENI infusion, maintain the same dose for two weeks, and taper over the following two weeks. The total corticosteroid dose should not exceed 60 mg/day.
- For prophylaxis against post-operative infection, administer antibiotics before incision.
- Administer age-appropriate vaccination prior to treatment with OTARMENI. Administer vaccines at least 1 month before the first corticosteroid dose and at least 1 month after the last dose.
- Vaccinate patients against micro-organisms that cause meningitis before surgery as meningitis is a known risk of inner ear surgery.
2.2 Recommended Dosage
For intracochlear infusion only.
The recommended dose of OTARMENI for each ear is 7.2 × 1012 vector genomes (vg) in a total volume of 0.24 mL, administered by a single-dose intracochlear infusion.
2.3 Preparation
Prepare syringe containing OTARMENI using sterile technique under aseptic conditions in a Class II biological safety cabinet (BSC). Administer OTARMENI within 4 hours of puncturing the vial. Below is the list of items required for administration syringe preparation. Confirm component packaging and inspect each item prior to use and do not use if any damage is identified.
- One single-dose vial of OTARMENI in a sealed barrier bag (package 1 of 2) [see How Supplied/Storage and Handling (16)]
- Administration Kit for use with OTARMENI (package 2 of 2) [see How Supplied/Storage and Handling (16)]
- Not supplied but required:
- One isopropyl alcohol pad
- One sterile sealable plastic bag
- Secondary container for delivery to surgical suite
- Label and a pen
Preparation of OTARMENI for Infusion
- For each treated ear, thaw one single-dose vial of OTARMENI at room temperature. The contents of the vial will thaw in approximately 30 minutes at room temperature. DO NOT thaw or warm the vial any other way.
- Gently invert the vial 5 times (DO NOT shake or vortex the vial).
- Inspect the OTARMENI vial to ensure that it is thawed, clear to slightly opalescent, and that the vial has no signs of damage. If there is any evidence of cloudiness, particulates, or color, do not use the vial.
- If not used immediately, store the unpunctured thawed vial in the refrigerator for up to 24 hours or at room temperature for up to 8 hours.
- Choose the smallest compatible syringe size, either 1 mL or 3 mL included in the kit, according to the syringe pump parameters. Discard the unused syringe. Do not open the catheter sterile packaging until transferred to the surgical suite.
- Prepare the septum of the OTARMENI vial using an isopropyl alcohol pad to wipe the top. Wait for the septum to dry. Secure the 21G, 1.5-inch needle onto the 1 mL or 3 mL syringe. When filling the syringe for administration, push the needle until it is completely inserted into the vial and the tip touches the bottom edge of the upright vial. Administer OTARMENI within 4 hours of puncturing the vial.
- Withdraw all OTARMENI vial contents into the syringe. Tilt the vial to ensure complete withdrawal. Confirm that at least 0.6 mL is available in the syringe. To prevent the introduction of air, keep the needle bevel submerged in the liquid at the bottom edge of the vial by orienting the bevel towards the vial wall (Figure 1).
Figure 1: 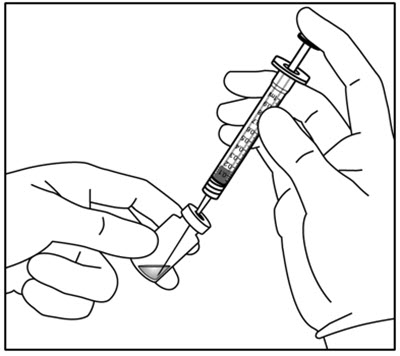
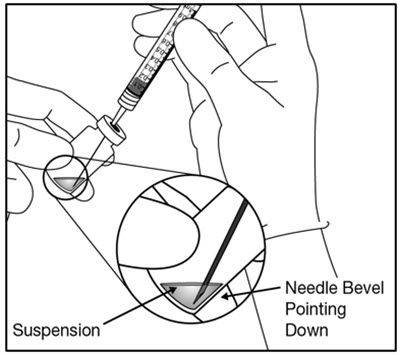
- Holding the syringe with the needle pointing up, check the syringe for bubbles. If there are bubbles, gently tap the syringe with your finger until the bubbles rise to the top.
