NDC 43598-921
Xeglyze
Abametapir
Xeglyze is a Topical Lotion in the Human Prescription Drug category. It is labeled and distributed by Dr. Reddy's Laboratories Inc.. The primary component is Abametapir.
| Product ID | 43598-921_79de62dc-8192-24c3-df79-2e75a4b6c85b |
| NDC | 43598-921 |
| Product Type | Human Prescription Drug |
| Proprietary Name | Xeglyze |
| Generic Name | Abametapir |
| Dosage Form | Lotion |
| Route of Administration | TOPICAL |
| Marketing Start Date | 2021-01-29 |
| Marketing Category | NDA / NDA |
| Application Number | NDA206966 |
| Labeler Name | Dr. Reddy's Laboratories Inc. |
| Substance Name | ABAMETAPIR |
| Active Ingredient Strength | 1 g/100g |
| NDC Exclude Flag | N |
| Listing Certified Through | 2023-12-31 |
Packaging
NDC 43598-921-11
1 BOTTLE in 1 CARTON (43598-921-11) > 200 g in 1 BOTTLE
| Marketing Start Date | 2021-01-29 |
| NDC Exclude Flag | N |
| Sample Package? | N |
Drug Details
Trademark Results [Xeglyze]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
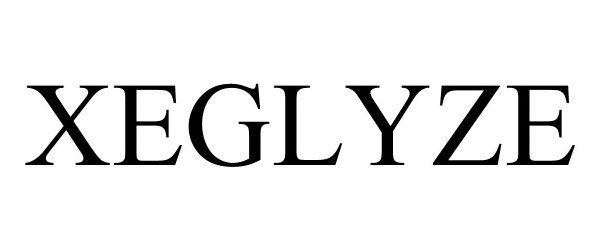 XEGLYZE 90812737 not registered Live/Pending |
Dr. Reddy's Laboratories SA 2021-07-06 |
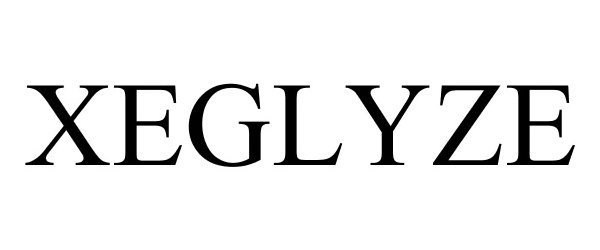 XEGLYZE 87514849 not registered Live/Pending |
Dr. Reddy's Laboratories, S. A. 2017-07-03 |
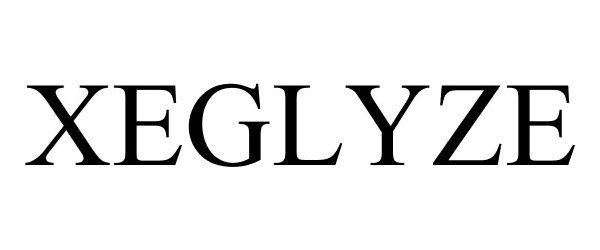 XEGLYZE 86164493 not registered Dead/Abandoned |
DR. REDDY'S LABORATORIES, S.A 2014-01-14 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.