AVANCE NERVE GRAFT- processed nerve allograft implant
Drug Labeling and Warnings
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use AVANCE® safely and effectively. See full prescribing information for AVANCE.
AVANCE (acellular nerve allograft-arwx), for surgical implantation.
Initial U.S. Approval: 2025INDICATIONS AND USAGE
AVANCE is an acellular nerve scaffold indicated for the treatment of adult and pediatric patients aged one month and older with:
- Sensory nerve discontinuity (≤25 mm). (1)
- Sensory nerve discontinuity (>25 mm). This indication is approved under accelerated approval based on static two-point discrimination (s2PD) at 12 months in sensory nerve gaps ≤25 mm, which provided empirical evidence to reasonably predict clinical benefit given similarities in pathophysiology and anticipated therapeutic effects. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory clinical trial. (1, 14)
- Mixed and motor nerve discontinuity. This indication is approved under accelerated approval based on s2PD at 12 months in sensory nerve gaps, which provided empirical evidence to reasonably predict clinical benefit given similarities in pathophysiology and anticipated therapeutic effects. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory clinical trial. (1, 14)
DOSAGE AND ADMINISTRATION
- AVANCE is intended for surgical implantation. (2.1)
- The recommended number of AVANCE required is based on the nerve deficits identified for repair. (2.1)
- AVANCE may be trimmed to accommodate the size of the deficits. Each AVANCE is for implantation in a single patient only. (2.1)
- See full prescribing information for AVANCE preparation and administration instructions. (2.2) (2.3)
DOSAGE FORMS AND STRENGTHS
AVANCE is available in 16 combination sizes with the following dimensions:
- Lengths: 15 mm, 30 mm, 50 mm, and 70 mm
- Diameters: 1 to 2 mm, 2 to 3 mm, 3 to 4 mm, and 4 to 5 mm
AVANCE may vary in color from white, off-white, pink, pale pink, and yellow to pale yellow. (3)
CONTRAINDICATIONS
None. (4)
WARNINGS AND PRECAUTIONS
- Procedural Complications – Monitor for procedural complications, including pain, hyperesthesia, infection, implant site swelling, adhesions, hypertrophic scar formation, impaired motor or sensory function, bleeding, and neuroma formation, and manage accordingly. (5.1)
- Transmission of Infectious Diseases – Because AVANCE is made from human donor tissue, it may carry a risk of transmitting infectious agents, e.g., viruses, the variant Creutzfeldt-Jakob disease (vCJD) agent, and theoretically, the Creutzfeldt-Jakob disease (CJD) agent. No cases of transmission of viral diseases, vCJD, or CJD have ever been identified for AVANCE. All infections thought to be transmitted by AVANCE should be reported to Axogen Corporation at 1-888-296-4361. (5.2)
ADVERSE REACTIONS
The most common adverse reactions (incidence ≥2%) were procedural pain (4%) and hyperesthesia (3%). (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Customer Care at 1-888-296-4361 or customercare@axogeninc.com or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 1/2026
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
1.1 Sensory Nerve Discontinuity (≤25 mm)
1.2 Sensory Nerve Discontinuity (>25 mm)
1.3 Mixed and Motor Nerve Discontinuity
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dose
2.2 Preparation
2.3 Administration
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Procedural Complications
5.2 Transmission of Infectious Diseases
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
16.2 Storage and Handling
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
1.1 Sensory Nerve Discontinuity (≤25 mm)
AVANCE is indicated for the treatment of adult and pediatric patients aged one month and older with sensory nerve discontinuity ≤25 mm.
1.2 Sensory Nerve Discontinuity (>25 mm)
AVANCE is indicated for the treatment of adult and pediatric patients aged one month and older with peripheral sensory nerve discontinuity >25 mm.
This indication is approved under accelerated approval based on improvement in static two-point discrimination (s2PD) at 12 months in sensory nerve gaps ≤ 25 mm, which provided empirical evidence to reasonably predict clinical benefit given similarities in pathophysiology and anticipated therapeutic effects. [see Clinical Studies (14)]. Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory clinical trials.
1.3 Mixed and Motor Nerve Discontinuity
AVANCE is indicated for the treatment of adult and pediatric patients aged one month and older with mixed and motor nerve discontinuity.
This indication is approved under accelerated approval based on improvement in s2PD at 12 months in sensory nerve gaps, which provided empirical evidence to reasonably predict clinical benefit given similarities in pathophysiology and anticipated therapeutic effects. [see Clinical Studies (14)]. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory clinical trial.
-
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dose
AVANCE is intended for surgical implantation.
- The recommended number of AVANCE required is based on the nerve deficits identified for repair.
- AVANCE may be trimmed to accommodate the size of the deficits. Each AVANCE is intended for implantation in a single patient only.
- AVANCE is available in 16 size combinations of different lengths and diameters to fit the size and diameter of the nerve(s) to be treated. Healthcare professionals should carefully assess the length and diameter of the nerve deficit(s) to be treated to determine the most suitable quantity and sizes of AVANCE required for treatment. [see Dosage and Administration (2.3)].
Refer to Table 1 for the available sizes of AVANCE:
Table 1. Available Sizes of AVANCE Length (mm) Diameter Range (mm)* Length Range (mm)* - * AVANCE is available in the lengths and diameters as shown in Table 1. The graft diameter is measured at each end of the graft, and variation along the length can occur in human tissue.
15
1 to 2
15 to 21
15
2 to 3
15 to 21
15
3 to 4
15 to 21
15
4 to 5
15 to 21
30
1 to 2
30 to 36
30
2 to 3
30 to 36
30
3 to 4
30 to 36
30
4 to 5
30 to 36
50
1 to 2
50 to 56
50
2 to 3
50 to 56
50
3 to 4
50 to 56
50
4 to 5
50 to 56
70
1 to 2
70 to 76
70
2 to 3
70 to 76
70
3 to 4
70 to 76
70
4 to 5
70 to 76
2.2 Preparation
Receipt and Storage of AVANCE:
AVANCE is shipped on dry ice in validated insulated shipping containers. AVANCE must remain frozen at ≤-40°C (≤-40°F) and be kept frozen until use. For a complete description of the AVANCE packaging configuration and storage conditions, [see How Supplied/Storage and Handling (16)].
