BORTEZOMIB injection, powder, lyophilized, for solution
Bortezomib by
Drug Labeling and Warnings
Bortezomib by is a Prescription medication manufactured, distributed, or labeled by Fresenius Kabi USA, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use BORTEZOMIB FOR INJECTION safely and effectively. See full prescribing information for BORTEZOMIB FOR INJECTION.
BORTEZOMIB for injection, for intravenous use
Initial U.S. Approval: 2003RECENT MAJOR CHANGES
Dosage and Administration ( 2.8) 6/2018 INDICATIONS AND USAGE
DOSAGE AND ADMINISTRATION
- For intravenous use only. Exercise caution when calculating the volume to be administered. ( 2.1, 2.8.)
- The recommended starting dose of Bortezomib for Injection is 1.3 mg/m 2 administered as a 3 to 5 second bolus intravenous injection. ( 2.2, 2.4)
- Hepatic Impairment: Use a lower starting dose for patients with moderate or severe hepatic impairment. ( 2.6)
- Dose must be individualized to prevent overdose. ( 2.8)
DOSAGE FORMS AND STRENGTHS
For injection: Single-dose vial contains 3.5 mg of bortezomib as lyophilized powder for reconstitution. ( 3)
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
- Peripheral Neuropathy: Manage with dose modification or discontinuation. ( 2.5) Patients with pre-existing severe neuropathy should be treated with Bortezomib for Injection only after careful risk-benefit assessment. ( 2.5, 5.1)
- Hypotension: Use caution when treating patients taking anti hypertensives, with a history of syncope, or with dehydration. ( 5.2)
- Cardiac Toxicity: Worsening of and development of cardiac failure has occurred. Closely monitor patients with existing heart disease or risk factors for heart disease. ( 5.3)
- Pulmonary Toxicity: Acute respiratory syndromes have occurred. Monitor closely for new or worsening symptoms. ( 5.4)
- Posterior Reversible Encephalopathy Syndrome: Consider MRI imaging for onset of visual or neurological symptoms; discontinue Bortezomib for Injection if suspected. ( 5.5)
- Gastrointestinal Toxicity: Nausea, diarrhea, constipation, and vomiting may require use of antiemetic and antidiarrheal medications or fluid replacement. ( 5.6)
- Thrombocytopenia or Neutropenia: Monitor complete blood counts regularly throughout treatment. ( 5.7)
- Tumor Lysis Syndrome: Closely monitor patients with high tumor burden. ( 5.8)
- Hepatic Toxicity: Monitor hepatic enzymes during treatment. ( 5.9)
- Embryo-fetal Toxicity: Bortezomib can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and to avoid pregnancy. ( 5.10)
ADVERSE REACTIONS
Most commonly reported adverse reactions (incidence ≥ 20%) in clinical studies include nausea, diarrhea, thrombocytopenia, neutropenia, peripheral neuropathy, fatigue, neuralgia, anemia, leukopenia, constipation, vomiting, lymphopenia, rash, pyrexia, and anorexia. ( 6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Fresenius Kabi USA, LLC at 1-800-551-7176 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- Coadministration with strong CYP3A4 inhibitors can increase bortezomib exposure. Monitor for signs of bortezomib toxicity in patients receiving Bortezomib for Injection with strong CYP3A4 inhibitors. ( 7.1)
- Coadministration with strong CYP3A4 inducers can decrease bortezomib exposure. Avoid strong CYP3A4 inducers. ( 7.3)
USE IN SPECIFIC POPULATIONS
- Patients with diabetes may require close monitoring of blood glucose and adjustment of anti-diabetic medication. ( 8.8)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 9/2018
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
1.1 Multiple Myeloma
1.2 Mantle Cell Lymphoma
2 DOSAGE AND ADMINISTRATION
2.1 Important Dosing Guidelines
2.2 Dosage in Previously Untreated Multiple Myeloma
2.3 Dose Modification Guidelines for Bortezomib for Injection When Given in Combination with Melphalan and Prednisone
2.4 Dosage and Dose Modifications for Relapsed Multiple Myeloma and Relapsed Mantle Cell Lymphoma
2.5 Dose Modifications for Peripheral Neuropathy
2.6 Dosage in Patients with Hepatic Impairment
2.7 Administration Precautions
2.8 Reconstitution/Preparation for Intravenous Administration
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Peripheral Neuropathy
5.2 Hypotension
5.3 Cardiac Toxicity
5.4 Pulmonary Toxicity
5.5 Posterior Reversible Encephalopathy Syndrome (PRES)
5.6 Gastrointestinal Toxicity
5.7 Thrombocytopenia/Neutropenia
5.8 Tumor Lysis Syndrome
5.9 Hepatic Toxicity
5.10 Embryo-fetal Toxicity
6 ADVERSE REACTIONS
6.1 Clinical Trials Safety Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Effect of Strong CYP3A4 Inhibitors on Bortezomib
7.3 Effect of Strong CYP3A4 Inducers on Bortezomib
7.4 Effect of Dexamethasone on Bortezomib
7.5 Effect of Melphalan-Prednisone on Bortezomib
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Patients with Renal Impairment
8.7 Patients with Hepatic Impairment
8.8 Patients with Diabetes
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
13.2 Animal Toxicology and/or Pharmacology
14 CLINICAL STUDIES
14.1 Multiple Myeloma
14.2 Mantle Cell Lymphoma
15 REFERENCES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 Important Dosing Guidelines
Bortezomib for Injection is for intravenous use only. Do not administer Bortezomib for Injection by any other route.
The recommended starting dose of Bortezomib for Injection is 1.3 mg/m 2. Bortezomib for Injection may be administered intravenously at a concentration of 1 mg/mL [see Dosage and Administration ( 2.8)] .
When administered intravenously, Bortezomib for Injection is administered as a 3 to 5 second bolus intravenous injection.
2.2 Dosage in Previously Untreated Multiple Myeloma
Bortezomib for Injection is administered in combination with oral melphalan and oral prednisone for nine 6-week treatment cycles as shown in Table 1. In Cycles 1 to 4, Bortezomib for Injection is administered twice weekly (days 1, 4, 8, 11, 22, 25, 29 and 32). In Cycles 5 to 9, Bortezomib for Injection is administered once weekly (days 1, 8, 22 and 29). At least 72 hours should elapse between consecutive doses of Bortezomib for Injection.