- Remove the needle and prime the air bubbles out of the syringe by depressing the plunger rod until the air is fully expelled and the meniscus is visible at the syringe opening.
- Cap and label the syringe with the following:
- Lot number of OTARMENI
- Expiration date of OTARMENI
- Time of preparation to ensure that product is administered within 4 hours after puncturing the vial
- Place the capped syringe into the sterile plastic bag and seal the bag. Place the bag into an appropriate secondary container for delivery to the surgical suite.
2.4 Administration
Administer OTARMENI in the surgical suite under general anesthesia with controlled aseptic conditions by a surgeon experienced in intracochlear surgery and trained in the OTARMENI administration procedure.
Administer OTARMENI within 4 hours of puncturing the vial.
The following items are required for administration:
- Syringe containing OTARMENI
- Catheter supplied with Administration Kit for use with OTARMENI (package 2 of 2) [see How Supplied/Storage and Handling (16)]
- Not supplied but required:
- Syringe pump, capable of delivering a volume of 0.24 mL at an infusion rate of 0.9 mL/hr and is compatible with the provided 1 mL or 3 mL syringe sizes
- Electrode insertion forceps
- Fine straight pick
- Mayo stand
Note: Only administer OTARMENI using the provided Administration Kit for use with OTARMENI.
Confirm the availability of OTARMENI (1 vial and 1 Administration Kit per treated ear) prior to administration of anesthesia.
Inspect each item prior to use and do not use if any damage is identified. Do not use if the sterile bag was damaged during transport or if the syringe was transported without the use of a sterile bag.
Follow the steps below for intracochlear infusion:
- Perform a standard mastoidectomy and an opening of the facial recess (also called a posterior tympanotomy). Consider monitoring the facial nerve using intraoperative facial nerve-monitoring (IFNM) throughout the procedure.
- With the help of an assistant, place the syringe pump on a sterile draped Mayo stand (positioned higher than the patient's cochlea) and ensure there is enough distance to allow the catheter to reach the round window membrane.
- Have the non-sterile assistant remove the cap from the syringe and help attach the provided catheter (the stylet will need to be removed from the catheter). Do not flush the catheter.
- Slowly prime the catheter manually with minimal loss by removing all air bubbles until OTARMENI is just visible at the tip of the catheter. The anticipated priming volume is approximately 0.11 mL.
- Have the non-sterile assistant hold the syringe and confirm that at least 0.4 mL of OTARMENI is available in the syringe after priming. Instruct the assistant to place the syringe onto the syringe pump while you maintain control of the catheter in the sterile field.
- Perform a 0.5 to 1 mm fenestration of the lateral semi-circular canal (Figure 2). Do not apply suction near the fenestration of the lateral semi-circular canal.
Figure 2: Lateral Semi-Circular Canal Fenestration 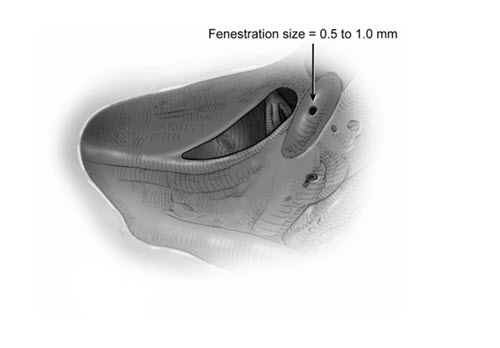
- Perforate the round window membrane using a fine straight pick or equivalent in the anterior-inferior aspect to create an opening that is sufficiently smaller than the size of the catheter to ensure a water-tight seal and prevent fluid backflow at the round window membrane (Figure 3). Do not apply suction near the round window opening. The diameter of the catheter is 1 Fr, 28 G, or 0.33 mm.