AVANCE is to be prepared in an appropriate surgical environment.
Supplies:
The following supplies are not included but are needed for preparation:
- Room temperature sterile normal saline or sterile Lactated Ringer’s solution (LRS).
Preparation Instructions:
Following standard surgical site preparation for exposure, mobilization, and trimming of the peripheral nerve to be treated, determine the size and/or number of AVANCE that need to be thawed. [see Dosage and Administration (2.3)].
The following instructions are for preparing one AVANCE. Two team members (one sterile field team member and one team member outside the sterile field) are required.
If two or more AVANCE are required, prepare them individually. AVANCE thawing should take approximately 6 minutes.
- 1. The team member outside the sterile field removes the desired AVANCE from the freezer and transports it to the operating room.
- 2. The team member outside the sterile field removes the zip top bag and opens and removes the carton.
- 3. The team member outside the sterile field peels open the seal of the outer foil-poly pouch and aseptically presents the inner Tyvek®-poly pouch to the sterile field team member for placement in the sterile field. The sterile field team member aseptically removes the inner Tyvek-poly pouch presented by the team member outside the sterile field from the inside of the outer foil-poly pouch and places it in the sterile field.
- 4. In the sterile field, the sterile field team member peels open the inner Tyvek-poly pouch and removes the clamshell tray.
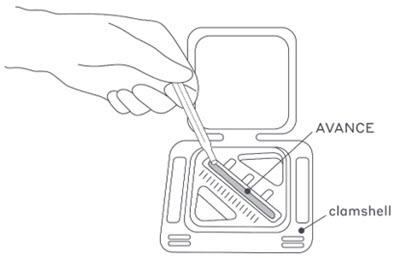
- 5. The sterile field team member opens the clamshell tray and fills the pre-molded thawing reservoir with room temperature sterile normal saline or sterile LRS (Figure 1). Do not heat AVANCE or add heated saline or LRS to the graft.
Figure 1: Thawing AVANCE with sterile saline or LRS.
Figure 1: Thawing AVANCE with sterile saline or LRS.
- 6. Do not implant a partially or fully frozen product. Allow AVANCE to thaw completely before use, which will take approximately 6 minutes. AVANCE must be thawed until it reaches room temperature without heating. Once thawed, AVANCE must be either implanted or discarded within 12 hours.
2.3 Administration
AVANCE is implanted in appropriate sterile conditions by a trained licensed physician. Follow the steps below for surgical implantation of AVANCE:
- 1. Prior to implanting AVANCE, it is important to trim back the nerve to be repaired until healthy nerve tissue is reached. Healthy nerve tissue can be identified by pouching fascicles and punctate bleeding.
- 2. After trimming, if applicable, place the extremity in extension and measure the deficit as well as the diameter of the nerve to be repaired.
- 3. Select an AVANCE of an appropriate length that ensures there will be no tension on the graft when implanted and of a diameter that most closely approximates the nerve to be repaired. AVANCE may be trimmed to accommodate the size of the deficits.
- 4. When handling AVANCE, only hold it by the outermost epineurium and avoid crimping or crushing the graft.
- 5. Coaptation can be achieved by multiple methods. Surgical methods should be tailored to the type of injury and goals of reconstruction, as well as body region, nerve type, and size of discontinuity. One method is to use appropriately sized non-absorbable sutures such as 8-‑0 or 9-0, which should be placed only through the epineurium of the nerve. Either end of AVANCE can be coapted to the proximal nerve to be repaired.
- 6. To bridge the defect, secure the sutures, ensuring that they are not overtightened. The nerve being repaired and AVANCE should be lightly touching, but they should not overlap.
-
3 DOSAGE FORMS AND STRENGTHS
AVANCE will vary in color from white, off-white, pink, pale pink, and yellow to pale yellow. AVANCE is a sterile, surgical implant and is intended for administration to a single patient. The available sizes of AVANCE are provided in Table 1.
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Procedural Complications
Procedural complications have occurred with the AVANCE implantation procedure and have included pain, hyperesthesia, infection, implant site swelling, adhesions, hypertrophic scar formation, impaired motor or sensory function, bleeding, and neuroma formation [see Adverse Reactions (6)]. Additional procedural complications may include risk of bleeding and complications related to anesthesia. Monitor patients for procedural complications with AVANCE implantation and manage accordingly.
5.2 Transmission of Infectious Diseases
Transmission of infectious diseases or agents may occur with AVANCE administration, as it is manufactured using tissues from human cadaveric donors. Donors are screened and tested for Human Immune-deficiency Virus 1 (HIV-1); Human Immune-deficiency Virus 2 (HIV-2); Hepatitis B Virus (HBV); Hepatitis C Virus (HCV); and Syphilis (Treponema pallidum). Additional testing for Human T-cell Leukemia-lymphoma Virus 1 (HTLV-1) and Human T-cell Leukemia-lymphoma Virus 2 (HTLV-2) may be performed as required by local regulatory authorities in US states. Only screening was performed for Creutzfeldt-Jakob disease (CJD). These measures do not eliminate the risk of transmitting these or other infectious diseases or agents.
All infections thought to be transmitted by AVANCE should be reported to Axogen Corporation at 1‑888-296-4361.
-
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in one clinical trial cannot be directly compared to rates in other clinical trials and may not reflect the rates observed in practice.
The safety database described in this section reflects exposure to AVANCE in the RECON Study. A total of 112 patients received AVANCE and 108 patients received NeuraGen® (bovine nerve cuff) and were followed for a duration of 12 months. [see Clinical Studies (14)].
A serious adverse reaction occurred in 1 patient, which was wound dehiscence.
Table 2 lists the most common adverse reactions that occurred in ≥ 2% of patients in the RECON Study.