Table 1: Dosage Regimen for Patients with Previously Untreated Multiple Myeloma Twice Weekly Bortezomib (Cycles 1 to 4) Week 1 2 3 4 5 6 Bortezomib for Injection
(1.3 mg/m 2)Day 1 -- -- Day 4 Day 8 Day 11 rest period Day 22 Day 25 Day 29 Day 32 rest period Melphalan (9 mg/m 2)
Prednisone (60 mg/m 2)Day 1 Day 2 Day 3 Day 4 -- -- rest period -- -- -- -- rest period Once Weekly Bortezomib for Injection (Cycles 5 to 9 when used in combination with Melphalan and Prednisone) Week 1 2 3 4 5 6 Bortezomib for Injection
(1.3 mg/m 2)Day 1 -- -- Day 8 rest period Day 22 Day 29 rest period Melphalan (9 mg/m 2)
Prednisone (60 mg/m 2)Day 1 Day 2 Day 3 Day 4 -- -- rest period -- -- -- -- rest period 2.3 Dose Modification Guidelines for Bortezomib for Injection When Given in Combination with Melphalan and Prednisone
Prior to initiating any cycle of therapy with Bortezomib for Injection in combination with melphalan and prednisone:
- Platelet count should be at least 70 x 10 9/L and the absolute neutrophil count (ANC) should be at least 1 x 10 9/L
- Non-hematological toxicities should have resolved to Grade 1 or baseline
Table 2: Dose Modifications during Cycles of Combination Bortezomib for Injection, Melphalan and Prednisone Therapy Toxicity Dose Modification or Delay Hematological toxicity during a cycle: If prolonged Grade 4 neutropenia or thrombocytopenia, or thrombocytopenia with bleeding is observed in the previous cycle Consider reduction of the melphalan dose by 25% in the next cycle If platelet count is not above 30 × 10 9/L or ANC is not above 0.75 x 10 9/L on a Bortezomib for Injection dosing day (other than day 1) Withhold Bortezomib for Injection dose If several Bortezomib for Injection doses in consecutive cycles are withheld due to toxicity
Reduce Bortezomib for Injection dose by 1 dose level (from 1.3 mg/m 2 to 1 mg/m 2, or from 1 mg/m 2 to 0.7 mg/m 2) Grade 3 or higher non-hematological toxicities Withhold Bortezomib for Injection therapy until symptoms of toxicity have resolved to Grade 1 or baseline. Then, Bortezomib for Injection may be reinitiated with one dose level reduction (from 1.3 mg/m 2 to 1 mg/m 2, or from 1 mg/m 2 to 0.7 mg/m 2). For bortezomib-related neuropathic pain and/or peripheral neuropathy, hold or modify Bortezomib for Injection as outlined in Table 3. For information concerning melphalan and prednisone, see manufacturer's prescribing information.
Dose modifications guidelines for peripheral neuropathy are provided [see Dosage and Administration ( 2.5)] .
2.4 Dosage and Dose Modifications for Relapsed Multiple Myeloma and Relapsed Mantle Cell Lymphoma
Bortezomib for Injection (1.3 mg/m 2/dose) is administered twice weekly for 2 weeks (Days 1, 4, 8, and 11) followed by a 10-day rest period (Days 12 to 21). For extended therapy of more than 8 cycles, Bortezomib for Injection may be administered on the standard schedule or, for relapsed multiple myeloma, on a maintenance schedule of once weekly for 4 weeks (Days 1, 8, 15, and 22) followed by a 13-day rest period (Days 23 to 35) [see Clinical Studies ( 14)] . At least 72 hours should elapse between consecutive doses of Bortezomib for Injection.
Bortezomib for Injection therapy should be withheld at the onset of any Grade 3 non-hematological or Grade 4 hematological toxicities excluding neuropathy as discussed below [see Warnings and Precautions ( 5)] . Once the symptoms of the toxicity have resolved, Bortezomib for Injection therapy may be reinitiated at a 25% reduced dose (1.3 mg/m 2/dose reduced to 1 mg/m 2/dose; 1 mg/m 2/dose reduced to 0.7 mg/m 2/dose).
For dose modifications guidelines for peripheral neuropathy see section 2.5.
2.5 Dose Modifications for Peripheral Neuropathy
Patients with pre-existing severe neuropathy should be treated with Bortezomib for Injection only after careful risk-benefit assessment.
Patients experiencing new or worsening peripheral neuropathy during Bortezomib for Injection therapy may require a decrease in the dose and/or a less dose-intense schedule.
For dose or schedule modification guidelines for patients who experience Bortezomib for Injection-related neuropathic pain and/or peripheral neuropathy see Table 3.
Table 3: Recommended Dose Modification for Bortezomib for Injection-related Neuropathic Pain and/or Peripheral Sensory or Motor Neuropathy *Grading based on NCI Common Terminology Criteria CTCAE v4.0
**Instrumental ADL: refers to preparing meals, shopping for groceries or clothes, using telephone, managing money etc
***Self care ADL: refers to bathing, dressing and undressing, feeding self, using the toilet, taking medications, and not bedridden
Severity of Peripheral Neuropathy
Signs and Symptoms*Modification of Dose and Regimen Grade 1 (asymptomatic; loss of deep tendon reflexes or paresthesia) without pain or loss of function No action Grade 1 with pain or Grade 2 (moderate symptoms; limiting instrumental Activities of Daily Living (ADL)**) Reduce Bortezomib for Injection to 1 mg/m 2 Grade 2 with pain or Grade 3 (severe symptoms; limiting self care ADL ***) Withhold Bortezomib for Injection therapy until toxicity resolves. When toxicity resolves reinitiate with a reduced dose of Bortezomib for Injection at 0.7 mg/m 2 once per week. Grade 4 (life-threatening consequences; urgent intervention indicated) Discontinue Bortezomib for Injection 2.6 Dosage in Patients with Hepatic Impairment
Do not adjust the starting dose for patients with mild hepatic impairment.
Start patients with moderate or severe hepatic impairment at a reduced dose of 0.7 mg/m 2 per injection during the first cycle, and consider subsequent dose escalation to 1 mg/m 2 or further dose reduction to 0.5 mg/m 2 based on patient tolerance (see Table 4) [see Use in Specific Populations ( 8.7) and Clinical Pharmacology ( 12.3)] .
Table 4: Recommended Starting Dose Modification for Bortezomib for Injection in Patients with Hepatic Impairment Abbreviations: SGOT = serum glutamic oxaloacetic transaminase; AST = aspartate aminotransferase; ULN = upper limit of the normal range.
Bilirubin Level SGOT (AST)
LevelsModification of Starting Dose Mild Less than or equal to 1 times ULN More than ULN None More than 1 to 1.5 times ULN Any None Moderate More than 1.5 to 3 times
ULNAny Reduce dose to 0.7 mg/m 2 in the first cycle. Consider dose escalation to 1 mg/m 2 or further dose reduction to 0.5 mg/m 2 in subsequent cycles based on patient tolerability. Severe More than 3 times ULN Any 2.7 Administration Precautions
The drug quantity contained in one vial (3.5 mg) may exceed the usual dose required. Use caution in calculating the dose to prevent overdose [see Dosage and Administration ( 2.8)] .
Bortezomib for Injection is a cytotoxic drug. Use procedures for proper handling and disposal [see How Supplied/Storage and Handling ( 16)] .
2.8 Reconstitution/Preparation for Intravenous Administration
Use proper aseptic technique. Reconstitute only with 0.9% sodium chloride. The reconstituted product should be a clear and colorless solution.