Figure 3: Round Window Perforation 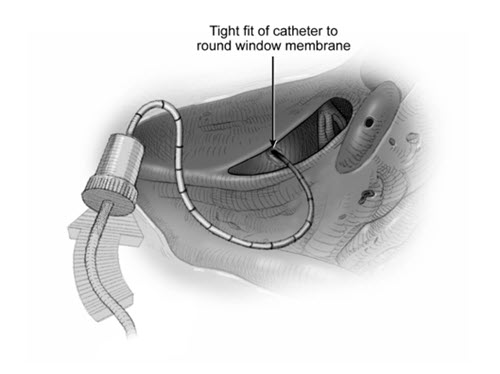
- Have the assistant program the syringe pump by selecting the brand, model, and size of the provided syringe to deliver fluid at 0.9 mL/hr with a total volume of 0.24 mL. To confirm the catheter's priming, have the assistant initiate the pump while the catheter tip is inspected. Once the presence of OTARMENI is noted at the tip of the catheter, stop the pump, and set the volume counter/limiter to zero. Instruct the assistant to note the initial volume in the syringe and verify that at least 0.3 mL of OTARMENI is available.
- Place the catheter tip through the round window membrane using electrode insertion forceps. Insert the catheter until the edge of the first black marker is just visible at the round window (approximately 4 mm), but no further (Figure 4). Do not over-insert the catheter. Allow the catheter to rest passively while minimizing all catheter movement during infusion. During this insertion process, some perilymph might extrude from the newly created fenestration of the lateral semicircular canal.
Figure 4: Catheter Insertion 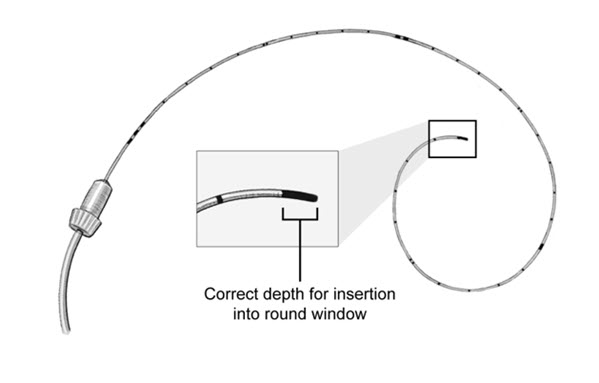
- If the catheter cannot be easily inserted through the round window membrane perforation, enlarge the perforation using a fine straight pick or equivalent.
- After confirming the pump parameters (0.24 mL at 0.9 mL/hr), instruct the assistant to initiate the pump and monitor the progression of the syringe plunger and volume counter during the entire infusion.
- Monitor the infusion continuously to ensure there are no interruptions, including pump alarms/alerts, flow interruption, system malfunction, or power failure. The pump should run continuously for 16 minutes. Monitor the round window membrane continuously for evidence of backflow and the fenestration in the lateral semicircular canal for evidence of forward flow.
IMPORTANT: If the infusion was interrupted for any reason, pause, inspect infusion system to identify the issue, and, using clinical judgement, follow the steps below:- Pause immediately and determine the time of interruption and volume delivered using the syringe markings and volume counter. Maintain catheter position.
- Inspect all infusion components to determine the source of the interruption.
- If a complete infusion of 0.24 mL has been delivered, then proceed to Step 13.
- If 0.24 mL has not been delivered, determine if the infusion can be resumed depending on the length and cause of the interruption, if medically appropriate.
- If the interruption cannot be resolved, abort the procedure. Proceed to Steps 14 – 17.
- After the completion of the infusion (16 minutes), leave the catheter in place for an additional 5-minute hold. Before removing the catheter, instruct the assistant to verify complete dose delivery by reading the final volume on the syringe, which should read approximately 0.24 mL less than the initial reading noted immediately before infusion.
- After the 5-minute hold is completed, remove the catheter from the round window membrane using the electrode insertion forceps. Do not flush the catheter.
- Close the round window membrane and the lateral semi-circular canal with a fascia overlay. Additionally, a bone cap can be used to reinforce the canal fenestration, while avoiding plugging the canal.
- Close the postauricular wound in a typical fashion.
- Following infusion, discard any remaining OTARMENI and disposable material according to local biosafety guidelines.
-
3 DOSAGE FORMS AND STRENGTHS
OTARMENI is a sterile suspension for intracochlear infusion. Each single-dose vial contains a nominal titer of 3.0 × 1013 vg/mL of OTARMENI and has an extractable volume of 0.63 mL.
Following product thaw, the suspension for infusion is a clear to slightly opalescent, colorless liquid, free of visible particulates [see How Supplied/Storage and Handling (16)].
