NDC 0071-0350
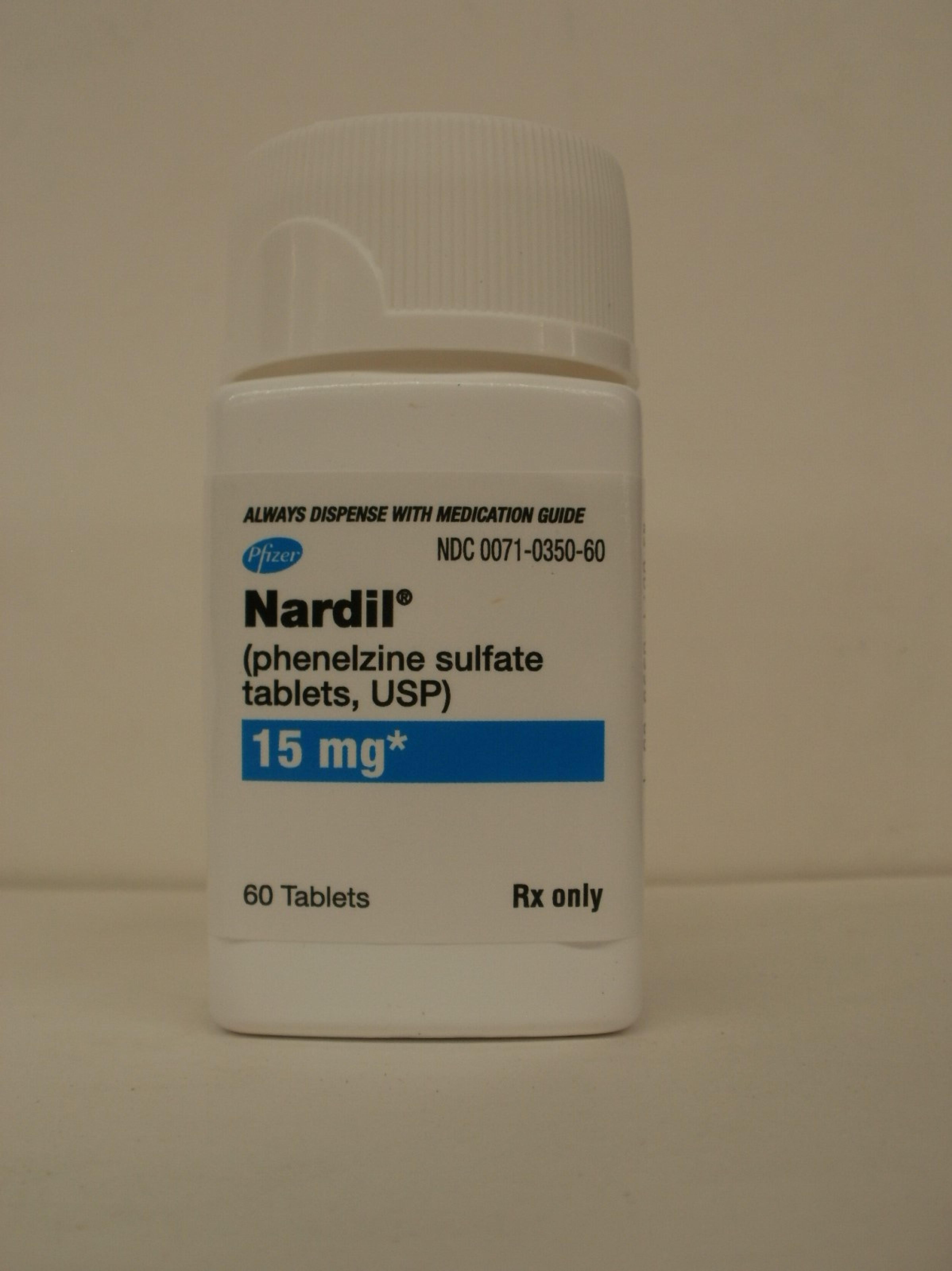
Nardil
Phenelzine Sulfate
Nardil is a Oral Tablet, Film Coated in the Human Prescription Drug category. It is labeled and distributed by Parke-davis Div Of Pfizer Inc. The primary component is Phenelzine Sulfate.
| Product ID | 0071-0350_521d17e0-13d4-4a60-aa3f-0b900aab24f7 |
| NDC | 0071-0350 |
| Product Type | Human Prescription Drug |
| Proprietary Name | Nardil |
| Generic Name | Phenelzine Sulfate |
| Dosage Form | Tablet, Film Coated |
| Route of Administration | ORAL |
| Marketing Start Date | 1961-06-09 |
| Marketing Category | NDA / NDA |
| Application Number | NDA011909 |
| Labeler Name | Parke-Davis Div of Pfizer Inc |
| Substance Name | PHENELZINE SULFATE |
| Active Ingredient Strength | 15 mg/1 |
| Pharm Classes | Monoamine Oxidase Inhibitor [EPC],Monoamine Oxidase Inhibitors [MoA] |
| NDC Exclude Flag | N |
| Listing Certified Through | 2020-12-31 |
Packaging
NDC 0071-0350-60
60 TABLET, FILM COATED in 1 BOTTLE (0071-0350-60)
| Marketing Start Date | 1961-06-09 |
| NDC Exclude Flag | N |
| Sample Package? | N |
NDC SPL Data Element Entries
NDC 0071-0350-60 [00071035060]
Nardil TABLET, FILM COATED
| Marketing Category | NDA |
| Application Number | NDA011909 |
| Product Type | HUMAN PRESCRIPTION DRUG |
| Billing Unit | EA |
| Marketing Start Date | 1961-06-09 |
Drug Details
Active Ingredients
| Ingredient | Strength |
|---|---|
| PHENELZINE SULFATE | 15 mg/1 |
OpenFDA Data
| SPL SET ID: | 513a41d0-37d4-4355-8a6d-a2c643bce6fa |
| Manufacturer | |
| UNII | |
| RxNorm Concept Unique ID - RxCUI | |
| UPC Code |
Pharmacological Class
- Monoamine Oxidase Inhibitor [EPC]
- Monoamine Oxidase Inhibitors [MoA]
- Monoamine Oxidase Inhibitor [EPC]
- Monoamine Oxidase Inhibitors [MoA]
NDC Crossover Matching brand name "Nardil" or generic name "Phenelzine Sulfate"
| NDC | Brand Name | Generic Name |
|---|---|---|
| 0071-0350 | Nardil | Phenelzine Sulfate |
| 24822-119 | Greenstone Brand Phenelzine Sulfate | PHENELZINE SULFATE |
| 24822-350 | Nardil Phenelzine Sulfate | PHENELZINE SULFATE |
| 40032-360 | Phenelzine Sulfate | Phenelzine Sulfate |
| 43386-360 | Phenelzine Sulfate | Phenelzine Sulfate |
| 59762-0119 | phenelzine sulfate | Phenelzine Sulfate |
Trademark Results [Nardil]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 NARDIL 72075232 0694808 Live/Registered |
Warner-Lambert Pharmaceutical Company 1959-06-05 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.