NIGHT TIME COLD AND FLU RELIEF LIQUID FILLED SOFTGELS- acetaminophen,dextromethrophan hbr,doxylamine succinate capsule, liquid filled
Drug Labeling and Warnings
Drug Details [pdf]
- ACTIVE INGREDIENT
- INDICATIONS & USAGE
- PURPOSE
-
WARNINGS
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take: more than 4 doses in
24 hours, which is the maximum daily amount for this product with other drugs containing acetaminophen 3 or
more alcoholic drinks daily while using this product.Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include: skin reddening blisters
rash If a skin reaction occurs, stop use and seek medical help right away.Sore throat warning: If sore throat is severe, lasts for more than 2 days, occurs with or is followed by fever, headache,
rash, nausea or vomiting, consult a doctor promptly. -
DO NOT USE
Do not use with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure
whether a drug contains acetaminophen, ask a doctor or pharmacist if you are now taking a prescription monoamine
oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or
for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or
pharmacist before taking this product to make a child sleep - ASK DOCTOR
- ASK DOCTOR/PHARMACIST
- WHEN USING
- STOP USE
- PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
- DOSAGE & ADMINISTRATION
- WHEN USING
- OTHER SAFETY INFORMATION
- INACTIVE INGREDIENT
- QUESTIONS
-
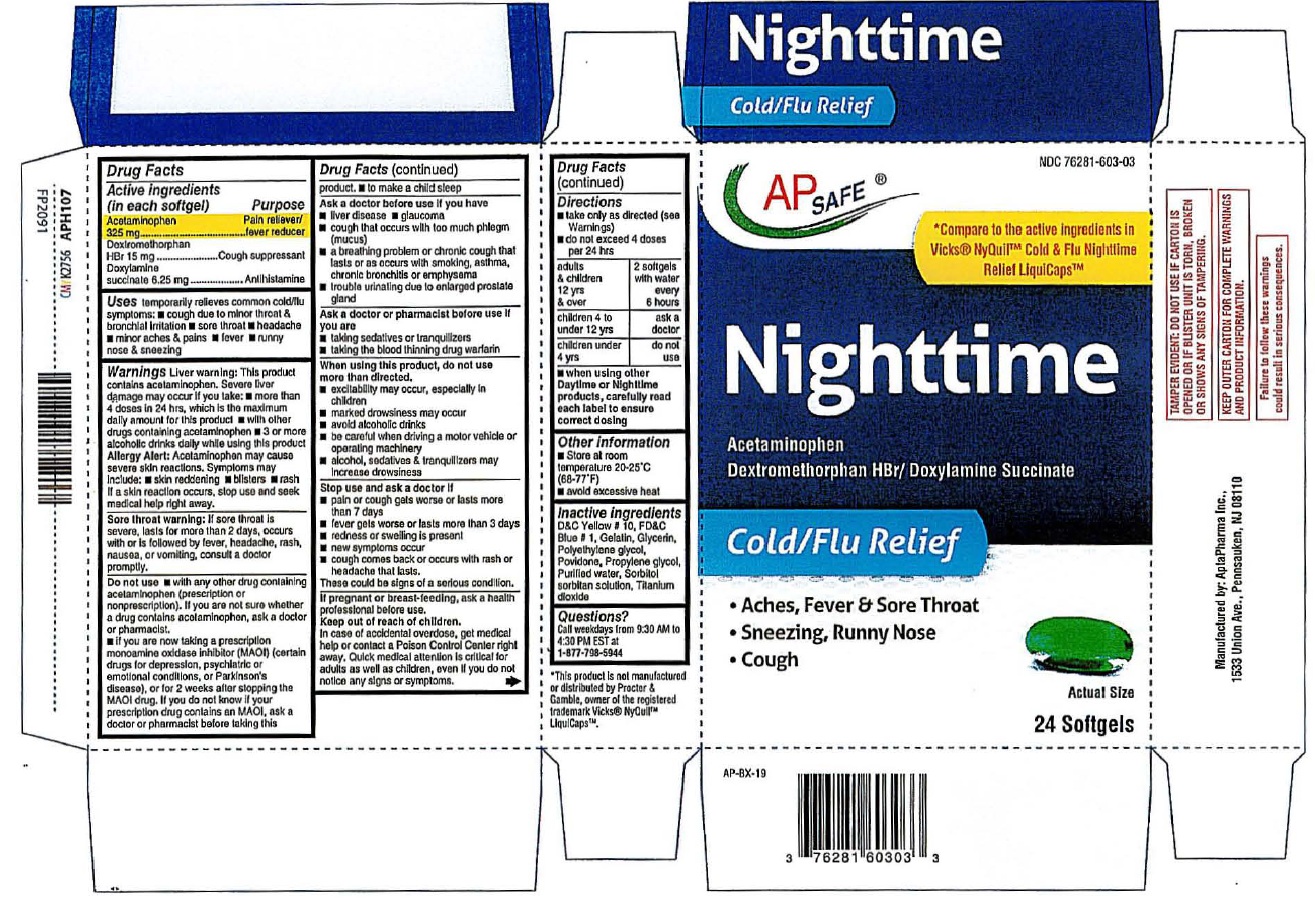
Product Label: AP SAFE Night time cold/flu Softgels
------------------------------------------------------
AP SAFE® Nighttime *Compare to the active ingredients
Cold/Flu Relief In Vicks® Nyquil™ Cold & Flu Nighttime
Relief LiquiCaps™
Acetaminophen
Dextromethorphan HBr/Doxylamine Succinate
24 Softgels
Manufactured by AptaPharma Inc.,
1533 Union Ave., Pennsauken, NJ 08110
This product is not manufactured or distributed by Procter & Gamble, owner of the registered
trademark Vicks® Nyquil™ Liquicaps™
TAMPER EVIDENT:DO NOT USE IF CARTON IS OPENED OR IF BLISTER UNIT IS TORN,
BROKEN OR SHOWS ANY SIGNS OF TAMPERINGKEEP OUTER CARTON FOR COMPLETE WARNING AND PRODUCT INFORMATION.
resRes
-
INGREDIENTS AND APPEARANCE
NIGHT TIME COLD AND FLU RELIEF LIQUID FILLED SOFTGELS
acetaminophen,dextromethrophan hbr,doxylamine succinate capsule, liquid filledProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 76281-603 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 325 mg DEXTROMETHORPHAN HYDROBROMIDE (UNII: 9D2RTI9KYH) (DEXTROMETHORPHAN - UNII:7355X3ROTS) DEXTROMETHORPHAN HYDROBROMIDE 15 mg DOXYLAMINE SUCCINATE (UNII: V9BI9B5YI2) (DOXYLAMINE - UNII:95QB77JKPL) DOXYLAMINE SUCCINATE 6.25 mg Inactive Ingredients Ingredient Name Strength D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) GELATIN (UNII: 2G86QN327L) GLYCERIN (UNII: PDC6A3C0OX) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POVIDONE (UNII: FZ989GH94E) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) SORBITOL (UNII: 506T60A25R) SORBITAN (UNII: 6O92ICV9RU) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color green Score no score Shape OVAL Size 20mm Flavor Imprint Code 603 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 76281-603-03 2 in 1 CARTON 01/01/2026 1 12 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M012 01/01/2026 Labeler - AptaPharma Inc. (790523323) Registrant - AptaPharma Inc. (790523323) Establishment Name Address ID/FEI Business Operations AptaPharma Inc. 790523323 manufacture(76281-603)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.