Table 2. Adverse Reactions occurring in > 2% of Patients in the RECON Study Adverse Reactions
AVANCE
n = 112n (%)
Bovine Nerve Cuff
n = 108n (%)
Implant site hyperesthesia
3 (3)
5 (5)
Procedural pain
4 (4)
0
Other clinically significant adverse reactions that occurred in <2% of patients in AVANCE group include: implant site swelling (n=2), paraesthesia (n=2), hypertrophic scar (n=2), pyogenic granuloma (n=2), grade 3 neuroma (n=1), wound dehiscence (n=1), tendon adhesion (n=1), implant site nodule (n=1), osteomyelitis (n=1), and dermal cyst (n=1).
6.2 Postmarketing Experience
Adverse Reactions from Observational Studies
The safety of AVANCE was evaluated in an ongoing, multicenter, observational registry study in patients undergoing nerve repair. The registry assessed repairs using the Medical Research Council Classification (MRCC) across nerve types (sensory, motor, and mixed), gap lengths (5–210 mm), and body regions (upper extremity, head and neck, torso including breast, and lower extremity).
Adverse reactions reported in the study include neuroma, seromas, implant site infection, tissue necrosis, nerve regeneration failure, hyperesthesia, hypoesthesia, AVANCE implantation error, and hematoma.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no data on the use of AVANCE in pregnant women. No animal reproductive and developmental toxicity studies have been conducted with AVANCE to assess whether it can cause harm to the mother or fetus when administered to a pregnant woman.
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
8.4 Pediatric Use
The safety and effectiveness of AVANCE have been established in pediatric patients aged one month and older. The use of AVANCE in pediatric patients for the treatment of nerve discontinuity was supported by extrapolation of adult data from the RECON Study. [see Clinical Studies (14)].
-
11 DESCRIPTION
AVANCE (acellular nerve allograft-arwx) is a sterile, acellular nerve scaffold derived from human cadaveric peripheral nerve tissue for surgical implantation. The processing method maintains the three-dimensional scaffold of the native peripheral nerve, including the endoneurial tubes, perineurium, epineurium, and microvasculature of the extracellular matrix (ECM). The processing removes cellular and noncellular components, including cells, fat, blood, axonal debris, and glycosaminoglycans. AVANCE is saturated in Lactated Ringer’s Solution prior to packaging and storage.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
AVANCE is an acellular nerve scaffold intended to restore nerve function in the distal tissue.
AVANCE provides structural and biochemical cues for axonal regeneration. The structural cue is provided by the intact endoneurial tubes. The biochemical cue is provided by the basement membrane proteins of the endoneurial tubes. The basement membrane of the endoneurial tubes includes many proteins, such as collagen, laminin, fibronectin, and proteoglycans. The laminin lining the endoneurial tubes of AVANCE has been shown in in vitro assays and animal studies to be bioactive by supporting Schwann cell migration, axon growth cone interactions, and neurite extension. Without the specific bioactive laminin composition, the neuroregenerative capacity is hindered. However, the exact mechanism of action is unknown.
- 13 NONCLINICAL TOXICOLOGY
-
14 CLINICAL STUDIES
The efficacy of AVANCE was evaluated in a multicenter, prospective, randomized, patient- and evaluator-blinded study that compared implantation of AVANCE to implantation of NeuraGen nerve cuff (RECON; NCT01809002). The study enrolled adult patients with sustained injury to at least one sensory nerve, distal to the superficial palmar arch and proximal to the distal interphalangeal joint, and a nerve gap between 5 mm and 25 mm. Patients with nerve injuries longer than 24 weeks, injuries requiring replantation of the target digit, injuries with vascular damage resulting in inadequate perfusion despite repair, prior history of type 1 Diabetes Mellitus or type 2 Diabetes Mellitus requiring regular insulin therapy, and prior history of neuropathy were excluded.
A total of 220 patients were intraoperatively randomized in a 1:1 ratio to receive either AVANCE (n=112) or NeuraGen (n=108). Randomization was stratified based on nerve gap length (5–14 mm: Group A; 15–25 mm: Group B).
The population demographics were as follows: the median age was 37 years (range 18 to 69 years), and 155 patients (71%) were male. 174 patients (79%) were White, 33 patients (15%) were Black, 3 patients (1.4%) were Hispanic or Latino, 2 patients (0.9%) were Asian, 2 patients (0.9%) were Native Hawaiian or Pacific Islander, and 6 patients (2.7%) were of “other race”.
The primary efficacy outcome measure was improvement in static two-point discrimination (s2PD) in the target digit, with a non-inferiority test (non-inferiority margin of -2 mm) between the groups at 12 months following reconstruction.
The efficacy results are summarized in Table 3 below.
Table 3: Efficacy Results of RECON Study a Estimated using repeated measures Analysis of Covariance (ANCOVA) model with fixed effects for repair type, visit, and gap length and a random effect for subject. Visits Pre-Op screening, Months 1, 3, 6, 9, and 12 were included.
CI=confidence interval; LS= least squares; mm=millimeterEndpoints
AVANCE
(n = 112)Nerve Cuff
(n = 108)LS mean difference
Static two-point discrimination (s2PD) in the target digit at 12 monthsa
LS mean
(95% CI)
9.1 mm
(8.11, 10.04)
9.4 mm
(8.50, 10.30)
0.3 mm
(-0.98, 1.64)
-
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
AVANCE is shipped on dry ice and supplied frozen, as a single, intact graft of varying color from white, off-white, pink, pale pink, and yellow to pale yellow. Do not use if broken or otherwise compromised.