For each 3.5 mg single-dose vial of Bortezomib for Injection reconstitute with the following volume of 0.9% sodium chloride ( Table 5):
Table 5: Reconstitution Volumes and Final Concentration for Intravenous Administration Route of
administrationBortezomib
(mg/vial)Diluent
(0.9% Sodium
Chloride)Final Bortezomib
concentration
(mg/mL)Intravenous 3.5 mg 3.5 mL 1 mg/mL Dose must be individualized to prevent overdosage. After determining patient body surface area (BSA) in square meters, use the following equation to calculate the total volume (mL) of reconstituted Bortezomib for Injection to be administered:
- Intravenous Administration [1 mg/mL concentration]
Bortezomib for Injection dose (mg/m 2) x patient BSA (m 2) = Total Bortezomib for Injection volume (mL) to be administered 1 mg/mL A sticker that indicates the route of administration is provided with each Bortezomib for Injection vial. The sticker should be placed directly on the syringe of Bortezomib for Injection once Bortezomib for Injection is prepared to help alert practitioners of the correct route of administration for Bortezomib for Injection.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit. If any discoloration or particulate matter is observed, the reconstituted product should not be used.
Bortezomib for Injection contains no antimicrobial preservative. Administer reconstituted Bortezomib for Injection within 8 hours of preparation. When reconstituted as directed, Bortezomib for Injection may be stored at 25°C (77°F). The reconstituted material may be stored in the original vial and/or the syringe prior to administration. The product may be stored for up to 8 hours in a syringe; however, total storage time for the reconstituted material must not exceed 8 hours when exposed to normal indoor lighting.
Stability: Unopened vials of Bortezomib for Injection are stable until the date indicated on the package when stored in the original package protected from light.
-
3 DOSAGE FORMS AND STRENGTHS
For injection: 3.5 mg of bortezomib as a white to off-white lyophilized powder in a single-dose vial for reconstitution [see Dosage and Administration ( 2.8)] .
-
4 CONTRAINDICATIONS
Bortezomib for Injection is contraindicated in patients with hypersensitivity (not including local reactions) to bortezomib, boron, boric acid or glycine. Reactions have included anaphylactic reactions [see Adverse Reactions ( 6.1)] .
Bortezomib for Injection is contraindicated for intrathecal administration. Fatal events have occurred with intrathecal administration of bortezomib.
-
5 WARNINGS AND PRECAUTIONS
5.1 Peripheral Neuropathy
Bortezomib treatment causes a peripheral neuropathy that is predominantly sensory; however, cases of severe sensory and motor peripheral neuropathy have been reported. Patients with pre-existing symptoms (numbness, pain or a burning feeling in the feet or hands) and/or signs of peripheral neuropathy may experience worsening peripheral neuropathy (including ≥ Grade 3) during treatment with bortezomib. Patients should be monitored for symptoms of neuropathy, such as a burning sensation, hyperesthesia, hypoesthesia, paresthesia, discomfort, neuropathic pain or weakness.
Patients experiencing new or worsening peripheral neuropathy during Bortezomib for Injection therapy may require a decrease in the dose and/or a less dose-intense schedule [see Dosage and Administration ( 2.5)] . In the bortezomib versus dexamethasone phase 3 relapsed multiple myeloma study, improvement in or resolution of peripheral neuropathy was reported in 48% of patients with ≥ Grade 2 peripheral neuropathy following dose adjustment or interruption. Improvement in or resolution of peripheral neuropathy was reported in 73% of patients who discontinued due to Grade 2 neuropathy or who had ≥ Grade 3 peripheral neuropathy in the phase 2 multiple myeloma studies [see Adverse Reactions ( 6.1)] . The long-term outcome of peripheral neuropathy has not been studied in mantle cell lymphoma.
5.2 Hypotension
The incidence of hypotension (postural, orthostatic, and hypotension NOS) was 8%. These events are observed throughout therapy. Caution should be used when treating patients with a history of syncope, patients receiving medications known to be associated with hypotension, and patients who are dehydrated. Management of orthostatic/postural hypotension may include adjustment of antihypertensive medications, hydration, and administration of mineralocorticoids and/or sympathomimetics [see Adverse Reactions ( 6.1)] .
5.3 Cardiac Toxicity
Acute development or exacerbation of congestive heart failure and new onset of decreased left ventricular ejection fraction have occurred during bortezomib therapy, including reports in patients with no risk factors for decreased left ventricular ejection fraction. Patients with risk factors for, or existing heart disease should be closely monitored. In the relapsed multiple myeloma study of bortezomib versus dexamethasone, the incidence of any treatment-related cardiac disorder was 8% and 5% in the bortezomib and dexamethasone groups, respectively. The incidence of adverse reactions suggestive of heart failure (acute pulmonary edema, pulmonary edema, cardiac failure, congestive cardiac failure, cardiogenic shock) was ≤ 1% for each individual reaction in the bortezomib group. In the dexamethasone group the incidence was ≤ 1% for cardiac failure and congestive cardiac failure; there were no reported reactions of acute pulmonary edema, pulmonary edema, or cardiogenic shock. There have been isolated cases of QT-interval prolongation in clinical studies; causality has not been established.
5.4 Pulmonary Toxicity
Acute Respiratory Distress Syndrome (ARDS) and acute diffuse infiltrative pulmonary disease of unknown etiology such as pneumonitis, interstitial pneumonia, lung infiltration have occurred in patients receiving bortezomib. Some of these events have been fatal.
In a clinical trial, the first two patients given high-dose cytarabine (2g/m 2 per day) by continuous infusion with daunorubicin and bortezomib for relapsed acute myelogenous leukemia died of ARDS early in the course of therapy.
There have been reports of pulmonary hypertension associated with bortezomib administration in the absence of left heart failure or significant pulmonary disease.
In the event of new or worsening cardiopulmonary symptoms, consider interrupting Bortezomib for Injection until a prompt and comprehensive diagnostic evaluation is conducted.
5.5 Posterior Reversible Encephalopathy Syndrome (PRES)
Posterior Reversible Encephalopathy Syndrome (PRES; formerly termed Reversible Posterior Leukoencephalopathy Syndrome (RPLS)) has occurred in patients receiving bortezomib. PRES is a rare, reversible, neurological disorder which can present with seizure, hypertension, headache, lethargy, confusion, blindness, and other visual and neurological disturbances. Brain imaging, preferably MRI (Magnetic Resonance Imaging), is used to confirm the diagnosis. In patients developing PRES, discontinue Bortezomib for Injection. The safety of reinitiating Bortezomib for Injection therapy in patients previously experiencing PRES is not known.
5.6 Gastrointestinal Toxicity
Bortezomib treatment can cause nausea, diarrhea, constipation, and vomiting [see Adverse Reactions ( 6.1)] sometimes requiring use of antiemetic and antidiarrheal medications. Ileus can occur. Fluid and electrolyte replacement should be administered to prevent dehydration. Interrupt Bortezomib for Injection for severe symptoms.
