iASSIST® Knee System 302.4021
GUDID 00887868558467
Orthosoft Inc
Robotic surgical navigation system| Primary Device ID | 00887868558467 |
| NIH Device Record Key | b2990a29-af7a-4a77-9d4f-2cc466a4b119 |
| Commercial Distribution Status | In Commercial Distribution |
| Brand Name | iASSIST® Knee System |
| Version Model Number | 302.4021 |
| Catalog Number | 302.4021 |
| Company DUNS | 205670032 |
| Company Name | Orthosoft Inc |
| Device Count | 1 |
| DM Exempt | false |
| Pre-market Exempt | false |
| MRI Safety Status | Labeling does not contain MRI Safety Information |
| Human Cell/Tissue Product | false |
| Device Kit | false |
| Device Combination Product | false |
| Single Use | false |
| Lot Batch | true |
| Serial Number | false |
| Manufacturing Date | true |
| Expiration Date | false |
| Donation Id Number | false |
| Contains Natural Rubber Latex | false |
| Labeled No Natural Rubber Latex | false |
| RX Perscription | true |
| OTC Over-The-Counter | false |
Customer Support Contacts
Device Dimensions
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
| Device Size Text, specify | 0 |
Device Identifiers
| Device Issuing Agency | Device ID |
|---|---|
| GS1 | 00887868558467 [Primary] |
FDA Pre-market Approvals/Notifications & deNovo
- Premarket Notification: K213033
FDA Product Code
| OLO | Orthopedic stereotaxic instrument |
Sterilization
| Steralize Prior To Use | true |
| Device Is Sterile | false |
[00887868558467]
Moist Heat or Steam Sterilization
[00887868558467]
Moist Heat or Steam Sterilization
[00887868558467]
Moist Heat or Steam Sterilization
[00887868558467]
Moist Heat or Steam Sterilization
[00887868558467]
Moist Heat or Steam Sterilization
[00887868558467]
Moist Heat or Steam Sterilization
[00887868558467]
Moist Heat or Steam Sterilization
[00887868558467]
Moist Heat or Steam Sterilization
[00887868558467]
Moist Heat or Steam Sterilization
[00887868558467]
Moist Heat or Steam Sterilization
[00887868558467]
Moist Heat or Steam Sterilization
[00887868558467]
Moist Heat or Steam Sterilization
[00887868558467]
Moist Heat or Steam Sterilization
[00887868558467]
Moist Heat or Steam Sterilization
[00887868558467]
Moist Heat or Steam Sterilization
[00887868558467]
Moist Heat or Steam Sterilization
[00887868558467]
Moist Heat or Steam Sterilization
[00887868558467]
Moist Heat or Steam Sterilization
[00887868558467]
Moist Heat or Steam Sterilization
[00887868558467]
Moist Heat or Steam Sterilization
[00887868558467]
Moist Heat or Steam Sterilization
[00887868558467]
Moist Heat or Steam Sterilization
Device Entry Metadata
| Public Version Status | New |
| Device Record Status | Published |
| Public Version Number | 1 |
| Public Version Date | 2022-09-29 |
| Device Publish Date | 2022-09-21 |
On-Brand Devices [iASSIST® Knee System]
| 00887868558474 | 20-8011-900-13 |
| 00887868558467 | 302.4021 |
| 00887868519161 | 20-8011-065-00 |
| 00887868519154 | 20-8011-063-00 |
| 00887868519147 | 20-8000-070-03 |
| 00887868519130 | 20-8000-070-02 |
| 00887868519017 | 20-8011-501-00 |
Trademark Results [iASSIST]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 IASSIST 97595075 not registered Live/Pending |
Triangle Tube Phase III Co. 2022-09-16 |
 IASSIST 90696907 not registered Live/Pending |
Triangle Tube Phase III Co. 2021-05-07 |
 IASSIST 86299323 not registered Dead/Abandoned |
Stuart C. Irby Company 2014-06-03 |
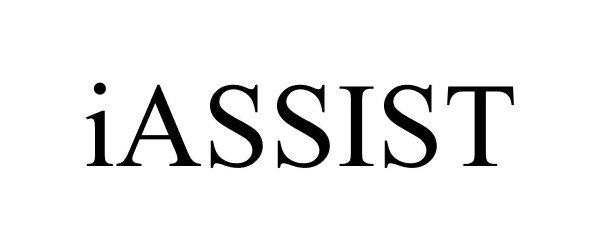 IASSIST 85705606 4351660 Live/Registered |
ZIMMER, INC. 2012-08-16 |
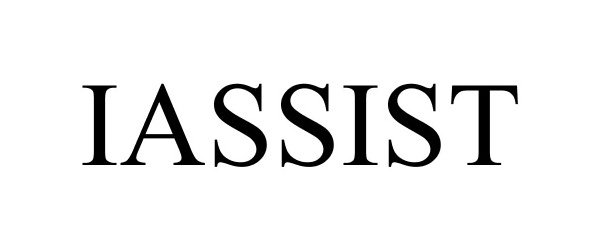 IASSIST 85125219 4061255 Live/Registered |
AssistRX, Inc. 2010-09-08 |
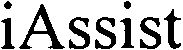 IASSIST 79101806 4103905 Dead/Cancelled |
SINTOKOGIO, LTD. 2011-07-19 |
 IASSIST 76609005 3510344 Dead/Cancelled |
Toshiba Medical Systems Corporation 2004-08-27 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.