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Procedure-Related Risks
Procedure-related adverse reactions may occur with OTARMENI administration including vertigo, tinnitus, cerebral spinal fluid leak, ipsilateral facial paresis, ipsilateral change in taste, meningitis, wound infection, mastoiditis, numbness around the ear, blood or fluid collection at surgical site, and labyrinthitis.
Monitor patients for procedure-related adverse reactions with OTARMENI administration and manage accordingly.
-
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety data described in this section reflect exposure to OTARMENI in one study (Study DB-OTO-001) which treated 24 patients with OTOF gene associated profound sensorineural hearing loss. A total of 10 patients received unilateral dosing, and a total of 14 patients received bilateral dosing of OTARMENI at a nominal dose of 7.2 × 1012 vg per ear. The median duration of follow-up was 45 weeks (range 9 to 115 weeks) [see Clinical Studies (14)].
Table 1 lists the most common adverse reactions that occurred in ≥5% patients in Study DB-OTO-001.
Table 1: Adverse Reactions Occurring in ≥5% of Patients in Study DB-OTO-001 (N=24) Adverse Reaction Overall
n (%)Otitis Media 9 (38) Vomiting 8 (33) Nausea 7 (29) Dizziness 5 (21) Procedural Pain 4 (17) Gait Disturbance 2 (8) Nystagmus 2 (8) Other clinically significant adverse reactions each occurring in 1 patient included transient balance disorder (4%), abnormal otoacoustic emissions (4%), and wound dehiscence (4%).
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no data from the use of OTARMENI in pregnant women. Animal reproductive and developmental toxicity studies have not been conducted with OTARMENI.
In the US general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
8.2 Lactation
Risk Summary
There are no data on the presence of OTARMENI in human milk, the effects on the breastfed infant, or the effects on milk production.
8.3 Females and Males of Reproductive Potential
Pregnancy Testing
Pregnancy status of females with reproductive potential should be verified. Sexually active females of reproductive potential should have a negative pregnancy test before administering OTARMENI.
Contraception
There are insufficient exposure data to provide a recommendation concerning duration of contraception following treatment with OTARMENI.
Infertility
There are no data on the effects of OTARMENI on fertility.
8.4 Pediatric Use
The safety and effectiveness of OTARMENI have been established in pediatric patients with OTOF gene associated profound sensorineural hearing loss. The use of OTARMENI was supported by evidence from one clinical study that treated 24 pediatric patients aged 10 months to 16 years [see Adverse Reactions (6) and Clinical Studies (14)].
-
11 DESCRIPTION
OTARMENI (lunsotogene parvec-cwha) is an adeno-associated virus vector-based gene therapy for intracochlear infusion. OTARMENI contains a dual adeno-associated virus serotype 1 vector (AAV1) that utilizes an engineered hair cell-specific promoter derived from regulatory elements of myosin 15 (Myo15) to drive complementary DNA (cDNA) expression of human OTOF transcript variant 5 encoding isoform e of the otoferlin protein (OTOF). The vector is produced in human embryonic kidney cells by recombinant DNA technology.
OTARMENI is a sterile, aqueous suspension administered by intracochlear infusion. Each single-dose vial contains a nominal vector genome titer of 3.0 × 1013 vg/mL in an extractable volume of 0.63 mL. Patients receive a total nominal dose of 7.2 × 1012 vg per ear in a total infused volume of 0.24 mL per ear.
OTARMENI is provided in a single-dose 2 mL vial containing a clear to slightly opalescent, colorless suspension, free of visible particulates following thaw from its frozen state. The excipients include sodium phosphate (10 mM), sodium chloride (180 mM), sucrose (5% w/v), and poloxamer 188 (0.001% w/v) with a pH of 7.3.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Intracochlear infusion of OTARMENI results in expression of the OTOF gene primarily in inner hair cells (IHCs), which facilitates production of functional otoferlin protein and restores synaptic transmission to the auditory nerve as shown in animal studies in mice.
12.3 Pharmacokinetics
Biodistribution (within the body) and Vector Shedding (excretion/secretion)
OTARMENI vector DNA levels in various matrices and nonclinical tissues were determined using a qualified quantitative polymerase chain reaction (qPCR) assay. Expression of human OTOF mRNA was determined in selected nonclinical tissues.
