NDC 24208-353
Alrex
Loteprednol Etabonate
Alrex is a Ophthalmic Suspension/ Drops in the Human Prescription Drug category. It is labeled and distributed by Bausch & Lomb Incorporated. The primary component is Loteprednol Etabonate.
| Product ID | 24208-353_138577af-1a4e-48fb-b088-47f54c896442 |
| NDC | 24208-353 |
| Product Type | Human Prescription Drug |
| Proprietary Name | Alrex |
| Generic Name | Loteprednol Etabonate |
| Dosage Form | Suspension/ Drops |
| Route of Administration | OPHTHALMIC |
| Marketing Start Date | 1998-03-09 |
| Marketing Category | NDA / NDA |
| Application Number | NDA020803 |
| Labeler Name | Bausch & Lomb Incorporated |
| Substance Name | LOTEPREDNOL ETABONATE |
| Active Ingredient Strength | 2 mg/mL |
| Pharm Classes | Corticosteroid Hormone Receptor Agonists [MoA], Corticosteroid [EPC] |
| NDC Exclude Flag | N |
| Listing Certified Through | 2023-12-31 |
Packaging
NDC 24208-353-01
1 BOTTLE, DROPPER in 1 CARTON (24208-353-01) > 1 mL in 1 BOTTLE, DROPPER
| Marketing Start Date | 1998-03-09 |
| NDC Exclude Flag | N |
| Sample Package? | Y |
NDC SPL Data Element Entries
NDC 24208-353-05 [24208035305]
ALREX SUSPENSION/ DROPS
| Marketing Category | NDA |
| Application Number | NDA020803 |
| Product Type | HUMAN PRESCRIPTION DRUG |
| Billing Unit | ML |
| Marketing Start Date | 1998-03-09 |
NDC 24208-353-10 [24208035310]
ALREX SUSPENSION/ DROPS
| Marketing Category | NDA |
| Application Number | NDA020803 |
| Product Type | HUMAN PRESCRIPTION DRUG |
| Billing Unit | ML |
| Marketing Start Date | 1998-03-09 |
NDC 24208-353-25 [24208035325]
ALREX SUSPENSION/ DROPS
| Marketing Category | NDA |
| Application Number | NDA020803 |
| Product Type | HUMAN PRESCRIPTION DRUG |
| Marketing Start Date | 2011-04-08 |
| Marketing End Date | 2013-04-12 |
NDC 24208-353-01 [24208035301]
ALREX SUSPENSION/ DROPS
| Marketing Category | NDA |
| Application Number | NDA020803 |
| Product Type | HUMAN PRESCRIPTION DRUG |
| Marketing Start Date | 1998-03-09 |
Drug Details
Active Ingredients
| Ingredient | Strength |
|---|---|
| LOTEPREDNOL ETABONATE | 2 mg/mL |
OpenFDA Data
| SPL SET ID: | 21c76da0-50a1-41c0-91b8-8cc4dcc27f60 |
| Manufacturer | |
| UNII | |
| RxNorm Concept Unique ID - RxCUI |
Pharmacological Class
- Corticosteroid [EPC]
- Corticosteroid Hormone Receptor Agonists [MoA]
Medicade Reported Pricing
24208035310 ALREX 0.2% EYE DROPS
Pricing Unit: ML | Drug Type:
24208035305 ALREX 0.2% EYE DROPS
Pricing Unit: ML | Drug Type:
NDC Crossover Matching brand name "Alrex" or generic name "Loteprednol Etabonate"
| NDC | Brand Name | Generic Name |
|---|---|---|
| 24208-353 | Alrex | loteprednol etabonate |
| 71571-333 | Eysuvis | loteprednol etabonate |
| 71571-121 | Inveltys | loteprednol etabonate |
| 24208-299 | LOTEMAX | loteprednol etabonate |
| 24208-443 | Lotemax | loteprednol etabonate |
| 24208-503 | LOTEMAX | loteprednol etabonate |
| 24208-507 | Lotemax SM | loteprednol etabonate |
| 17478-830 | Loteprednol Etabonate | Loteprednol Etabonate |
| 24208-508 | Loteprednol Etabonate | loteprednol etabonate |
| 50383-265 | Loteprednol Etabonate | Loteprednol Etabonate |
| 62756-232 | Loteprednol Etabonate | Loteprednol Etabonate |
| 68682-299 | Loteprednol Etabonate | loteprednol etabonate |
Trademark Results [Alrex]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 ALREX 75442434 2360654 Dead/Cancelled |
BAUSCH & LOMB INCORPORATED 1998-03-02 |
 ALREX 75283586 2265425 Live/Registered |
BAUSCH & LOMB INCORPORATED 1997-04-29 |
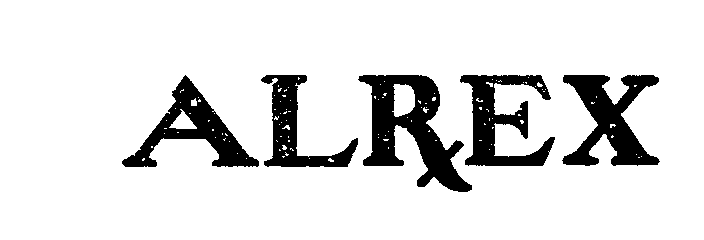 ALREX 72325406 0887079 Dead/Expired |
KENNETH SMITH, INC. 1969-04-23 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.