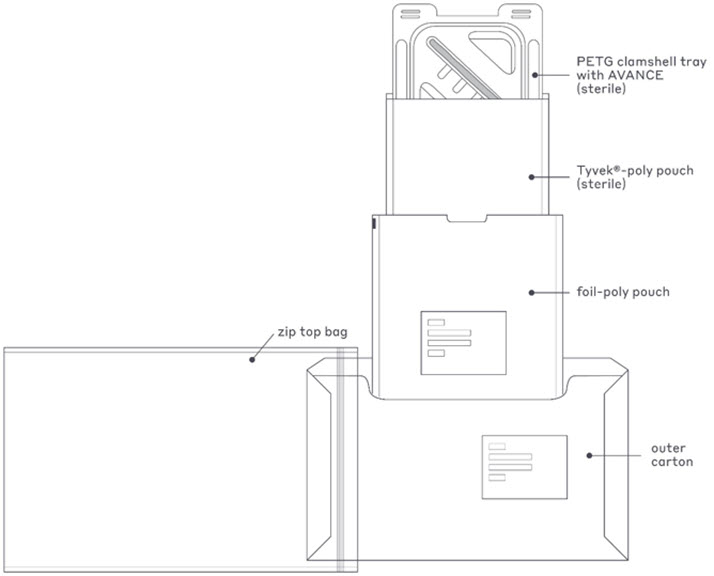
AVANCE is sterile packaged. The immediate container closure system consists of a clamshell (thermoformed tray) within two pouches, a sealed Tyvek-poly pouch within a foil-poly pouch. The foil-poly pouch is the sterile barrier for AVANCE. The immediate container closure system is packaged in a commercial carton, which is sealed with a Carton Seal Tape and placed in a zip top bag for freezer storage protection, see Figure 2.
- AVANCE is available in the following lengths and diameters (Table 4):
Table 4: Commercially Available Sizes of AVANCE Length (mm) Diameter (mm) NDC Numbers 15
1 to 2
84545-115-01
15
2 to 3
84545-215-01
15
3 to 4
84545-315-01
15
4 to 5
84545-415-01
30
1 to 2
84545-130-01
30
2 to 3
84545-230-01
30
3 to 4
84545-330-01
30
4 to 5
84545-430-01
50
1 to 2
84545-150-01
50
2 to 3
84545-250-01
50
3 to 4
84545-350-01
50
4 to 5
84545-450-01
70
1 to 2
84545-170-01
70
2 to 3
84545-270-01
70
3 to 4
84545-370-01
70
4 to 5
84545-470-01
16.2 Storage and Handling
- Ensure the shipping container was received by the date indicated on the shipping container label and that sufficient dry ice is present within the shipping container. The amount of dry ice present within the shipping container can be assessed by the overall shipping container weight. The overall shipping container weight should be at least 12.0 lbs. for the 48-hour shipping container, 15.0 lbs. for the 72-hour shipping container, and 21.0 lbs. for the 120-hour shipping container.
- Store AVANCE at ≤-40°C (≤-40°F) and keep frozen until use.
- Do not use AVANCE if any component of the immediate container closure system or commercial carton has been compromised.
- Thaw AVANCE to room temperature without heating using room temperature sterile normal saline or Lactated Ringer’s Solution (LRS). Do not implant a partially or fully frozen product. Once thawed, it should not be placed back into the freezer. Thawed AVANCE must be implanted or discarded within 12 hours.
- Do not use if the lot number on the outer foil-poly pouch does not match the lot number on the outer carton.
- Any expired or thawed AVANCE that is not used in a surgical procedure should be destroyed in accordance with local, state, and federal or country regulations for disposal of human tissue.
Contact Axogen Customer Care at 888-296-4361 for any issues with the condition of the packaging or graft, or for further instructions.
-
17 PATIENT COUNSELING INFORMATION
Discuss the following with the patient and/or caregivers.
-
Procedural Complications
Inform patients that procedure-related complications such as infection, pain, swelling, bleeding, scarring, neuroma formation, or delayed wound healing may occur after nerve repair surgery using AVANCE. Advise that careful postoperative monitoring is important. [see Warnings and Precautions (5.1) and Adverse Reactions (6)]. -
Transmission of Infectious Diseases
Inform patients that AVANCE is made from cadaveric human tissue and, despite donor screening and validated processing steps, the risk of transmitting infectious diseases cannot be completely eliminated. [see Warnings and Precautions (5.2)]. - Advise the patient to closely follow the physician-prescribed rehabilitation program.
-
Procedural Complications
-
Axogen Corporation
13631 Progress Blvd., Suite 400
Alachua, FL 32615