5.7 Thrombocytopenia/Neutropenia
Bortezomib is associated with thrombocytopenia and neutropenia that follow a cyclical pattern with nadirs occurring following the last dose of each cycle and typically recovering prior to initiation of the subsequent cycle. The cyclical pattern of platelet and neutrophil decreases and recovery remain consistent in the studies of multiple myeloma and mantle cell lymphoma, with no evidence of cumulative thrombocytopenia or neutropenia in the treatment regimens studied.
Monitor complete blood counts (CBC) frequently during treatment with Bortezomib for Injection. Measure platelet counts prior to each dose of Bortezomib for Injection. Adjust dose/schedule for thrombocytopenia [see Table 2 and Dosage and Administration ( 2.4)] . Gastrointestinal and intracerebral hemorrhage has occurred during thrombocytopenia in association with Bortezomib. Support with transfusions and supportive care, according to published guidelines.
In the single-agent, relapsed multiple myeloma study of bortezomib versus dexamethasone, the mean platelet count nadir measured was approximately 40% of baseline. The severity of thrombocytopenia related to pretreatment platelet count is shown in Table 6. The incidence of bleeding (≥ Grade 3) was 2% on the bortezomib arm and was < 1% in the dexamethasone arm.
Table 6: Severity of Thrombocytopenia Related to Pretreatment Platelet Count in the Relapsed Multiple Myeloma Study of Bortezomib versus Dexamethasone *A baseline platelet count of 50,000/μL was required for study eligibility
**Data were missing at baseline for 1 patient
Pretreatment
Platelet Count*
Number of Patients (N=331)**Number (%) of Patients with Platelet Count
< 10,000/μLNumber (%) of Patients with Platelet Count 10,000 to 25,000/μL ≥ 75,000/μL 309 8 (3%) 36 (12%) ≥ 50,000/μL to < 75,000/μL 14 2 (14%) 11 (79%) ≥ 10,000/μL to < 50,000/μL 7 1 (14%) 5 (71%) 5.8 Tumor Lysis Syndrome
Tumor lysis syndrome has been reported with bortezomib therapy. Patients at risk of tumor lysis syndrome are those with high tumor burden prior to treatment. Monitor patients closely and take appropriate precautions.
5.9 Hepatic Toxicity
Cases of acute liver failure have been reported in patients receiving multiple concomitant medications and with serious underlying medical conditions. Other reported hepatic reactions include hepatitis, increases in liver enzymes, and hyperbilirubinemia. Interrupt Bortezomib for injection therapy to assess reversibility. There is limited rechallenge information in these patients.
5.10 Embryo-fetal Toxicity
Based on the mechanism of action and findings in animals, bortezomib can cause fetal harm when administered to a pregnant woman. Bortezomib administered to rabbits during organogenesis at a dose approximately 0.5 times the clinical dose of 1.3 mg/m 2 based on body surface area caused post-implantation loss and a decreased number of live fetuses [see Use in Specific Populations ( 8.1)].
Females of reproductive potential should avoid becoming pregnant while being treated with Bortezomib for Injection. Advise females of reproductive potential that they must use contraception during treatment with Bortezomib for Injection and for 7 months following cessation of therapy. Advise males with female sexual partners of reproductive potential that they must use contraception during treatment with Bortezomib for Injection and for 4 months following cessation of therapy. If Bortezomib for Injection is used during pregnancy or if the patient becomes pregnant during Bortezomib for Injection treatment, the patient should be apprised of the potential risk to the fetus [see Use in Specific Populations ( 8.1, 8.3) and Nonclinical Toxicology ( 13.1)].
-
6 ADVERSE REACTIONS
The following adverse reactions are also discussed in other sections of the labeling:
- Peripheral Neuropathy [see Warnings and Precautions ( 5.1)]
- Hypotension [see Warnings and Precautions ( 5.2)]
- Cardiac Toxicity [see Warnings and Precautions ( 5.3)]
- Pulmonary Toxicity [see Warnings and Precautions ( 5.4)]
- Posterior Reversible Encephalopathy Syndrome (PRES) [see Warnings and Precautions ( 5.5)]
- Gastrointestinal Toxicity [see Warnings and Precautions ( 5.6)]
- Thrombocytopenia/Neutropenia [see Warnings and Precautions ( 5.7)]
- Tumor Lysis Syndrome [see Warnings and Precautions ( 5.8)]
- Hepatic Toxicity [see Warnings and Precautions ( 5.9)]
6.1 Clinical Trials Safety Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
Summary of Clinical Trial in Patients with Previously Untreated Multiple Myeloma
Table 7 describes safety data from 340 patients with previously untreated multiple myeloma who received bortezomib (1.3 mg/m 2) administered intravenously in combination with melphalan (9 mg/m 2) and prednisone (60 mg/m 2) in a prospective randomized study.
The safety profile of bortezomib in combination with melphalan/prednisone is consistent with the known safety profiles of both bortezomib and melphalan/prednisone.
Table 7: Most Commonly Reported Adverse Reactions (≥ 10% in the Bortezomib, Melphalan and Prednisone arm) with Grades 3 and ≥ 4 Intensity in the Previously Untreated Multiple Myeloma Study a Represents High Level Term Peripheral Neuropathies NEC
Bortezomib, Melphalan and
Prednisone
(n=340)Melphalan and Prednisone
(n=337)Toxicity Grade, n (%) Toxicity Grade, n (%) System Organ Class
Preferred Term
Total n (%) 3 ≥ 4 Total n (%) 3 ≥ 4 Blood and lymphatic system disorders Thrombocytopenia 164 (48) 60 (18) 57 (17) 140 (42) 48 (14) 39 (12) Neutropenia 160 (47) 101 (30) 33 (10) 143 (42) 77 (23) 42 (12) Anemia 109 (32) 41 (12) 4 (1) 156 (46) 61 (18) 18 (5) Leukopenia 108 (32) 64 (19) 8 (2) 93 (28) 53 (16) 11 (3) Lymphopenia 78 (23) 46 (14) 17 (5) 51 (15) 26 (8) 7 (2) Gastrointestinal disorders Nausea 134 (39) 10 (3) 0 70 (21) 1 (< 1) 0 Diarrhea 119 (35) 19 (6) 2 (1) 20 (6) 1 (< 1) 0 Vomiting 87 (26) 13 (4) 0 41 (12) 2 (1) 0 Constipation 77 (23) 2 (1) 0 14 (4) 0 0 Abdominal Pain Upper 34 (10) 1 (< 1) 0 20 (6) 0 0 Nervous system disorders Peripheral Neuropathy a 156 (46) 42 (12) 2 (1) 4 (1) 0 0 Neuralgia 117 (34) 27 (8) 2 (1) 1 (< 1) 0 0 Paresthesia 42 (12) 6 (2) 0 4 (1) 0 0 General disorders and administration site conditions Fatigue 85 (25) 19 (6) 2 (1) 48 (14) 4 (1) 0 Asthenia 54 (16) 18 (5) 0 23 (7) 3 (1) 0 Pyrexia 53 (16) 4 (1) 0 19 (6) 1 (< 1) 1 (< 1) Infections and infestations Herpes Zoster 39 (11) 11 (3) 0 9 (3) 4 (1) 0 Metabolism and nutrition disorders Anorexia 64 (19) 6 (2) 0 19 (6) 0 0 Skin and subcutaneous tissue disorders Rash 38 (11) 2 (1) 0 7 (2) 0 0 Psychiatric disorders Insomnia 35 (10) 1 (< 1) 0 21 (6) 0 0 Relapsed Multiple Myeloma Randomized Study of Bortezomib versus Dexamethasone
The safety data described below and in Table 8 reflect exposure to either bortezomib (n=331) or dexamethasone (n=332) in a study of patients with relapsed multiple myeloma. Bortezomib was administered intravenously at doses of 1.3 mg/m 2 twice weekly for 2 out of 3 weeks (21-day cycle). After eight 21-day cycles patients continued therapy for three 35-day cycles on a weekly schedule. Duration of treatment was up to 11 cycles (9 months) with a median duration of 6 cycles (4.1 months). For inclusion in the trial, patients must have had measurable disease and 1 to 3 prior therapies. There was no upper age limit for entry. Creatinine clearance could be as low as 20 mL/min and bilirubin levels as high as 1.5 times the upper limit of normal. The overall frequency of adverse reactions was similar in men and women, and in patients < 65 and ≥ 65 years of age. Most patients were Caucasian [see Clinical Studies ( 14.1)] .
