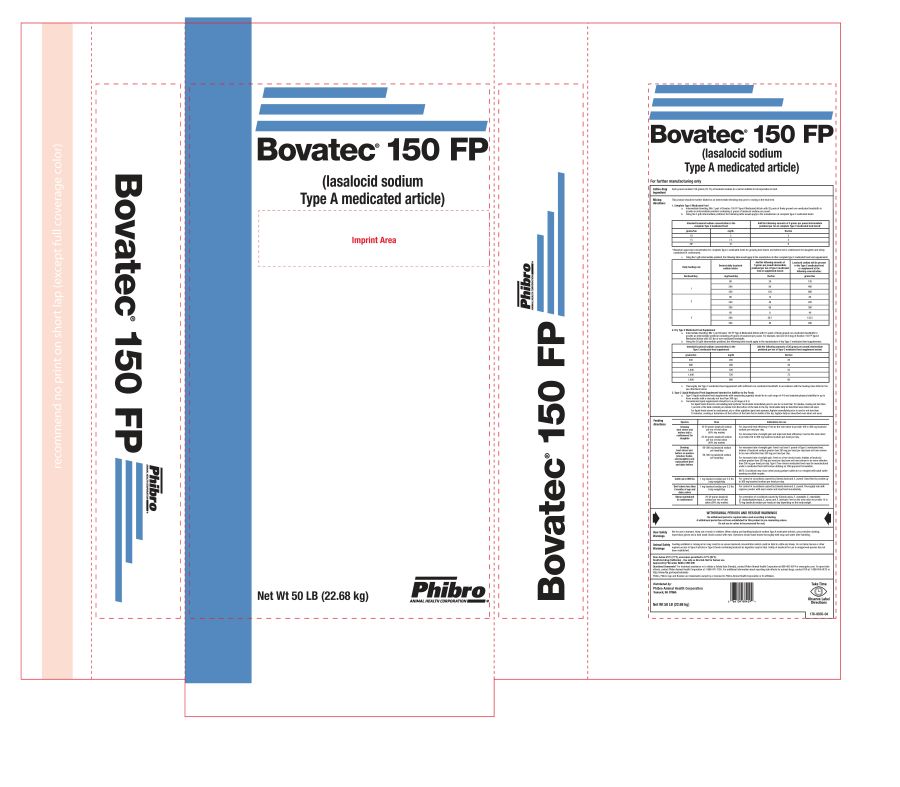
BOVATEC 150FP- lasalocid sodium powder
Drug Labeling and Warnings
Drug Details [pdf]
- SPL UNCLASSIFIED SECTION
- Active drug ingredient
-
Mixing directions
This product should be further diluted in an intermediate blending step prior to mixing in the final feed.
1. Complete Type C Medicated Feed
a. Intermediate blending: Mix 1 part of Bovatec 150 FP (Type A Medicated Article) with 29 parts of finely ground non-medicated feedstuffs to provide an intermediate preblend containing 5 grams of lasalocid sodium per pound.
b. Using the 5 g/lb intermediate preblend, the following table would apply to the manufacture of complete Type C medicated feeds:Intended lasalocid sodium concentration in the complete Type C medicated feed Add the following amounts of 5 grams per pound intermediate preblend per ton of complete Type C medicated feed mixed: grams/ton mg/lb lbs/ton 10 5 2 15 7.5 3 30* 15 6 * Maximum approved concentration for complete Type C medicated feeds for growing beef steers and heifers fed in confinement for slaughter and sheep maintained in confinement.
c. Using the 5 g/lb intermediate preblend, the following table would apply to the manufacture of other complete Type C medicated feeds and supplements:Daily feeding rate Desired daily lasalocid sodium intake Add the following amounts of 5 grams per pound intermediate preblend per ton of Type C medicated feed or supplement mixed: Lasalocid sodium will be present in the Type C medicated feed or supplement at the following concentration: lbs/head/day mg/head/day lbs/ton grams/ton 1 60 24 120 200 80 400 300 120 600 2 60 12 60 200 40 200 300 60 300 3 60 8 40 200 26.7 133.5 300 40 200 2. Dry Type C Medicated Feed Supplement
a. Intermediate blending: Mix 1 part Bovatec 150 FP Type A Medicated Article with 6.5 parts of finely ground non-medicated feedstuffs to provide an intermediate preblend containing 20 grams of lasalocid per pound. For example, mix one 50 lb bag of Bovatec 150 FP Type A Medicated Article with 325 lbs of non-medicated feedstuffs.
b. Using the 20 g/lb intermediate preblend, the following table would apply to the manufacture of dry Type C medicated supplements:Intended lasalocid sodium concentration in the Type C medicated feed supplement Add the following amounts of 20 grams per pound intermediate preblend per ton of Type C medicated feed supplement mixed: grams/ton mg/lb lbs/ton 400 200 20 600 300 30 1000 500 50 1440 720 72 1600 800 80 c. Thoroughly mix Type C medicated feed supplement with sufficient non-medicated feedstuffs in accordance with the feeding rates listed in the use directions below.
