NDC 0006-0031
FOSAMAX
Alendronate Sodium
FOSAMAX is a Oral Tablet in the Human Prescription Drug category. It is labeled and distributed by Merck Sharp & Dohme Corp.. The primary component is Alendronate Sodium.
| Product ID | 0006-0031_37333f48-be6f-455d-878b-328bc51259c4 |
| NDC | 0006-0031 |
| Product Type | Human Prescription Drug |
| Proprietary Name | FOSAMAX |
| Generic Name | Alendronate Sodium |
| Dosage Form | Tablet |
| Route of Administration | ORAL |
| Marketing Start Date | 1995-09-29 |
| Marketing Category | NDA / NDA |
| Application Number | NDA020560 |
| Labeler Name | Merck Sharp & Dohme Corp. |
| Substance Name | ALENDRONATE SODIUM |
| Active Ingredient Strength | 70 mg/1 |
| Pharm Classes | Bisphosphonate [EPC],Diphosphonates [CS] |
| NDC Exclude Flag | N |
| Listing Certified Through | 2021-12-31 |
Packaging
NDC 0006-0031-44
4 TABLET in 1 BLISTER PACK (0006-0031-44)
| Marketing Start Date | 1995-09-29 |
| NDC Exclude Flag | N |
| Sample Package? | N |
NDC SPL Data Element Entries
NDC 0006-0031-44 [00006003144]
FOSAMAX TABLET
| Marketing Category | NDA |
| Application Number | NDA020560 |
| Product Type | HUMAN PRESCRIPTION DRUG |
| Billing Unit | EA |
| Marketing Start Date | 1995-09-29 |
NDC 0006-0031-21 [00006003121]
FOSAMAX TABLET
| Marketing Category | NDA |
| Application Number | NDA020560 |
| Product Type | HUMAN PRESCRIPTION DRUG |
| Billing Unit | EA |
| Marketing Start Date | 1995-09-29 |
| Marketing End Date | 2012-07-09 |
NDC 0006-0031-01 [00006003101]
FOSAMAX TABLET
| Marketing Category | NDA |
| Application Number | NDA020560 |
| Product Type | HUMAN PRESCRIPTION DRUG |
| Marketing Start Date | 1995-09-29 |
| Marketing End Date | 2012-07-09 |
Drug Details
Active Ingredients
| Ingredient | Strength |
|---|---|
| ALENDRONATE SODIUM | 70 mg/1 |
OpenFDA Data
| SPL SET ID: | f2dcfb39-a7c2-4e39-9e35-852a3a74f947 |