Among the 331 bortezomib-treated patients, the most commonly reported (> 20%) adverse reactions overall were nausea (52%), diarrhea (52%), fatigue (39%), peripheral neuropathies (35%), thrombocytopenia (33%), constipation (30%), vomiting (29%), and anorexia (21%). The most commonly reported (> 20%) adverse reaction reported among the 332 patients in the dexamethasone group was fatigue (25%). Eight percent (8%) of patients in the bortezomib-treated arm experienced a Grade 4 adverse reaction; the most common reactions were thrombocytopenia (4%) and neutropenia (2%). Nine percent (9%) of dexamethasone-treated patients experienced a Grade 4 adverse reaction. All individual dexamethasone-related Grade 4 adverse reactions were less than 1%.
Serious Adverse Reactions and Adverse Reactions Leading to Treatment Discontinuation in the Relapsed Multiple Myeloma Study of Bortezomib versus Dexamethasone
Serious adverse reactions are defined as any reaction that results in death, is life-threatening, requires
hospitalization or prolongs a current hospitalization, results in a significant disability, or is deemed to be an important medical event. A total of 80 (24%) patients from the bortezomib treatment arm experienced a serious adverse reaction during the study, as did 83 (25%) dexamethasone-treated patients. The most commonly reported serious adverse reactions in the bortezomib treatment arm were diarrhea (3%), dehydration, herpes zoster, pyrexia, nausea, vomiting, dyspnea, and thrombocytopenia (2% each). In the dexamethasone treatment group, the most commonly reported serious adverse reactions were pneumonia (4%), hyperglycemia (3%), pyrexia, and psychotic disorder (2% each).
A total of 145 patients, including 84 (25%) of 331 patients in the bortezomib treatment group and 61 (18%) of
332 patients in the dexamethasone treatment group were discontinued from treatment due to adverse reactions. Among the 331 bortezomib-treated patients, the most commonly reported adverse reaction leading to discontinuation was peripheral neuropathy (8%). Among the 332 patients in the dexamethasone group, the most commonly reported adverse reactions leading to treatment discontinuation were psychotic disorder and hyperglycemia (2% each).
Four deaths were considered to be bortezomib-related in this relapsed multiple myeloma study: 1 case each of cardiogenic shock, respiratory insufficiency, congestive heart failure and cardiac arrest. Four deaths were considered dexamethasone-related: 2 cases of sepsis, 1 case of bacterial meningitis, and 1 case of sudden death at home.
Most Commonly Reported Adverse Reactions in the Relapsed Multiple Myeloma Study of Bortezomib versus Dexamethasone
The most common adverse reactions from the relapsed multiple myeloma study are shown in Table 8. All adverse reactions with incidence ≥ 10% in the bortezomib arm are included.
Table 8: Most Commonly Reported Adverse Reactions (≥ 10% in Bortezomib arm), with Grades 3 and 4 Intensity in the Relapsed Multiple Myeloma Study of Bortezomib versus Dexamethasone (N=663) a Represents High Level Term Peripheral Neuropathies NEC
Preferred Term
All
Bortezomib
(N=331)
Grade 3
Grade 4
All
Dexamethasone
(N=332)
Grade 3
Grade 4Adverse Reactions 324 (98) 193 (58) 28 (8) 297 (89) 110 (33) 29 (9) Nausea 172 (52) 8 (2) 0 31 (9) 0 0 Diarrhea NOS 171 (52) 22 (7) 0 36 (11) 2 (< 1) 0 Fatigue 130 (39) 15 (5) 0 82 (25) 8 (2) 0 Peripheral neuropathies a 115 (35) 23 (7) 2 (< 1) 14 (4) 0 1 (< 1) Thrombocytopenia 109 (33) 80 (24) 12 (4) 11 (3) 5 (2) 1 (< 1) Constipation 99 (30) 6 (2) 0 27 (8) 1 (< 1) 0 Vomiting NOS 96 (29) 8 (2) 0 10 (3) 1 (< 1) 0 Anorexia 68 (21) 8 (2) 0 8 (2) 1 (< 1) 0 Pyrexia 66 (20) 2 (< 1) 0 21 (6) 3 (< 1) 1 (< 1) Paresthesia 64 (19) 5 (2) 0 24 (7) 0 0 Anemia NOS 63 (19) 20 (6) 1 (< 1) 21 (6) 8 (2) 0 Headache NOS 62 (19) 3 (< 1) 0 23 (7) 1 (< 1) 0 Neutropenia 58 (18) 37 (11) 8 (2) 1 (< 1) 1 (< 1) 0 Rash NOS 43 (13) 3 (< 1) 0 7 (2) 0 0 Appetite decreased NOS 36 (11) 0 0 12 (4) 0 0 Dyspnea NOS 35 (11) 11 (3) 1 (< 1) 37 (11) 7 (2) 1 (< 1) Abdominal pain NOS 35 (11) 5 (2) 0 7 (2) 0 0 Weakness 34 (10) 10 (3) 0 28 (8) 8 (2) 0 Safety Experience from the Phase 2 Open-Label Extension Study in Relapsed Multiple Myeloma
In the phase 2 extension study of 63 patients, no new cumulative or new long-term toxicities were observed with prolonged bortezomib treatment. These patients were treated for a total of 5.3 to 23 months, including time on bortezomib in the prior bortezomib study [see Clinical Studies ( 14.1)] .