Nonclinical data
Systemic distribution of vector DNA was evaluated in cynomolgus monkeys at weeks 7 and 27 after a single bilateral intracochlear infusion of dose levels up to 4.4 × 1012 vg/ear (7.3 × 1013 vg/mL), and in juvenile mice at days 30 and 90 and in adult mice at days 30 and 60 after a single unilateral inner ear infusion of dose levels up to 1.3 × 1011 vg/ear (6.5 × 1013 vg/mL).
The highest levels of vector DNA were detected in the cochleae. Human OTOF mRNA expression in the cochleae was sustained over the study durations (27 weeks in monkeys, 60-90 days in mice), with peak expression at 6 weeks in monkeys and 4 weeks in mice, then plateauing through the final time points.
Vector DNA distribution to non-otic tissues was limited, with levels at least 10-fold lower than cochlear levels in monkeys and at least 3-fold lower in mice. The highest levels of OTARMENI vector DNA were detected in the parotid lymph node, cervical dorsal root ganglion, cervical spinal cord, and brain, while sporadic or low levels were detected in other non-otic tissues. No measurable vector DNA was detected in ovaries, and only minimal levels were detected in testes (1 of 4 mouse samples near detection limit).
Human OTOF mRNA expression in non-otic tissues was restricted to neural tissues at substantially lower levels than in the cochlea. In monkeys, human OTOF mRNA was detected only in cervical and lumbar dorsal root ganglia of one monkey at Week 7 (no detection at Week 27). In mice, human OTOF mRNA was detected in brain, cervical dorsal root ganglia, and cervical spinal cord at levels 4.9- to 96.4-fold lower than cochlear expression. No human OTOF mRNA was detected in reproductive tissues or other non-neural tissues in either species.
Clinical data
Systemic distribution of vector DNA in plasma and vector DNA shedding in urine, saliva, and stool were evaluated in 15 patients administered OTARMENI at the dose of 7.2 × 1012 vg/ear in Study DB-OTO-001. Samples were collected at baseline, at Day 1, and Week 2, 4, 6, 12, 24, and 36 following OTARMENI administration. Vector DNA was detectable in plasma, urine, and saliva at Day 1 after OTARMENI administration and was undetectable by Week 2 (the next sampling timepoint). In stool, vector DNA was detectable through Week 6 and became undetectable by Week 12. Peak concentration was observed at Day 1 after OTARMENI administration and declined substantially thereafter.
12.6 Immunogenicity
The observed incidence of anti-AAV1 neutralizing antibodies and anti-OTOF antibodies is highly dependent on the sensitivity and specificity of the assays. Differences in assay methods preclude meaningful comparisons of the incidence of antibodies in the study described below with the incidence of antibodies in other studies.
In the DB-OTO-001 study, patients were not excluded based on pre-existing serum anti-AAV1 neutralizing antibodies. Immunogenicity was assessed from baseline through Week 12.
A total of 12/15 (80%) patients had anti-AAV1 neutralizing antibodies at baseline (titer <1,000). All patients developed anti-AAV1 neutralizing antibody responses through Week 12 with 10/15 (67%) having titer >10,000, 3/15 (20%) having titer 1,000 to 10,000, and 2/15 (13%) having titer <1,000 responses.
A total of 1/15 (7%) patients had anti-OTOF antibodies at baseline (titer <1,000). A total of 7/15 (47%) patients developed anti-OTOF antibody responses through Week 12. Of the 7 patients who developed anti-OTOF responses, 6 had titer <1,000 and 1 had titer between 1,000 and 10,000.
There were no identified effects of immunogenicity on the safety or efficacy of OTARMENI.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No animal studies have been performed to evaluate the effects of OTARMENI on carcinogenicity, mutagenesis, or impairment of fertility.
13.2 Animal Toxicology and/or Pharmacology
In a 6-month study, cynomolgus monkeys (22 to 63 months old) received bilateral intracochlear administration of OTARMENI at dose levels up to 4.4 × 1012 vg per ear. Vehicle control animals underwent the same surgical procedure, which was designed to closely mimic the intended clinical procedure.
