FIBER THERAPY- methylcellulose powder, for solution
FIBER THERAPY by
Drug Labeling and Warnings
FIBER THERAPY by is a Otc medication manufactured, distributed, or labeled by Preferred Pharmaceuticals Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Active ingredient (in each 19g adult dose/ rounded tablespoon)
- Purpose
- Uses
-
Warnings
CHOKING: taking this product without adequate fluid may cause it to swell and block your throat or esophagus and may cause choking. Do not take this product if you have difficulty in swallowing. If you experience chest pain, vomiting, or difficulty in swallowing or breathing after taking this product, seek immediate medical attention.
Ask a doctor before use if you have
- abdominal pain, nausea or vomiting
- a sudden change in bowel habits that persists over a period of 2 weeks
- sensitivity to any of the ingredients
Stop use and ask a doctor if
- you have rectal bleeding
- you fail to have a bowl movement after use.
- These could be signs of a serious condition.
-
Directions
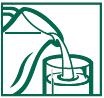
- MIX THIS PRODUCT (CHILD OR ADULT DOSE) WITH AT LEAST 8 OUNCES (A FULL GLASS) OF WATER OR OTHER FLUID. TAKING THIS PRODUCT WITHOUT ENOUGH LIQUID MAY CAUSE CHOKING. SEE CHOKING WARNING
- use product at the first sign of constipation or irregularity
- put one dose in a full glass of cold water
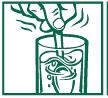
- stir briskly and drink promptly
- drinking another glass of water is helpful
Age
Dose
adults & children above 12 years of age and over
one rounded tablespoon. (19 g) in 8 ounces of water fluid up to 3 times daily at the first sign of constipation or irregularity
children 6 - 12 years of age
half the adult dose in 8 ounces of water once daily.
children under 6 years of age
ask a doctor
- Other information
- Inactive ingredients
-
Questions or comments?
1-800-616-2471
*This product is not manufactured or distributed by GlaxoSmithKline the distributor of CITRUCEL®
Distributed by:
MAJOR® PHARMACEUTICALS
17177 N Laurel Park Drive, Suite 233
Relabeled By: Preferred Pharmaceuticals Inc.
Livonia, MI 48152, USA
Relabeled By: Preferred Pharmaceuticals Inc.
DIRECTIONS FOR USE
1. Fill glass with at least 8 ounces of cold water.
2. Add the adult or child dose listed in the directions. 3. Stir briskly until dissolved and drink promptly. Drinking an additional glass of water is helpful.
-
Principal Display Panel
NDC: 68788-7995-4
Compare to the Active Ingredient in CITRUCEL®*
FIBER THERAPY
Methylcellulose
Easy to Mix No Gritty Texture
100% Soluble Fiber for Controlled Regularity
ORANGE FLAVOR
NET WT 16 OZ (454 GRAMS)
-
INGREDIENTS AND APPEARANCE
FIBER THERAPY
methylcellulose powder, for solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 68788-7995(NDC:0904-5675) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength METHYLCELLULOSE (4000 MPA.S) (UNII: MRJ667KA5E) (METHYLCELLULOSE (4000 MPA.S) - UNII:MRJ667KA5E) METHYLCELLULOSE (4000 MPA.S) 2 g in 19 g Inactive Ingredients Ingredient Name Strength CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) MALTODEXTRIN (UNII: 7CVR7L4A2D) POTASSIUM CITRATE (UNII: EE90ONI6FF) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SUCROSE (UNII: C151H8M554) Product Characteristics Color ORANGE Score Shape Size Flavor ORANGE Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 68788-7995-4 454 g in 1 BOTTLE; Type 0: Not a Combination Product 08/09/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 334 08/09/2021 Labeler - Preferred Pharmaceuticals Inc. (791119022) Registrant - Preferred Pharmaceuticals Inc. (791119022) Establishment Name Address ID/FEI Business Operations Preferred Pharmaceuticals Inc. 791119022 RELABEL(68788-7995)
Trademark Results [FIBER THERAPY]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 FIBER THERAPY 87195645 5444200 Live/Registered |
Henkel AG & Co. KGaA 2016-10-06 |
 FIBER THERAPY 78137531 not registered Dead/Abandoned |
BISSELL Homecare, Inc. 2002-06-21 |
 FIBER THERAPY 73671259 1495554 Dead/Cancelled |
SANDOZ PHARMACEUTICALS CORPORATION 1987-07-10 |
© 2024 FDA.report
This site is not affiliated with or endorsed by the FDA.