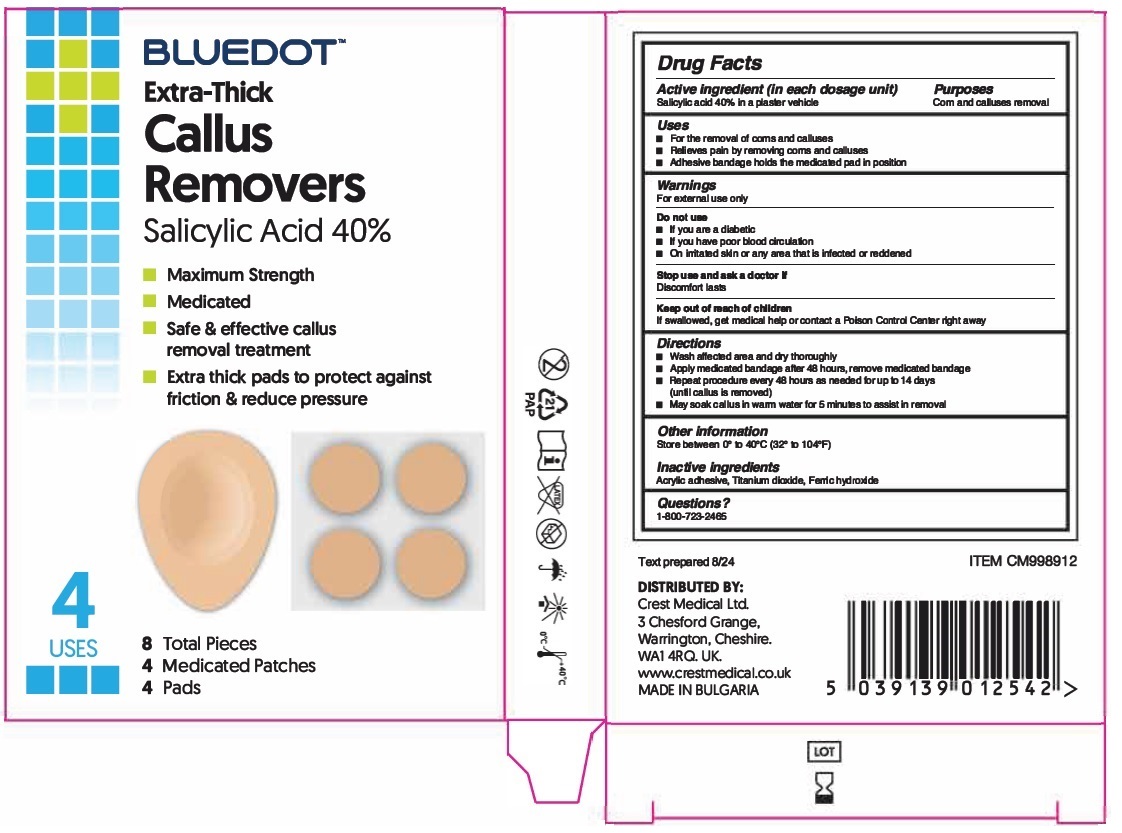
BLUEDOTTM Extra-Thick Callus Removers
BLUEDOT Callus Removers by
Drug Labeling and Warnings
BLUEDOT Callus Removers by is a Otc medication manufactured, distributed, or labeled by CREST MEDICAL LIMITED, KRE EOOD. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
BLUEDOT CALLUS REMOVERS- salicylic acid patch
CREST MEDICAL LIMITED
----------
BLUEDOTTM Extra-Thick Callus Removers
Uses
For the removal of corns and calluses
Relieves pain by removing corns and calluses
Adhesive bandage holds the medicated pad in position
Warnings
For external use only
Do not use
If you are a diabetic
If you have poor blood circulation
On irritated skin or any area that is infected or reddened
Stop use and ask a doctor if
Discomfort lasts
Directions
Wash affected area and dry thoroughly
Apply medicated bandage after 48 hours, remove medicated bandage
Repeat procedure every 48 hours as needed for up to 14 days (until callus is removed)
May soak callus in warm water for 5 minutes to assist in removal
| BLUEDOT CALLUS REMOVERS
salicylic acid patch |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Labeler - CREST MEDICAL LIMITED (238858539) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| KRE EOOD | 565504983 | manufacture(84713-103) , label(84713-103) , pack(84713-103) | |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.