www.axogeninc.com
U.S. License #2340AVANCE® is a registered trademark of Axogen Corporation.
NeuraGen® is a registered trademark of Integra LifeSciences Corporation.
Tyvek® is a registered trademark of E.I. DuPont De Nemours & Co.
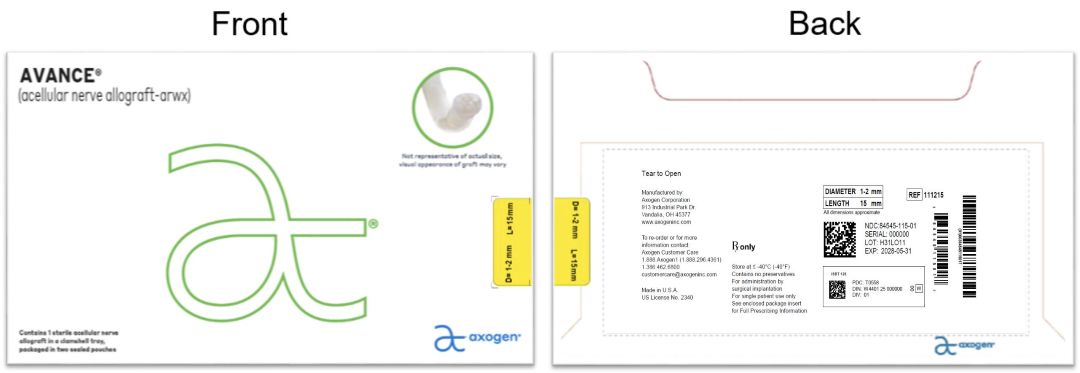
© 2025 Axogen Corporation - PRINCIPAL DISPLAY PANEL - 1-2 mm x 15 mm NDC: 84545-115-01
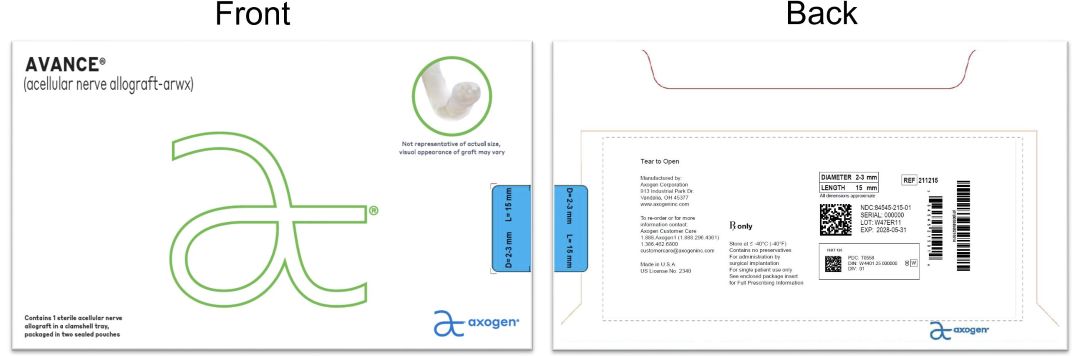
- PRINCIPAL DISPLAY PANEL - 2-3 mm x 15 mm NDC: 84545-215-01
- PRINCIPAL DISPLAY PANEL - 3-4 mm x 15 mm NDC: 84545-315-01
- PRINCIPAL DISPLAY PANEL - 4-5 mm x 15 mm NDC: 84545-415-01
- PRINCIPAL DISPLAY PANEL - 1-2 mm x 30 mm NDC: 84545-130-01
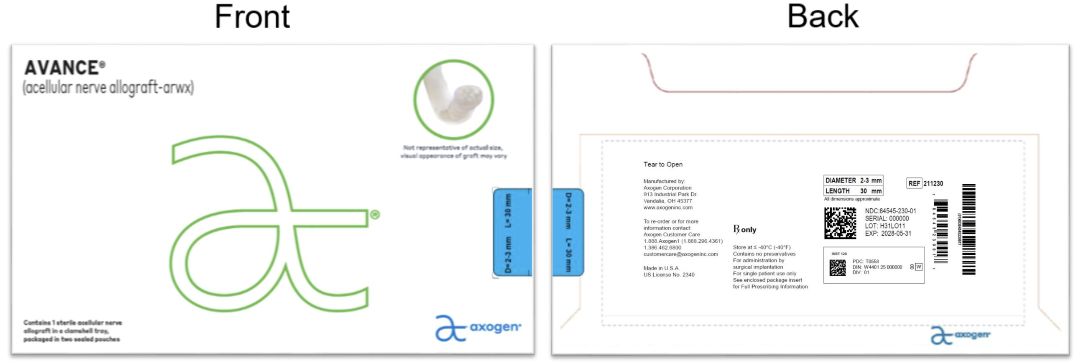
- PRINCIPAL DISPLAY PANEL - 2-3 mm x 30 mm NDC: 84545-230-01
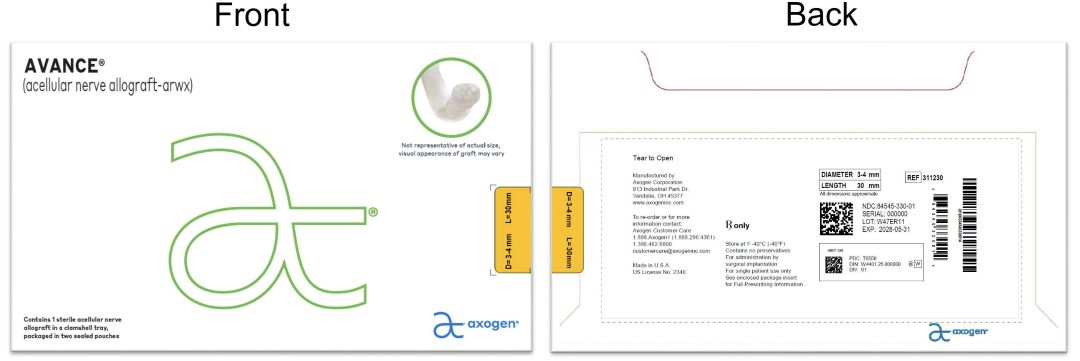
- PRINCIPAL DISPLAY PANEL - 3-4 mm x 30 mm NDC: 84545-330-01
- PRINCIPAL DISPLAY PANEL - 4-5 mm x 30 mm NDC: 84545-430-01
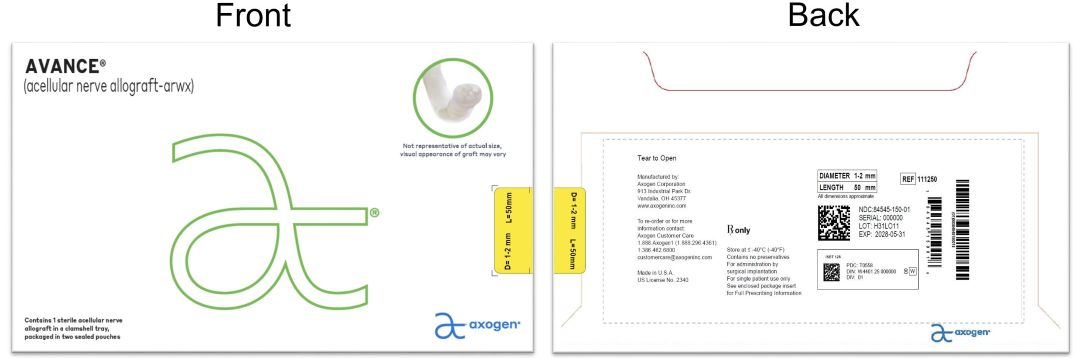
- PRINCIPAL DISPLAY PANEL - 1-2 mm x 50 mm NDC: 84545-150-01
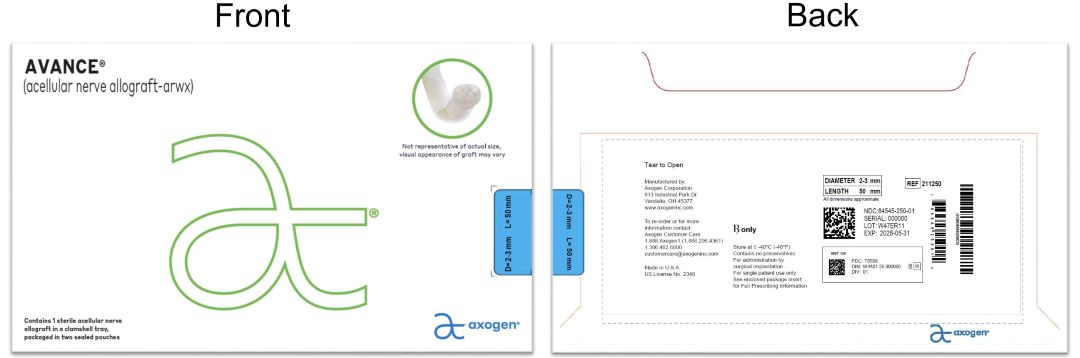
- PRINCIPAL DISPLAY PANEL - 2-3 mm x 50 mm NDC: 84545-250-01
- PRINCIPAL DISPLAY PANEL - 3-4 mm x 50 mm NDC: 84545-350-01
- PRINCIPAL DISPLAY PANEL - 4-5 mm x 50 mm NDC: 84545-450-01
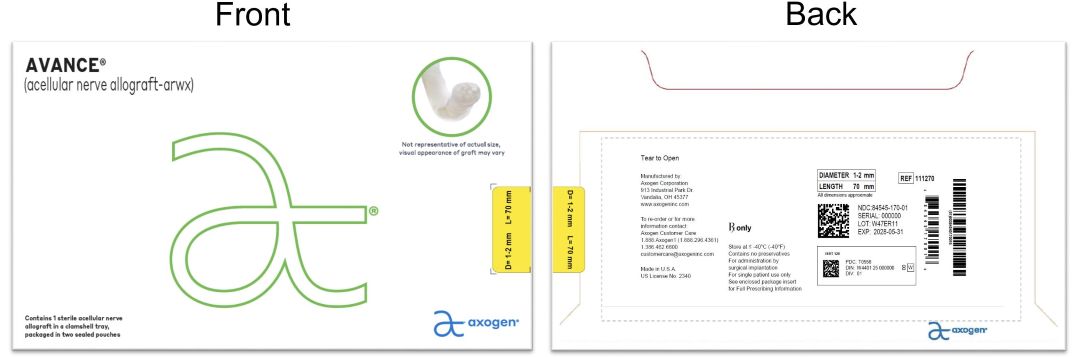
- PRINCIPAL DISPLAY PANEL.-. 1-2 mm x 70 mm NDC: 84545-170-01
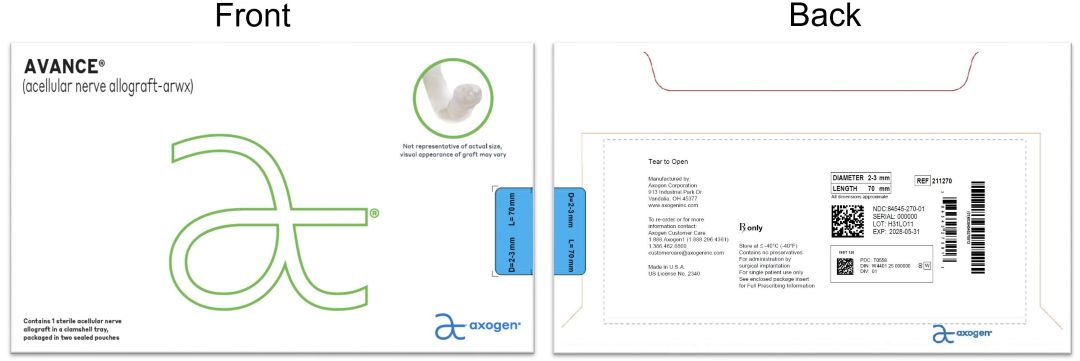
- PRINCIPAL DISPLAY PANEL - 2-3 mm x 70 mm NDC: 84545-270-01
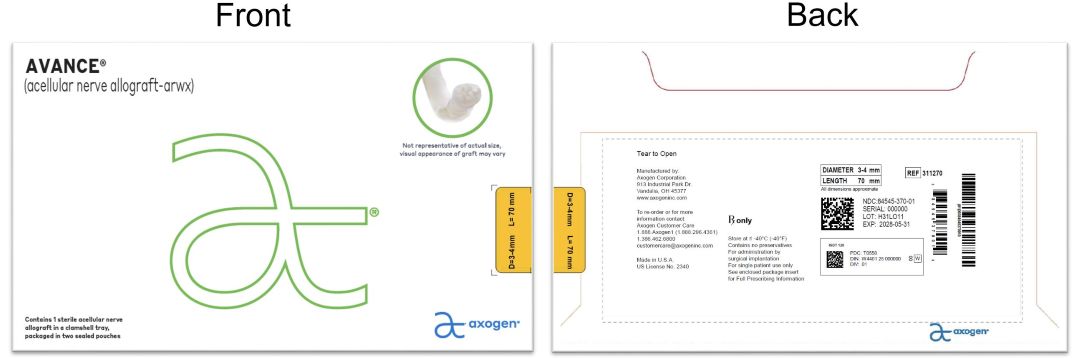
- PRINCIPAL DISPLAY PANEL - 3-4 mm x 70 mm NDC: 84545-370-01
- PRINCIPAL DISPLAY PANEL - 4-5 mm x 70 mm NDC: 84545-470-01
-
INGREDIENTS AND APPEARANCE
AVANCE NERVE GRAFT
processed nerve allograft implantProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 84545-115 Route of Administration SOFT TISSUE Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HUMAN PERIPHERAL NERVE TISSUE (UNII: 725P2LR8RG) (HUMAN PERIPHERAL NERVE TISSUE - UNII:725P2LR8RG) HUMAN PERIPHERAL NERVE TISSUE 1 mm in 15 mm Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM LACTATE (UNII: TU7HW0W0QT) POTASSIUM CHLORIDE (UNII: 660YQ98I10) CALCIUM CHLORIDE (UNII: M4I0D6VV5M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 84545-115-01 1 in 1 BAG 04/06/2026 1 1 in 1 CARTON 1 1 in 1 POUCH 1 1 in 1 POUCH 1 1 mm in 1 CONTAINER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125816 04/06/2026 AVANCE NERVE GRAFT
processed nerve allograft implantProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 84545-215 Route of Administration SOFT TISSUE Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HUMAN PERIPHERAL NERVE TISSUE (UNII: 725P2LR8RG) (HUMAN PERIPHERAL NERVE TISSUE - UNII:725P2LR8RG) HUMAN PERIPHERAL NERVE TISSUE 2 mm in 15 mm Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM LACTATE (UNII: TU7HW0W0QT) POTASSIUM CHLORIDE (UNII: 660YQ98I10) CALCIUM CHLORIDE (UNII: M4I0D6VV5M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 84545-215-01 1 in 1 BAG 04/06/2026 1 1 in 1 CARTON 1 1 in 1 POUCH 1 1 in 1 POUCH 1 1 mm in 1 CONTAINER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125816 04/06/2026 AVANCE NERVE GRAFT