Integrated Summary of Safety (Relapsed Multiple Myeloma and Relapsed Mantle Cell Lymphoma)
Safety data from phase 2 and 3 studies of single agent bortezomib 1.3 mg/m 2/dose twice weekly for 2 weeks followed by a 10-day rest period in 1,163 patients with previously-treated multiple myeloma (N=1,008) and previously-treated mantle cell lymphoma (N=155) were integrated and tabulated. This analysis does not include data from the Phase 3 Open-Label Study of bortezomib subcutaneous versus intravenous in relapsed multiple myeloma. In the integrated studies, the safety profile of bortezomib was similar in patients with multiple myeloma and mantle cell lymphoma.
In the integrated analysis, the most commonly reported (> 20%) adverse reactions were nausea (49%), diarrhea (46%), asthenic conditions including fatigue (41%) and weakness (11%), peripheral neuropathies (38%), thrombocytopenia (32%), vomiting (28%), constipation (25%), and pyrexia (21%). Eleven percent (11%) of patients experienced at least 1 episode of ≥ Grade 4 toxicity, most commonly thrombocytopenia (4%) and neutropenia (2%).
In the Phase 2 relapsed multiple myeloma clinical trials of bortezomib administered intravenously, local skin irritation was reported in 5% of patients, but extravasation of bortezomib was not associated with tissue damage.
Serious Adverse Reactions and Adverse Reactions Leading to Treatment Discontinuation in the Integrated Summary of Safety
A total of 26% of patients experienced a serious adverse reaction during the studies. The most commonly reported serious adverse reactions included diarrhea, vomiting and pyrexia (3% each), nausea, dehydration, and thrombocytopenia (2% each) and pneumonia, dyspnea, peripheral neuropathies, and herpes zoster (1% each).
Adverse reactions leading to discontinuation occurred in 22% of patients. The reasons for discontinuation included peripheral neuropathy (8%), and fatigue, thrombocytopenia, and diarrhea (2% each).
In total, 2% of the patients died and the cause of death was considered by the investigator to be possibly related to study drug: including reports of cardiac arrest, congestive heart failure, respiratory failure, renal failure, pneumonia and sepsis.
Most Commonly Reported Adverse Reactions in the Integrated Summary of Safety
The most common adverse reactions are shown in Table 9. All adverse reactions occurring at ≥ 10% are included. In the absence of a randomized comparator arm, it is often not possible to distinguish between adverse events that are drug-caused and those that reflect the patient's underlying disease. Please see the discussion of specific adverse reactions that follows.
Table 9: Most Commonly Reported (≥ 10% Overall) Adverse Reactions in Integrated Analyses of Relapsed Multiple Myeloma and Relapsed Mantle Cell Lymphoma Studies using the 1.3 mg/m 2 Dose (N=1,163) a Represents High Level Term Peripheral Neuropathies NEC
All Patients Multiple Myeloma Mantle Cell Lymphoma (N=1,163)
(N=1,008) (N=155) Preferred Term All ≥ Grade 3 All ≥ Grade 3 All ≥ Grade 3 Nausea 567 (49) 36 (3) 511 (51) 32 (3) 56 (36) 4 (3) Diarrhea NOS 530 (46) 83 (7) 470 (47) 72 (7) 60 (39) 11 (7) Fatigue 477 (41) 86 (7) 396 (39) 71 (7) 81 (52) 15 (10) Peripheral neuropathies a 443 (38) 129 (11) 359 (36) 110 (11) 84 (54) 19 (12) Thrombocytopenia 369 (32) 295 (25) 344 (34) 283 (28) 25 (16) 12 (8) Vomiting NOS 321 (28) 44 (4) 286 (28) 40 (4) 35 (23) 4 (3) Constipation 296 (25) 17 (1) 244 (24) 14 (1) 52 (34) 3 (2) Pyrexia 249 (21) 16 (1) 233 (23) 15 (1) 16 (10) 1 (< 1) Anorexia 227 (20) 19 (2) 205 (20) 16 (2) 22 (14) 3 (2) Anemia NOS 209 (18) 65 (6) 190 (19) 63 (6) 19 (12) 2 (1) Headache NOS 175 (15) 8 (< 1) 160 (16) 8 (< 1) 15 (10) 0 Neutropenia 172 (15) 121 (10) 164 (16) 117 (12) 8 (5) 4 (3) Rash NOS 156 (13) 8 (< 1) 120 (12) 4 (< 1) 36 (23) 4 (3) Paresthesia 147 (13) 9 (< 1) 136 (13) 8 (< 1) 11 (7) 1 (< 1) Dizziness (excl vertigo) 129 (11) 13 (1) 101 (10) 9 (< 1) 28 (18) 4 (3) Weakness 124 (11) 31 (3) 106 (11) 28 (3) 18 (12) 3 (2) Description of Selected Adverse Reactions from the Integrated Phase 2 and 3 Relapsed Multiple Myeloma and Phase 2 Relapsed Mantle Cell Lymphoma Studies
Gastrointestinal Toxicity
A total of 75% of patients experienced at least one gastrointestinal disorder. The most common gastrointestinal disorders included nausea, diarrhea, constipation, vomiting, and appetite decreased. Other gastrointestinal disorders included dyspepsia and dysgeusia. Grade 3 adverse reactions occurred in 14% of patients; ≥ Grade 4 adverse reactions were ≤ 1%. Gastrointestinal adverse reactions were considered serious in 7% of patients. Four percent (4%) of patients discontinued due to a gastrointestinal adverse reaction. Nausea was reported more often in patients with multiple myeloma (51%) compared to patients with mantle cell lymphoma (36%).
Thrombocytopenia
Across the studies, bortezomib-associated thrombocytopenia was characterized by a decrease in platelet count during the dosing period (days 1 to 11) and a return toward baseline during the 10-day rest period during each treatment cycle. Overall, thrombocytopenia was reported in 32% of patients. Thrombocytopenia was Grade 3 in 22%, ≥ Grade 4 in 4%, and serious in 2% of patients, and the reaction resulted in bortezomib discontinuation in 2% of patients [see Warnings and Precautions ( 5.7)] . Thrombocytopenia was reported more often in patients with multiple myeloma (34%) compared to patients with mantle cell lymphoma (16%). The incidence of ≥ Grade 3 thrombocytopenia also was higher in patients with multiple myeloma (28%) compared to patients with mantle cell lymphoma (8%).
Peripheral Neuropathy
Overall, peripheral neuropathies occurred in 38% of patients. Peripheral neuropathy was Grade 3 for 11% of patients and ≥ Grade 4 for < 1% of patients. Eight percent (8%) of patients discontinued bortezomib due to peripheral neuropathy. The incidence of peripheral neuropathy was higher among patients with mantle cell lymphoma (54%) compared to patients with multiple myeloma (36%).
In the bortezomib versus dexamethasone phase 3 relapsed multiple myeloma study, among the 62 bortezomib-treated patients who experienced ≥ Grade 2 peripheral neuropathy and had dose adjustments, 48% had improved or resolved with a median of 3.8 months from first onset.