Procedure-related complications were observed across all treatment groups, including vehicle controls. Facial nerve paralysis occurred in 6 of 36 animals (3 of 12 control animals and 3 of 24 OTARMENI-administered animals). Of the OTARMENI-administered animals that developed facial paralysis, 2 showed complete recovery by Day 3, while 1 required euthanasia on Day 3 due to severe facial paralysis and associated corneal complications. The 3 control animals with facial paralysis did not recover by their Week 7 euthanasia time point. Permanent auditory damage was documented, including spiral ganglion cell degeneration and hair cell loss in the cochlea observed in 2 of 6 control animals and 7 of 13 OTARMENI-administered animals at Week 27. Damage was primarily limited to the injection site area in the basal turn of the cochlea, though one animal experienced more extensive cochlear damage. Hearing threshold elevations affected 55-84% of ears at high frequencies (8-16 kHz) and 21-33% of ears at low frequencies (1-4 kHz), with no recovery observed over the 6-month study duration. Additional post-operative adverse reactions included vestibular symptoms (lasting through Day 5) such as ataxia, head tilt, and coordination problems.
-
14 CLINICAL STUDIES
The efficacy of OTARMENI was evaluated in an ongoing, multi-center, single-arm study (Study DB-OTO-001; NCT05788536). The study enrolled pediatric patients with molecularly confirmed OTOF gene associated profound sensorineural hearing loss (defined as >90 dB HL on pure tone audiometry averaged across 0.5, 1, 2, and 4 kHz and auditory brainstem response), evidence of outer hair cell activity by otoacoustic emissions testing, and who were considered candidates for cochlear implant. Patients with cochlear implants in the same ear to be treated with OTARMENI were excluded from the study.
A total of 24 patients (10 patients received unilateral treatment and 14 patients received bilateral treatment) received a single dose of OTARMENI at a dose of 7.2 × 1012 vector genomes (vg) in 0.24 mL per ear.
The demographic characteristics of the population (N=24): The median age was 2 years (range 10 months to 16 years), 16 patients (67%) were female, 17 patients (71%) were White, 4 patients (17%) were Asian, and 3 patients (13%) had race not reported. Eleven patients (46%) were Hispanic.
Of the 24 patients, 20 had completed efficacy assessments at 24 weeks. Among these 20 patients, five had a contralateral cochlear implant in place prior to enrollment, and two received a cochlear implant in the contralateral ear during the same surgical session for OTARMENI administration. Two patients had bilateral hearing aids before OTARMENI treatment.
The primary efficacy endpoint was the achievement of a hearing sensitivity threshold ≤70 dB HL assessed by average pure tone audiometry (i.e., average of PTA thresholds at 0.5, 1.0, 2.0, and 4.0 kHz) at Week 24 after product administration. The key secondary efficacy endpoint was the presence of auditory brainstem response (ABR) to a click (broadband sound) stimulus of ≤90 dB nHL at Week 24 after product administration.
Efficacy results for Study DB-OTO-001 are presented in Table 2 below.
Table 2: Efficacy Results for Study DB-OTO-001 Endpoint N Participants Achieving Threshold/Response* ABR: Auditory Brainstem Response; CI: Confidence Interval; PTA: Pure Tone Audiometry - * For patients who were treated bilaterally, the data reflect the treated ear with better average PTA/better ABR to click at Week 24.
- † Patient without available Week 24 data (n=1) was imputed as a non-responder.
Primary Endpoint Achievement of a hearing sensitivity threshold of ≤70 dB HL assessed by average PTA 20†
n (%) 16 (80%) 95% CI 56%, 94% Key Secondary Endpoint ABR to Click at ≤90 dB nHL 20 n (%) 14 (70%) 95% CI 46%, 88% At Week 24, a total of 9/20 (45%) patients achieved an average PTA threshold ≤45 dB HL (ability to hear soft conversational speech level) and 3/20 (15%) achieved ≤25 dB HL (normal hearing level i.e., ability to hear whispers).
One patient received a cochlear implant as rescue treatment approximately 8 months after OTARMENI administration in the same ear following determination of treatment failure.
