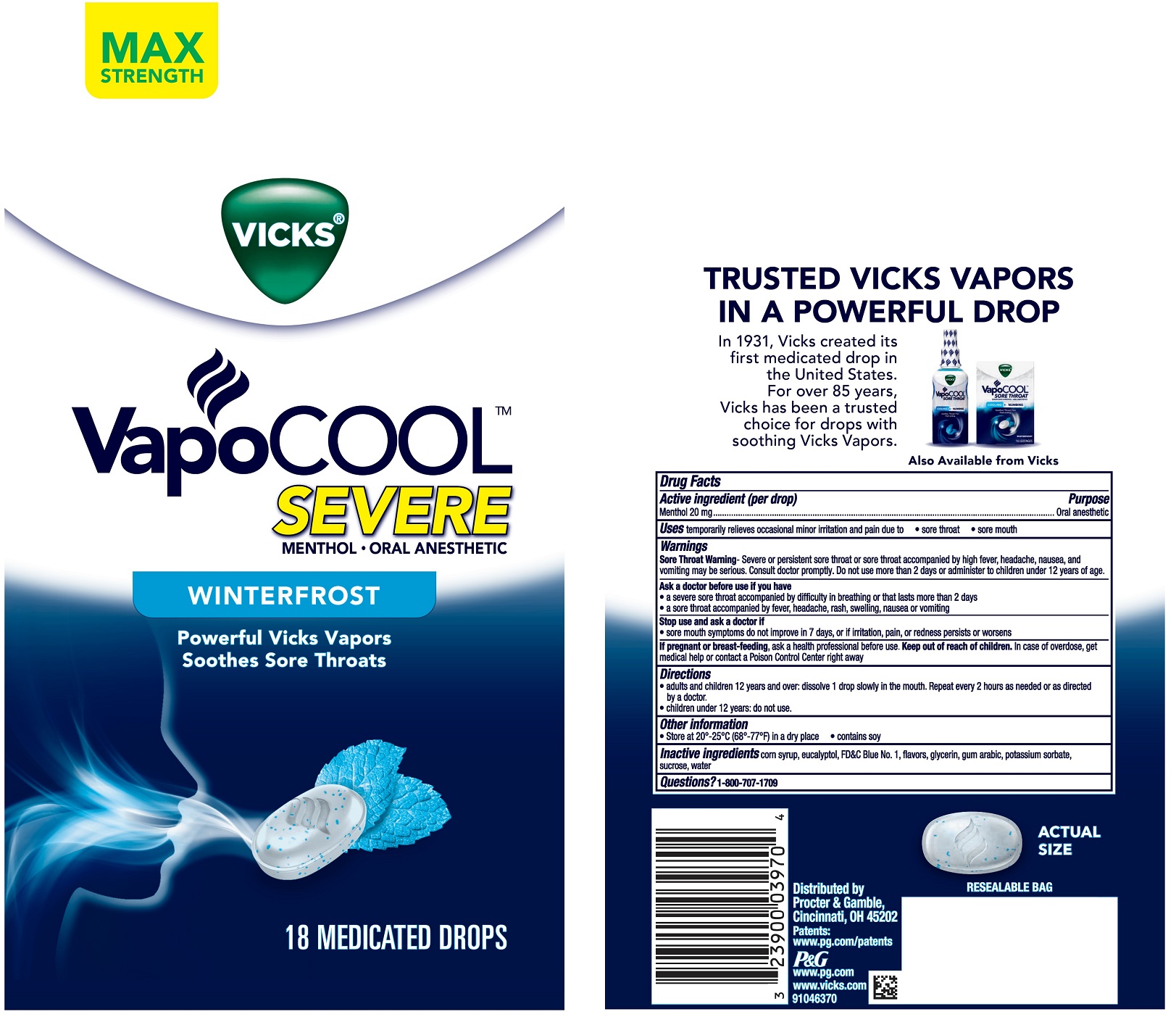
Vicks ®VapoCOOL ™SEVERE MENTHOL • ORAL ANESTHETIC DROPS WINTERFROST
Vicks VapoCOOL by
Drug Labeling and Warnings
Vicks VapoCOOL by is a Otc medication manufactured, distributed, or labeled by The Procter & Gamble Manufacturing Company. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
VICKS VAPOCOOL SEVERE WINTERFROST- menthol lozenge
The Procter & Gamble Manufacturing Company
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Vicks
®VapoCOOL
™SEVERE MENTHOL ORAL ANESTHETIC DROPS
WINTERFROST
Ask a doctor before use if you have
- a severe sore throat accompanied by difficulty in breathing or that lasts more than 2 days
- a sore throat accompanied by fever, headache, rash, swelling, nausea or vomiting
Stop use and ask a doctor if
- sore mouth symptoms do not improve in 7 days, or if irritation, pain, or redness persists or worsens.
Directions
- adults and children 12 years and over: dissolve 1 drop slowly in the mouth. Repeat every 2 hours as needed or as directed by a doctor.
- children 12 years and under: do not use
| VICKS VAPOCOOL
SEVERE WINTERFROST
menthol lozenge |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - The Procter & Gamble Manufacturing Company (004238200) |
Revised: 9/2023
Document Id: 0657c9bc-9277-94e8-e063-6394a90a35cd
Set id: 01ce6525-ef2e-bf24-e063-6294a90abb73
Version: 4
Effective Time: 20230927
The Pr
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.