In the phase 2 relapsed multiple myeloma studies, among the 30 patients who experienced Grade 2 peripheral neuropathy resulting in discontinuation or who experienced ≥ Grade 3 peripheral neuropathy, 73% reported improvement or resolution with a median time of 47 days to improvement of one Grade or more from the last dose of bortezomib.
Hypotension
The incidence of hypotension (postural, orthostatic and hypotension NOS) was 8% in patients treated with bortezomib. Hypotension was Grade 1 or 2 in the majority of patients and Grade 3 in 2% and ≥ Grade 4 in
< 1%. Two percent (2%) of patients had hypotension reported as a serious adverse reaction, and 1% discontinued due to hypotension. The incidence of hypotension was similar in patients with multiple myeloma (8%) and those with mantle cell lymphoma (9%). In addition, < 1% of patients experienced hypotension associated with a syncopal reaction.
Neutropenia
Neutrophil counts decreased during the bortezomib dosing period (days 1 to 11) and returned toward baseline during the 10-day rest period during each treatment cycle. Overall, neutropenia occurred in 15% of patients and was Grade 3 in 8% of patients and ≥ Grade 4 in 2%. Neutropenia was reported as a serious adverse reaction in < 1% of patients and < 1% of patients discontinued due to neutropenia. The incidence of neutropenia was higher in patients with multiple myeloma (16%) compared to patients with mantle cell lymphoma (5%). The incidence of ≥ Grade 3 neutropenia also was higher in patients with multiple myeloma (12%) compared to patients with mantle cell lymphoma (3%).
Asthenic conditions (Fatigue, Malaise, Weakness, Asthenia)
Asthenic conditions were reported in 54% of patients. Fatigue was reported as Grade 3 in 7% and ≥ Grade 4 in < 1% of patients. Asthenia was reported as Grade 3 in 2% and ≥ Grade 4 in < 1% of patients. Two percent (2%) of patients discontinued treatment due to fatigue and < 1% due to weakness and asthenia. Asthenic conditions were reported in 53% of patients with multiple myeloma and 59% of patients with mantle cell lymphoma.
Pyrexia
Pyrexia (> 38ºC) was reported as an adverse reaction for 21% of patients. The reaction was Grade 3 in 1% and ≥ Grade 4 in < 1%. Pyrexia was reported as a serious adverse reaction in 3% of patients and led to bortezomib discontinuation in < 1% of patients. The incidence of pyrexia was higher among patients with multiple myeloma (23%) compared to patients with mantle cell lymphoma (10%). The incidence of ≥ Grade 3 pyrexia was 1% in patients with multiple myeloma and < 1% in patients with mantle cell lymphoma.
Herpes Virus Infection
Consider using antiviral prophylaxis in subjects being treated with Bortezomib for Injection. In the randomized studies in previously untreated and relapsed multiple myeloma, herpes zoster reactivation was more common in subjects treated with bortezomib (ranging between 6 to 11%) than in the control groups (3 to 4%). Herpes simplex was seen in 1 to 3% in subjects treated with bortezomib and 1 to 3% in the control groups. In the previously untreated multiple myeloma study, herpes zoster virus reactivation in the bortezomib, melphalan and prednisone arm was less common in subjects receiving prophylactic antiviral therapy (3%) than in subjects who did not receive prophylactic antiviral therapy (17%).
Additional Adverse Reactions from Clinical Studies
The following clinically important serious adverse reactions that are not described above have been reported in clinical trials in patients treated with bortezomib administered as monotherapy or in combination with other chemotherapeutics. These studies were conducted in patients with hematological malignancies and in solid tumors.
Blood and lymphatic system disorders: Anemia, disseminated intravascular coagulation, febrile neutropenia, lymphopenia, leukopenia
Cardiac disorders: Angina pectoris, atrial fibrillation aggravated, atrial flutter, bradycardia, sinus arrest, cardiac amyloidosis, complete atrioventricular block, myocardial ischemia, myocardial infarction, pericarditis, pericardial effusion, Torsades de pointes, ventricular tachycardia
Ear and labyrinth disorders: Hearing impaired, vertigo
Eye disorders: Diplopia and blurred vision, conjunctival infection, irritation
Gastrointestinal disorders: Abdominal pain, ascites, dysphagia, fecal impaction, gastroenteritis, gastritis hemorrhagic, hematemesis, hemorrhagic duodenitis, ileus paralytic, large intestinal obstruction, paralytic intestinal obstruction, peritonitis, small intestinal obstruction, large intestinal perforation, stomatitis, melena, pancreatitis acute, oral mucosal petechiae, gastroesophageal reflux
General disorders and administration site conditions: Chills, edema, edema peripheral, injection site erythema, neuralgia, injection site pain, irritation, malaise, phlebitis
Hepatobiliary disorders: Cholestasis, hepatic hemorrhage, hyperbilirubinemia, portal vein thrombosis, hepatitis, liver failure
Immune system disorders: Anaphylactic reaction, drug hypersensitivity, immune complex mediated hypersensitivity, angioedema, laryngeal edema
Infections and infestations: Aspergillosis, bacteremia, bronchitis, urinary tract infection, herpes viral infection, listeriosis, nasopharyngitis, pneumonia, respiratory tract infection, septic shock, toxoplasmosis, oral candidiasis, sinusitis, catheter-related infection
Injury, poisoning and procedural complications: Catheter-related complication, skeletal fracture, subdural hematoma
Investigations: Weight decreased
Metabolism and nutrition disorders: Dehydration, hypocalcemia, hyperuricemia, hypokalemia, hyperkalemia, hyponatremia, hypernatremia
Musculoskeletal and connective tissue disorders: Arthralgia, back pain, bone pain, myalgia, pain in extremity
Nervous system disorders: Ataxia, coma, dizziness, dysarthria, dysesthesia, dysautonomia, encephalopathy, cranial palsy, grand mal convulsion, headache, hemorrhagic stroke, motor dysfunction, neuralgia, spinal cord compression, paralysis, postherpetic neuralgia, transient ischemic attack
Psychiatric disorders: Agitation, anxiety, confusion, insomnia, mental status change, psychotic disorder, suicidal ideation
Renal and urinary disorders: Calculus renal, bilateral hydronephrosis, bladder spasm, hematuria, hemorrhagic cystitis, urinary incontinence, urinary retention, renal failure (acute and chronic), glomerular nephritis proliferative
Respiratory, thoracic and mediastinal disorders: Acute respiratory distress syndrome, aspiration pneumonia, atelectasis, chronic obstructive airways disease exacerbated, cough, dysphagia, dyspnea, dyspnea exertional, epistaxis, hemoptysis, hypoxia, lung infiltration, pleural effusion, pneumonitis, respiratory distress, pulmonary hypertension
Skin and subcutaneous tissue disorders: Urticaria, face edema, rash (which may be pruritic), leukocytoclastic vasculitis, pruritus
Vascular disorders: Cerebrovascular accident, cerebral hemorrhage, deep venous thrombosis, hypertension, peripheral embolism, pulmonary embolism, pulmonary hypertension
6.2 Postmarketing Experience
The following adverse reactions have been identified from the worldwide postmarketing experience with bortezomib. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure:
Cardiac disorders: Cardiac tamponade
Ear and labyrinth disorders: Deafness bilateral
Eye disorders: Optic neuropathy, blindness Gastrointestinal disorders: Ischemic colitis
Infections and infestations: Progressive multifocal leukoencephalopathy (PML), ophthalmic herpes, herpes meningoencephalitis
Nervous system disorders: Posterior reversible encephalopathy syndrome (PRES, formerly RPLS)
Respiratory, thoracic and mediastinal disorders: Acute diffuse infiltrative pulmonary disease
Skin and subcutaneous tissue disorders: Stevens-Johnson syndrome/toxic epidermal necrolysis (SJS/TEN), acute febrile neutrophilic dermatosis (Sweet's syndrome)
-
7 DRUG INTERACTIONS
7.1 Effect of Strong CYP3A4 Inhibitors on Bortezomib
Monitor patients for signs of bortezomib toxicity and consider a Bortezomib for Injection dose reduction if Bortezomib for Injection must be given with strong CYP3A4 inhibitors.