3. Type C Liquid Medicated Feed Supplement Intended for Addition to Dry Feeds
a. Type C liquid medicated feed supplements with suspending agent(s) should be in a pH range of 4-8 and maintain physical stability for up to three months with a viscosity not less than 300 cps.
b. Conventional liquid supplements should be in a pH range of 4-8.
For liquid feeds stored in recirculating tank systems: Recirculate immediately prior to use for no less than 10 minutes, moving not less than 1 percent of the tank contents per minute from the bottom of the tank to the top. Recirculate daily as described even when not used.
For liquid feeds stored in mechanical, air, or other agitation-type tank systems: Agitate immediately prior to use for not less than 10 minutes, creating a turbulence at the bottom of the tank that is visible at the top. Agitate daily as described even when not used. -
Feeding directions
Species Dose Indications for use Growing
beef steers and
heifers fed in
confinement for
slaughter10-30 grams lasalocid sodium
per ton of total ration
(90% dry matter)For improved feed efficiency: Feed as the sole ration to provide 100 to 360 mg lasalocid sodium per head per day. 25-30 grams lasalocid sodium
per ton of total ration
(90% dry matter)For increased rate of weight gain and improved feed efficiency: Feed as the sole ration to provide 250 to 360 mg lasalocid sodium per head per day. Growing
beef steers and
heifers on pasture
(stocker, feeder,
and slaughter) and
replacement beef
and dairy heifers60-300 mg lasalocid sodium per
head/dayFor increased rate of weight gain: Feed in at least 1 pound of Type C medicated feed. Intakes of lasalocid sodium greater than 200 mg per head per day have not been shown to be more effective than 200 mg per head per day. 60-300 mg lasalocid sodium per
head/dayFor increased rate of weight gain: Feed on a free-choice basis. Intakes of lasalocid sodium greater than 200 mg per head per day have not been shown to be more effective than 200 mg per head per day. Type C free-choice medicated feed must be manufactured under a medicated feed mill license utilizing an FDA approved formulation.
NOTE: Coccidiosis may occur when young pasture cattle are co-mingled with adult cattle passing coccidial oocysts. Cattle up to 800 lbs 1 mg lasalocid sodium per 2.2 lbs
body weight/dayFor control of coccidiosis caused by Eimeria bovis and E. zuernii. Hand feed to provide up to 360 mg lasalocid sodium per head per day. Beef calves less than
2 months of age and
dairy calves1 mg lasalocid sodium per 2.2 lbs
body weight/dayFor control of coccidiosis caused by Eimeria bovis and E. zuernii. Thoroughly mix milk replacer powder with warm water and hand feed immediately.
Sheep maintained
in confinement20-30 grams lasalocid sodium
per ton of total ration
(90% dry matter)For prevention of coccidiosis caused by Eimeria ovina, E. crandallis, E. ovinoidalis (E. ninakohlyakimovae), E. parva, and E. intricata: Feed as the sole ration to provide 15 to 70 mg lasalocid sodium per head per day depending on the body weight.
- WITHDRAWAL PERIODS AND RESIDUE WARNINGS
- User Safety Warnings
-
Animal Safety Warnings
Feeding undiluted or mixing errors may result in an excess lasalocid concentration which could be fatal to cattle and sheep. Do not allow horses or other
equines access to Type A articles or Type B feeds containing lasalocid as ingestion may be fatal. Safety of lasalocid for use in unapproved species has not
been established. -
SPL UNCLASSIFIED SECTION
Questions/Comments? For technical assistance or to obtain a Safety Data Sheet(s), contact Phibro Animal Health Corporation at 888-403-0074 or www.pahc.com. To report side
effects, contact Phibro Animal Health Corporation at 1-888-475-7355. For additional information about reporting side effects for animal drugs, contact FDA at 1-888-FDA-VETS or
http://www.fda.gov/reportanimalaePhibro, Phibro logo and Bovatec are trademarks owned by or licensed to Phibro Animal Health Corporation or its affiliates.
Distributed by:
Phibro Animal Health Corporation
Teaneck, NJ 07666
Net Wt 50 LB (22.68 kg)
176-9035-04

Take TIme
- Storage Conditions
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
BOVATEC 150FP
lasalocid sodium powderProduct Information Product Type OTC TYPE A MEDICATED ARTICLE ANIMAL DRUG Item Code (Source) NDC: 66104-1457 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LASALOCID SODIUM (UNII: W2S5C71Y3G) (LASALOCID - UNII:W7V2ZZ2FWB) LASALOCID SODIUM 150 g in 0.45 kg Product Characteristics Color white (white to tan) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 66104-1457-1 22.68 kg in 1 BAG Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NADA NADA096298 01/01/2009 Labeler - Phibro Animal Health (006989008)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.