Among the 20 patients, 12 patients were evaluated at Week 48 after product administration. Of these, all 9 patients who previously had a response at Week 24 and who were evaluated at Week 48 maintained their response. One additional patient who had not initially achieved response at Week 24, achieved an average PTA threshold ≤70 dB HL by Week 48, resulting in a total of 10/12 (83%) patients who achieved an average PTA threshold ≤70 dB HL by Week 48. Eight of the 12 (67%) patients achieved an average PTA threshold ≤45 dB HL and 5 of the 12 patients (42%) achieved ≤25 dB HL by Week 48. Additionally, 9 of the 12 patients (75%) demonstrated presence of auditory brainstem response (ABR) to a click (broadband sound) stimulus of ≤90 dB nHL by Week 48.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
OTARMENI is supplied as a single-dose vial enclosed in a sealed barrier bag. Following thaw from its frozen state, OTARMENI is a sterile, clear to slightly opalescent colorless liquid, free of visible particulates. Each vial contains a nominal titer of 3.0 × 1013 vg/mL of OTARMENI and has an extractable volume of 0.63 mL.
The Administration Kit for use with OTARMENI is supplied separately as package 2 of 2 [see Dosage and Administration (2.3 and 2.4)].
Contents of Package 1 NDC Number OTARMENI 2 mL vial 61755-062-00 OTARMENI vial carton 61755-062-01 OTARMENI sealed barrier bag 61755-062-99 Contents of Package 2 NDC Number Administration Kit for use with OTARMENI - One BD PrecisionGlide™ 21G, 1.5-inch sterile needle
- One BD 1 mL sterile syringe, Luer-Lok™ tip
- One BD PlastiPak™ 3 mL sterile syringe, Luer-Lok™ tip
- One BD Luer-Lok™ syringe tip cap
- One Vygon Premicath® catheter
61755-062-11 Storage and Handling
OTARMENI suspension (2 mL vial):
Store and transport OTARMENI (package 1) frozen at -80°C (-112°F). Keep the vial in the supplied carton. Thaw OTARMENI at room temperature prior to administration. If not used immediately, the unpunctured thawed vial can be stored in the refrigerator (2°C to 8°C [36°F to 46°F]) for up to 24 hours or at room temperature (up to 25°C [77°F]) for up to 8 hours [see Dosage and Administration (2.3)]. Do not refreeze vial once thawed.
Administration Kit for use with OTARMENI:
Store at room temperature in original carton separately from OTARMENI until time of use. Do not freeze. Avoid excessive heat and direct sunlight.
-
17 PATIENT COUNSELING INFORMATION
Inform patients or caregivers that:
- Prior to treatment with OTARMENI, it is recommended that patients receive the appropriate vaccinations for their age. Vaccines should be administered at least 1 month before the first corticosteroid dose and at least 1 month after the last dose [see Dosage and Administration (2.1)].
- Before surgery, patients should consult their primary care physician and surgeon regarding vaccination status against microorganisms that cause meningitis. Meningitis is a known risk of inner ear surgery [see Dosage and Administration (2.1)].
- Procedural-related adverse reactions may occur following OTARMENI administration. These may include vertigo, tinnitus, cerebral spinal fluid leak, ipsilateral facial paresis, ipsilateral change in taste, meningitis, wound infection, mastoiditis, numbness around the ear, blood or fluid collection at surgical site, and labyrinthitis. [see Warnings and Precautions (5.1)].
- Temporary vector shedding of OTARMENI occurs primarily through body waste. Practice proper hand hygiene, such as hand washing, when coming into direct contact with patient body waste. Place potentially contaminated materials that may have the patient's bodily fluids/waste in a sealable bag and dispose into regular trash. These precautions should be followed for 2 weeks after OTARMENI infusion [see Clinical Pharmacology (12.3)].
- SPL UNCLASSIFIED SECTION
-
PRINCIPAL DISPLAY PANEL - 0.63 mL Vial Carton Barrier Bag Label
NDC: 61755-062-99
Rx onlylunsotogene parvec-cwha
Otarmeni™
3.0 x 1013 vg/mL
Suspension for Intracochlear Infusion
Single-Dose Vial, 0.63 mL per vial.
Discard unused portion.Store frozen at -80°C (-112°F).
Manufactured by:
Regeneron Pharmaceuticals, Inc.
U.S. License No. 1760
-
PRINCIPAL DISPLAY PANEL - 0.63 mL Vial Carton
NDC: 61755-062-01
Rx only
lunsotogene parvec-cwha
Otarmeni™
3.0 x 1013 vg/mL
Suspension for Intracochlear Infusion
Single-Dose Vial. Discard unused portion.