| Manufacturer | |
| UNII | |
| RxNorm Concept Unique ID - RxCUI |
Pharmacological Class
- Bisphosphonate [EPC]
- Diphosphonates [CS]
NDC Crossover Matching brand name "FOSAMAX" or generic name "Alendronate Sodium"
| NDC | Brand Name | Generic Name |
|---|---|---|
| 0006-0031 | FOSAMAX | alendronate sodium |
| 10135-696 | Alendronate | Alendronate Sodium |
| 10135-697 | Alendronate | Alendronate Sodium |
| 0054-0282 | Alendronate Sodium | Alendronate Sodium |
| 0115-1676 | Alendronate Sodium | Alendronate Sodium |
| 0115-1678 | Alendronate Sodium | Alendronate Sodium |
| 0115-1679 | Alendronate Sodium | Alendronate Sodium |
| 0115-1680 | Alendronate Sodium | Alendronate Sodium |
| 0115-1681 | Alendronate Sodium | Alendronate Sodium |
| 0378-3566 | Alendronate Sodium | alendronate sodium |
| 16252-599 | alendronate sodium | alendronate sodium |
| 16252-601 | alendronate sodium | alendronate sodium |
| 16714-631 | Alendronate Sodium | Alendronate Sodium |
| 16714-632 | Alendronate Sodium | Alendronate Sodium |
| 16714-633 | Alendronate Sodium | Alendronate Sodium |
| 21695-901 | Alendronate Sodium | alendronate sodium |
| 21695-902 | Alendronate Sodium | alendronate sodium |
| 33261-554 | Alendronate Sodium | Alendronate Sodium |
| 33261-820 | Alendronate Sodium | Alendronate Sodium |
| 0178-0101 | Binosto | alendronate sodium |
| 17139-400 | Binosto | alendronate sodium |
Trademark Results [FOSAMAX]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 FOSAMAX 76057522 2747775 Live/Registered |
MERCK SHARP & DOHME CORP. 2000-05-26 |
 FOSAMAX 75044414 2073389 Dead/Cancelled |
MERCK & CO., Inc. 1996-01-16 |
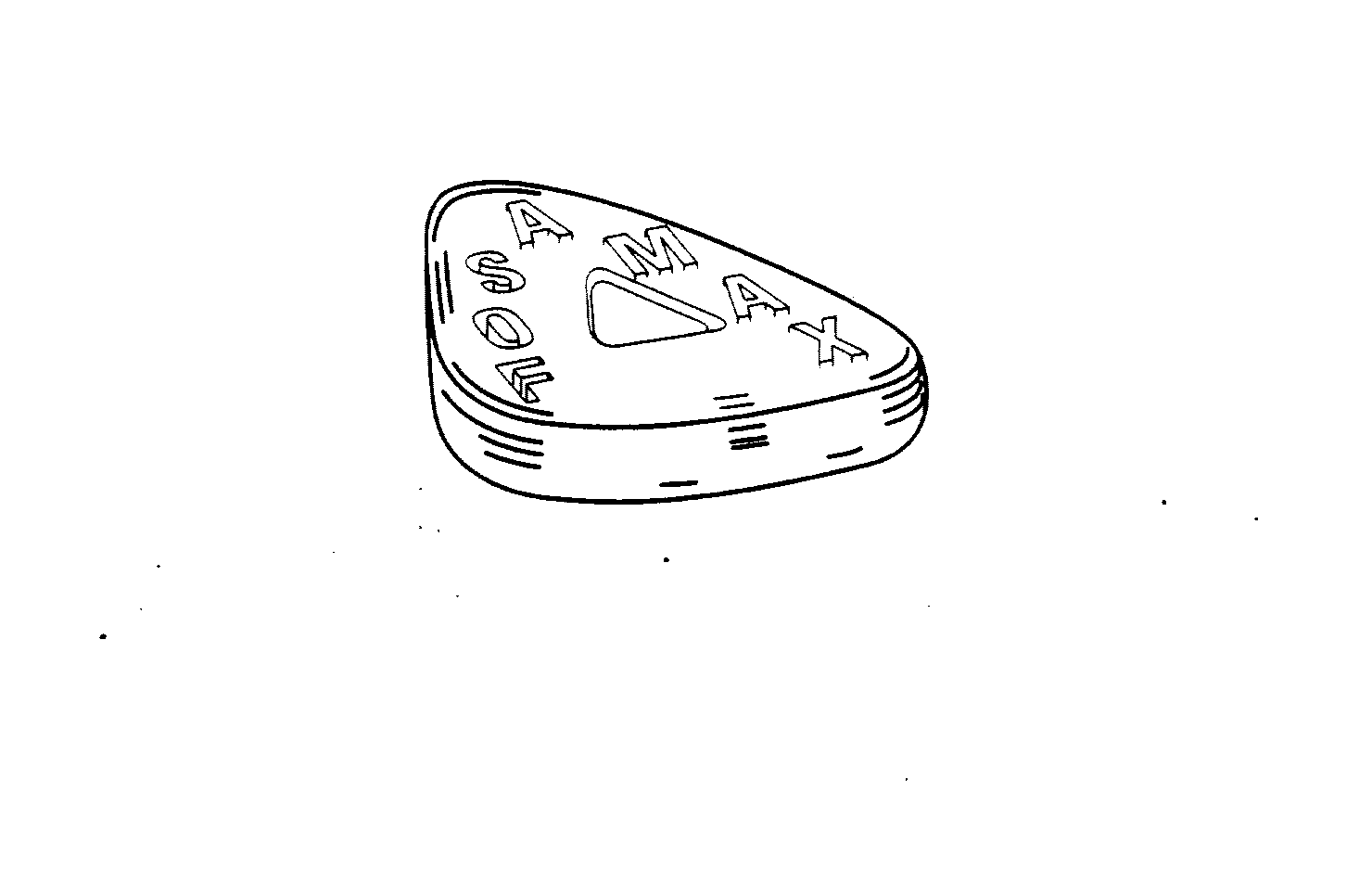 FOSAMAX 75044413 2438692 Live/Registered |
MERCK SHARP & DOHME CORP. 1996-01-16 |
 FOSAMAX 74132851 1710682 Live/Registered |
MERCK SHARP & DOHME CORP. 1991-01-22 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.