processed nerve allograft implantProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 84545-315 Route of Administration SOFT TISSUE Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HUMAN PERIPHERAL NERVE TISSUE (UNII: 725P2LR8RG) (HUMAN PERIPHERAL NERVE TISSUE - UNII:725P2LR8RG) HUMAN PERIPHERAL NERVE TISSUE 3 mm in 15 mm Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM LACTATE (UNII: TU7HW0W0QT) POTASSIUM CHLORIDE (UNII: 660YQ98I10) CALCIUM CHLORIDE (UNII: M4I0D6VV5M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 84545-315-01 1 in 1 BAG 04/06/2026 1 1 in 1 CARTON 1 1 in 1 POUCH 1 1 in 1 POUCH 1 1 mm in 1 CONTAINER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125816 04/06/2026 AVANCE NERVE GRAFT
processed nerve allograft implantProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 84545-415 Route of Administration SOFT TISSUE Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HUMAN PERIPHERAL NERVE TISSUE (UNII: 725P2LR8RG) (HUMAN PERIPHERAL NERVE TISSUE - UNII:725P2LR8RG) HUMAN PERIPHERAL NERVE TISSUE 4 mm in 15 mm Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM LACTATE (UNII: TU7HW0W0QT) POTASSIUM CHLORIDE (UNII: 660YQ98I10) CALCIUM CHLORIDE (UNII: M4I0D6VV5M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 84545-415-01 1 in 1 BAG 04/06/2026 1 1 in 1 CARTON 1 1 in 1 POUCH 1 1 in 1 POUCH 1 1 mm in 1 CONTAINER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125816 04/06/2026 AVANCE NERVE GRAFT
processed nerve allograft implantProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 84545-130 Route of Administration SOFT TISSUE Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HUMAN PERIPHERAL NERVE TISSUE (UNII: 725P2LR8RG) (HUMAN PERIPHERAL NERVE TISSUE - UNII:725P2LR8RG) HUMAN PERIPHERAL NERVE TISSUE 1 mm in 30 mm Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM LACTATE (UNII: TU7HW0W0QT) POTASSIUM CHLORIDE (UNII: 660YQ98I10) CALCIUM CHLORIDE (UNII: M4I0D6VV5M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 84545-130-01 1 in 1 BAG 04/06/2026 1 1 in 1 CARTON 1 1 in 1 POUCH 1 1 in 1 POUCH 1 1 mm in 1 CONTAINER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125816 04/06/2026 AVANCE NERVE GRAFT
processed nerve allograft implantProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 84545-230 Route of Administration SOFT TISSUE Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HUMAN PERIPHERAL NERVE TISSUE (UNII: 725P2LR8RG) (HUMAN PERIPHERAL NERVE TISSUE - UNII:725P2LR8RG) HUMAN PERIPHERAL NERVE TISSUE 2 mm in 30 mm Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM LACTATE (UNII: TU7HW0W0QT) POTASSIUM CHLORIDE (UNII: 660YQ98I10) CALCIUM CHLORIDE (UNII: M4I0D6VV5M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 84545-230-01 1 in 1 BAG 04/06/2026 1 1 in 1 CARTON 1 1 in 1 POUCH 1 1 in 1 POUCH 1 1 mm in 1 CONTAINER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125816 04/06/2026 AVANCE NERVE GRAFT
processed nerve allograft implantProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 84545-330 Route of Administration SOFT TISSUE Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HUMAN PERIPHERAL NERVE TISSUE (UNII: 725P2LR8RG) (HUMAN PERIPHERAL NERVE TISSUE - UNII:725P2LR8RG) HUMAN PERIPHERAL NERVE TISSUE 3 mm in 30 mm Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM LACTATE (UNII: TU7HW0W0QT) POTASSIUM CHLORIDE (UNII: 660YQ98I10) CALCIUM CHLORIDE (UNII: M4I0D6VV5M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 84545-330-01 1 in 1 BAG 04/06/2026 1 1 in 1 CARTON 1 1 in 1 POUCH 1 1 in 1 POUCH 1 1 mm in 1 CONTAINER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125816 04/06/2026 AVANCE NERVE GRAFT
