HYDROCORTISONE SODIUM SUCCINATE injection
Drug Labeling and Warnings
Drug Details [pdf]
- SPL UNCLASSIFIED SECTION
-
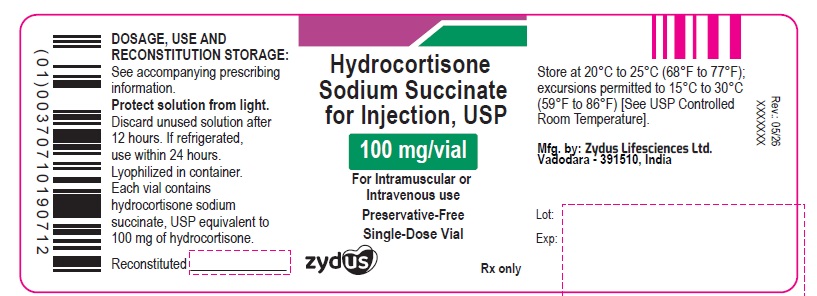
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
NDC: 70771-1966-1
Hydrocortisone sodium succinate for injection, USP
100 mg/vial
For Intravenous or Intramuscular use
Rx only
-
INGREDIENTS AND APPEARANCE
HYDROCORTISONE SODIUM SUCCINATE
hydrocortisone sodium succinate injectionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 70771-1966 Route of Administration INTRAMUSCULAR, INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HYDROCORTISONE SODIUM SUCCINATE (UNII: 50LQB69S1Z) (HYDROCORTISONE - UNII:WI4X0X7BPJ) HYDROCORTISONE 100 mg in 2 mL Inactive Ingredients Ingredient Name Strength SODIUM PHOSPHATE, DIBASIC, ANHYDROUS (UNII: 22ADO53M6F) SODIUM PHOSPHATE, MONOBASIC, ANHYDROUS (UNII: KH7I04HPUU) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 70771-1966-1 1 in 1 CARTON 05/09/2026 1 2 mL in 1 VIAL; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA217191 05/09/2026 Labeler - Zydus Lifesciences Limited (918596198) Establishment Name Address ID/FEI Business Operations Zydus Lifesciences Limited 873671928 MANUFACTURE(70771-1966) , ANALYSIS(70771-1966) , API MANUFACTURE(70771-1966)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.