Do not use if seal is broken or damaged.Package 1 of 2
The Administration Kit for use with
OTARMENI is supplied separately
as package 2 of 2.OTARMENI must be
administered with the
Administration Kit provided.Single-Dose Vial,
0.63 mL per vialREGENERON
-
PRINCIPAL DISPLAY PANEL - 0.63 mL Vial Label
NDC: 61755-062-00
Rx onlylunsotogene parvec-cwha
Otarmeni™3.0 x 1013 vg/mL
Suspension for Intracochlear Infusion
Single-Dose Vial, 0.63 mL per vial.
Discard unused portion.
-
PRINCIPAL DISPLAY PANEL - Device Kit Carton
NDC: 61755-062-11
Rx only
Administration Kit
FOR USE ONLY WITHlunsotogene parvec-cwha
Otarmeni™
For Intracochlear Infusion Only
Dosage: See enclosed prescribing information.
Contents of Administration Kit:
- One BD PrecisionGlide™ 21G, 1.5-inch sterile needle
- One BD 1 mL sterile syringe, Luer-Lok™ tip
- One BD PlastiPak™ 3 mL sterile syringe, Luer-Lok™ tip
- One BD Luer-Lok™ syringe tip cap
- One Vygon Premicath® Catheter
Package 2 of 2
OTARMENI vial is supplied separately in package 1 of 2.
Caution:
- –
Store at room temperature in original carton separately
from OTARMENI until time of use. Do not freeze. - – Do not use if seal is broken or damaged.
- – Single use only. Do not reuse.
- – Avoid excessive heat and direct sunlight.
NOTE:
- –
This Administration Kit has no components made of natural
latex rubber. - – Individual devices are provided in separate sterile packaging.
- – Use the expiration date on this carton.
REGENERON

-
INGREDIENTS AND APPEARANCE
OTARMENI
lunsotogene parvec injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 61755-062 Route of Administration INTRACOCHLEAR Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength lunsotogene parvec 3' vector (UNII: PA2E3JN26W) (lunsotogene parvec 3' vector - UNII:PA2E3JN26W) lunsotogene parvec 3' vector 30000000000000 {GC} in 1 mL Inactive Ingredients Ingredient Name Strength sodium phosphate (UNII: SE337SVY37) sodium chloride (UNII: 451W47IQ8X) sucrose (UNII: C151H8M554) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 61755-062-99 1 in 1 BAG 04/23/2026 1 NDC: 61755-062-01 1 in 1 CARTON 1 NDC: 61755-062-00 1 mL in 1 VIAL, SINGLE-DOSE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125874 04/23/2026 Labeler - Regeneron Pharmaceuticals, Inc. (194873139) Establishment Name Address ID/FEI Business Operations Regeneron Pharmaceuticals, Inc. 945589711 API MANUFACTURE(61755-062) , ANALYSIS(61755-062) Establishment Name Address ID/FEI Business Operations Brammer Bio MA, LLC 118645749 ANALYSIS(61755-062) Establishment Name Address ID/FEI Business Operations Catalent Maryland, Inc. (BWI) 116950534 MANUFACTURE(61755-062) Establishment Name Address ID/FEI Business Operations BioReliance Corporation 119271065 ANALYSIS(61755-062) Establishment Name Address ID/FEI Business Operations Charles River Laboratories, Inc. 125469424 ANALYSIS(61755-062) Establishment Name Address ID/FEI Business Operations Nelson Laboratories, LLC 766951479 ANALYSIS(61755-062) Establishment Name Address ID/FEI Business Operations Eurofins Lancaster Laboratories, LLC 069777290 ANALYSIS(61755-062) Establishment Name Address ID/FEI Business Operations Primera Analytical Solutions Corporation 080456034 ANALYSIS(61755-062) Establishment Name Address ID/FEI Business Operations Fisher BioServices, Inc. 081352696 LABEL(61755-062) , PACK(61755-062) Establishment Name Address ID/FEI Business Operations Fisher Clinical Services, Inc. 199879800 LABEL(61755-062) , PACK(61755-062) Establishment Name Address ID/FEI Business Operations Charles River Laboratories 078495006 ANALYSIS(61755-062)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.