processed nerve allograft implantProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 84545-430 Route of Administration SOFT TISSUE Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HUMAN PERIPHERAL NERVE TISSUE (UNII: 725P2LR8RG) (HUMAN PERIPHERAL NERVE TISSUE - UNII:725P2LR8RG) HUMAN PERIPHERAL NERVE TISSUE 4 mm in 30 mm Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM LACTATE (UNII: TU7HW0W0QT) POTASSIUM CHLORIDE (UNII: 660YQ98I10) CALCIUM CHLORIDE (UNII: M4I0D6VV5M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 84545-430-01 1 in 1 BAG 04/06/2026 1 1 in 1 CARTON 1 1 in 1 POUCH 1 1 in 1 POUCH 1 1 mm in 1 CONTAINER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125816 04/06/2026 AVANCE NERVE GRAFT
processed nerve allograft implantProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 84545-150 Route of Administration SOFT TISSUE Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HUMAN PERIPHERAL NERVE TISSUE (UNII: 725P2LR8RG) (HUMAN PERIPHERAL NERVE TISSUE - UNII:725P2LR8RG) HUMAN PERIPHERAL NERVE TISSUE 1 mm in 50 mm Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM LACTATE (UNII: TU7HW0W0QT) POTASSIUM CHLORIDE (UNII: 660YQ98I10) CALCIUM CHLORIDE (UNII: M4I0D6VV5M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 84545-150-01 1 in 1 BAG 04/06/2026 1 1 in 1 CARTON 1 1 in 1 POUCH 1 1 in 1 POUCH 1 1 mm in 1 CONTAINER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125816 04/06/2026 AVANCE NERVE GRAFT
processed nerve allograft implantProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 84545-250 Route of Administration SOFT TISSUE Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HUMAN PERIPHERAL NERVE TISSUE (UNII: 725P2LR8RG) (HUMAN PERIPHERAL NERVE TISSUE - UNII:725P2LR8RG) HUMAN PERIPHERAL NERVE TISSUE 2 mm in 50 mm Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM LACTATE (UNII: TU7HW0W0QT) POTASSIUM CHLORIDE (UNII: 660YQ98I10) CALCIUM CHLORIDE (UNII: M4I0D6VV5M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 84545-250-01 1 in 1 BAG 04/06/2026 1 1 in 1 CARTON 1 1 in 1 POUCH 1 1 in 1 POUCH 1 1 mm in 1 CONTAINER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125816 04/06/2026 AVANCE NERVE GRAFT
processed nerve allograft implantProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 84545-350 Route of Administration SOFT TISSUE Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HUMAN PERIPHERAL NERVE TISSUE (UNII: 725P2LR8RG) (HUMAN PERIPHERAL NERVE TISSUE - UNII:725P2LR8RG) HUMAN PERIPHERAL NERVE TISSUE 3 mm in 50 mm Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM LACTATE (UNII: TU7HW0W0QT) POTASSIUM CHLORIDE (UNII: 660YQ98I10) CALCIUM CHLORIDE (UNII: M4I0D6VV5M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 84545-350-01 1 in 1 BAG 04/06/2026 1 1 in 1 CARTON 1 1 in 1 POUCH 1 1 in 1 POUCH 1 1 mm in 1 CONTAINER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125816 04/06/2026 AVANCE NERVE GRAFT
processed nerve allograft implantProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 84545-450 Route of Administration SOFT TISSUE Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HUMAN PERIPHERAL NERVE TISSUE (UNII: 725P2LR8RG) (HUMAN PERIPHERAL NERVE TISSUE - UNII:725P2LR8RG) HUMAN PERIPHERAL NERVE TISSUE 4 mm in 50 mm Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM LACTATE (UNII: TU7HW0W0QT) POTASSIUM CHLORIDE (UNII: 660YQ98I10) CALCIUM CHLORIDE (UNII: M4I0D6VV5M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 84545-450-01 1 in 1 BAG 04/06/2026 1 1 in 1 CARTON 1 1 in 1 POUCH 1 1 in 1 POUCH 1 1 mm in 1 CONTAINER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125816 04/06/2026 AVANCE NERVE GRAFT
