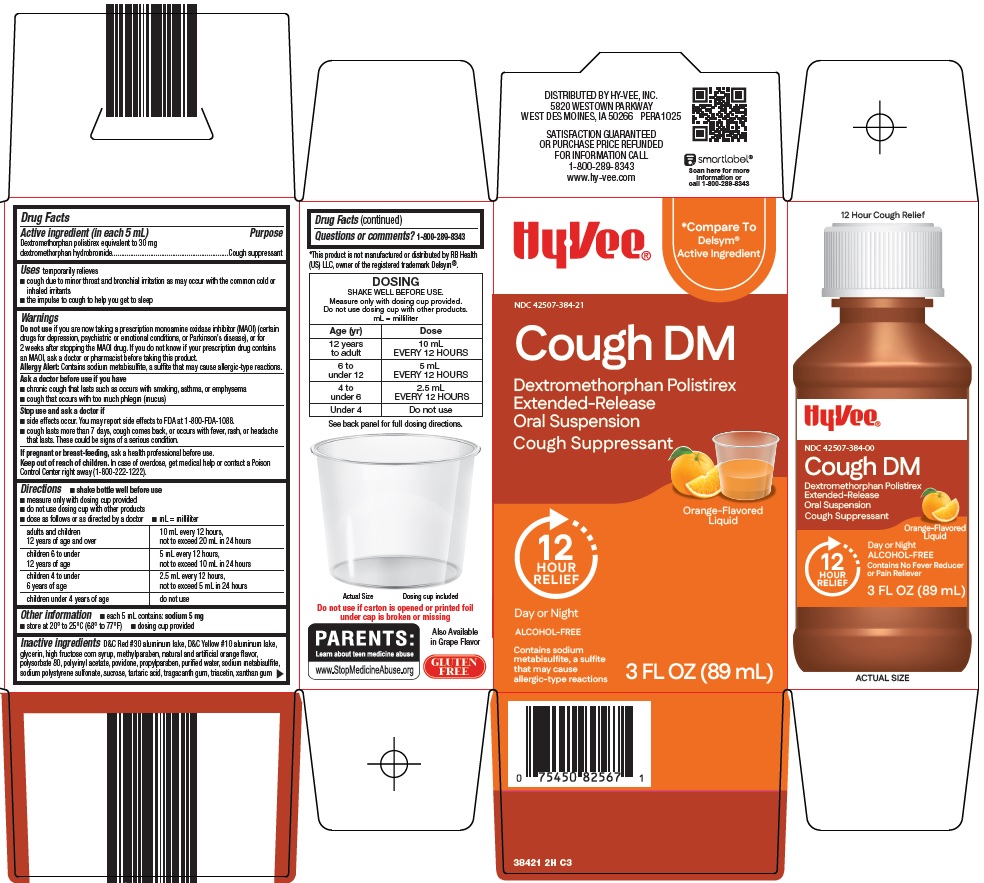
COUGH DM- dextromethorphan polistirex suspension
cough dm by
Drug Labeling and Warnings
cough dm by is a Otc medication manufactured, distributed, or labeled by HyVee Inc. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Active ingredient (in each 5 mL)
- Purpose
- Uses
-
Warnings
Do not use
if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Allergy Alert: Contains sodium metabisulfite, a sulfite that may cause allergic-type reactions.
Ask a doctor before use if you have
- chronic cough that lasts as occurs with smoking, asthma, or emphysema
- cough that occurs with too much phlegm (mucus)
-
Directions
- shake bottle well before use
- measure only with dosing cup provided
- do not use dosing cup with other products
- dose as follows or as directed by doctor
- mL = milliliter
adults and children 12 years of age and over
10 mL every 12 hours, not to exceed 20 mL in 24 hours
children 6 to under 12 years of age
5 mL every 12 hours, not to exceed 10 mL in 24 hours
children 4 to under 6 years of age
2.5 mL every 12 hours, not to exceed 5 mL in 24 hours
children under 4 years of age
do not use
- Other information
-
Inactive Ingredients
D&C Red #30 aluminum lake, D&C Yellow #10 aluminum lake, glycerin, high fructose corn syrup, methylparaben, natural and artificial orange flavor, polysorbate 80, polyvinyl acetate, povidone, propylparaben, purified water, sodium metabisulfite, sodium polystyrene sulfonate, sucrose, tartaric acid, tragacanth gum, triacetin, xanthan gum
- Questions or comments?
- Package/Label Principal Display Panel
-
INGREDIENTS AND APPEARANCE
COUGH DM
dextromethorphan polistirex suspensionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 42507-384 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DEXTROMETHORPHAN HYDROBROMIDE (UNII: 9D2RTI9KYH) (DEXTROMETHORPHAN - UNII:7355X3ROTS) DEXTROMETHORPHAN HYDROBROMIDE 30 mg in 5 mL Inactive Ingredients Ingredient Name Strength D&C RED NO. 30 ALUMINUM LAKE (UNII: GE75M6YV5W) D&C YELLOW NO. 10 ALUMINUM LAKE (UNII: CQ3XH3DET6) GLYCERIN (UNII: PDC6A3C0OX) HIGH FRUCTOSE CORN SYRUP (UNII: XY6UN3QB6S) METHYLPARABEN (UNII: A2I8C7HI9T) POLYSORBATE 80 (UNII: 6OZP39ZG8H) POLYVINYL ACETATE (UNII: 32K497ZK2U) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) PROPYLPARABEN (UNII: Z8IX2SC1OH) WATER (UNII: 059QF0KO0R) SODIUM METABISULFITE (UNII: 4VON5FNS3C) SODIUM POLYSTYRENE SULFONATE (UNII: 1699G8679Z) SUCROSE (UNII: C151H8M554) TARTARIC ACID (UNII: W4888I119H) TRAGACANTH (UNII: 2944357O2O) TRIACETIN (UNII: XHX3C3X673) XANTHAN GUM (UNII: TTV12P4NEE) Product Characteristics Color ORANGE Score Shape Size Flavor ORANGE Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 42507-384-21 1 in 1 CARTON 09/02/2012 1 NDC: 42507-384-00 89 mL in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC: 42507-384-28 1 in 1 CARTON 08/05/2013 2 NDC: 42507-384-01 148 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA091135 09/02/2012 Labeler - HyVee Inc (006925671)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.