processed nerve allograft implantProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 84545-170 Route of Administration SOFT TISSUE Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HUMAN PERIPHERAL NERVE TISSUE (UNII: 725P2LR8RG) (HUMAN PERIPHERAL NERVE TISSUE - UNII:725P2LR8RG) HUMAN PERIPHERAL NERVE TISSUE 1 mm in 70 mm Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM LACTATE (UNII: TU7HW0W0QT) POTASSIUM CHLORIDE (UNII: 660YQ98I10) CALCIUM CHLORIDE (UNII: M4I0D6VV5M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 84545-170-01 1 in 1 BAG 04/06/2026 1 1 in 1 CARTON 1 1 in 1 POUCH 1 1 in 1 POUCH 1 1 mm in 1 CONTAINER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125816 04/06/2026 AVANCE NERVE GRAFT
processed nerve allograft implantProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 84545-270 Route of Administration SOFT TISSUE Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HUMAN PERIPHERAL NERVE TISSUE (UNII: 725P2LR8RG) (HUMAN PERIPHERAL NERVE TISSUE - UNII:725P2LR8RG) HUMAN PERIPHERAL NERVE TISSUE 2 mm in 70 mm Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM LACTATE (UNII: TU7HW0W0QT) POTASSIUM CHLORIDE (UNII: 660YQ98I10) CALCIUM CHLORIDE (UNII: M4I0D6VV5M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 84545-270-01 1 in 1 BAG 04/06/2026 1 1 in 1 CARTON 1 1 in 1 POUCH 1 1 in 1 POUCH 1 1 mm in 1 CONTAINER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125816 04/06/2026 AVANCE NERVE GRAFT
processed nerve allograft implantProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 84545-370 Route of Administration SOFT TISSUE Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HUMAN PERIPHERAL NERVE TISSUE (UNII: 725P2LR8RG) (HUMAN PERIPHERAL NERVE TISSUE - UNII:725P2LR8RG) HUMAN PERIPHERAL NERVE TISSUE 3 mm in 70 mm Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM LACTATE (UNII: TU7HW0W0QT) POTASSIUM CHLORIDE (UNII: 660YQ98I10) CALCIUM CHLORIDE (UNII: M4I0D6VV5M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 84545-370-01 1 in 1 BAG 04/06/2026 1 1 in 1 CARTON 1 1 in 1 POUCH 1 1 in 1 POUCH 1 1 mm in 1 CONTAINER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125816 04/06/2026 AVANCE NERVE GRAFT
processed nerve allograft implantProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 84545-470 Route of Administration SOFT TISSUE Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HUMAN PERIPHERAL NERVE TISSUE (UNII: 725P2LR8RG) (HUMAN PERIPHERAL NERVE TISSUE - UNII:725P2LR8RG) HUMAN PERIPHERAL NERVE TISSUE 4 mm in 70 mm Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM LACTATE (UNII: TU7HW0W0QT) POTASSIUM CHLORIDE (UNII: 660YQ98I10) CALCIUM CHLORIDE (UNII: M4I0D6VV5M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 84545-470-01 1 in 1 BAG 04/06/2026 1 1 in 1 CARTON 1 1 in 1 POUCH 1 1 in 1 POUCH 1 1 mm in 1 CONTAINER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date BLA BLA125816 04/06/2026 Labeler - Axogen Corporation (113424464)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.