7.3 Effect of Strong CYP3A4 Inducers on Bortezomib
Avoid strong CYP3A4 inducers. The coadministration of a strong CYP3A4 inducer is expected to decrease the exposure of Bortezomib for Injection. Efficacy may be reduced when Bortezomib for Injection is coadministered with strong CYP3A4 inducers.
Avoid St. John's Wort ( Hypericum perforatum), as it may decrease Bortezomib exposure unpredictably.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Based on its mechanism of action [see Clinical Pharmacology ( 12.1)] and findings in animals, Bortezomib for Injection can cause fetal harm when administered to a pregnant woman. There are no studies with the use of bortezomib in pregnant women to inform drug-associated risks. Bortezomib caused embryo-fetal lethality in rabbits at doses lower than the clinical dose [see Data] . Advise pregnant women of the potential risk to the fetus.
Adverse outcomes in pregnancy occur regardless of the health of the mother or the use of medications. The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Data
Animal Data
Bortezomib was not teratogenic in nonclinical developmental toxicity studies in rats and rabbits at the highest dose tested (0.075 mg/kg; 0.5 mg/m 2 in the rat and 0.05 mg/kg; 0.6 mg/m 2 in the rabbit) when administered during organogenesis. These dosages are approximately 0.5 times the clinical dose of 1.3 mg/m 2 based on body surface area.
Bortezomib caused embryo-fetal lethality in rabbits at doses lower than the clinical dose (approximately
0.5 times the clinical dose of 1.3 mg/m 2 based on body surface area). Pregnant rabbits given bortezomib during organogenesis at a dose of 0.05 mg/kg (0.6 mg/m 2) experienced significant post-implantation loss and decreased number of live fetuses. Live fetuses from these litters also showed significant decreases in fetal weight.
8.2 Lactation
Risk Summary
There are no data on the presence of bortezomib or its metabolites in human milk, the effects of the drug on the breastfed child, or the effects of the drug on milk production. Because many drugs are excreted in human milk and because the potential for serious adverse reactions in a breastfed child from Bortezomib for Injection is unknown, advise nursing women not to breastfeed during treatment with Bortezomib for Injection and for 2 months after treatment.
8.3 Females and Males of Reproductive Potential
Based on its mechanism of action and findings in animals, bortezomib can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations ( 8.1)].
Pregnancy Testing
Verify the pregnancy status of females of reproductive potential prior to initiating Bortezomib for Injection treatment.
Contraception
Females
Advise females of reproductive potential to avoid pregnancy during treatment with Bortezomib for Injection and for 7 months following cessation of therapy.
Males
Males with female sexual partners of reproductive potential should use effective contraception during treatment with Bortezomib for Injection and for 4 months following cessation of therapy.
Infertility
Based on the mechanism of action and findings in animals, Bortezomib for Injection may have an effect on either male or female fertility [see Nonclinical Toxicology ( 13.1)] .
8.4 Pediatric Use
Additional information describing a clinical study in which efficacy was not demonstrated in pediatric patients is in the approved label for Millennium Pharmaceuticals, Inc.'s VELCADE (bortezomib) Injection. However, due to Millennium Pharmaceuticals, Inc.'s marketing exclusivity rights, this drug product is not labeled with that pediatric information.
8.5 Geriatric Use
Of the 669 patients enrolled in the relapsed multiple myeloma study, 245 (37%) were 65 years of age or older: 125 (38%) on the bortezomib arm and 120 (36%) on the dexamethasone arm. Median time to progression and median duration of response for patients ≥ 65 were longer on bortezomib compared to dexamethasone [5.5 mo versus 4.3 mo, and 8 mo versus 4.9 mo, respectively]. On the bortezomib arm, 40% (n=46) of evaluable patients aged ≥ 65 experienced response (CR+PR) versus 18% (n=21) on the dexamethasone arm. The incidence of Grade 3 and 4 events was 64%, 78% and 75% for bortezomib patients ≤ 50, 51 to 64 and ≥ 65 years old, respectively [see Adverse Reactions ( 6.1) and Clinical Studies ( 14.1)] .
No overall differences in safety or effectiveness were observed between patients ≥ age 65 and younger patients receiving bortezomib; but greater sensitivity of some older individuals cannot be ruled out.
8.6 Patients with Renal Impairment
The pharmacokinetics of bortezomib are not influenced by the degree of renal impairment. Dosing adjustments of Bortezomib for Injection are not necessary for patients with renal insufficiency. Since dialysis may reduce bortezomib concentrations, Bortezomib for Injection should be administered after the dialysis procedure [see Clinical Pharmacology ( 12.3)] .
8.7 Patients with Hepatic Impairment
Reduce the starting dose in patients with moderate (bilirubin greater than 1.5 to 3 times upper limit of normal (ULN) and any AST) and severe (bilirubin greater than 3 times ULN and any AST) hepatic impairment [see Dosage and Administration ( 2.6), Clinical Pharmacology ( 12.3)].
8.8 Patients with Diabetes
During clinical trials, hypoglycemia and hyperglycemia were reported in diabetic patients receiving oral hypoglycemics. Patients on oral antidiabetic agents receiving Bortezomib for Injection treatment may require close monitoring of their blood glucose levels and adjustment of the dose of their antidiabetic medication.
-
10 OVERDOSAGE
There is no known specific antidote for bortezomib overdosage. In humans, fatal outcomes following the administration of more than twice the recommended therapeutic dose have been reported, which were associated with the acute onset of symptomatic hypotension ( 5.2) and thrombocytopenia ( 5.7). In the event of an overdosage, the patient's vital signs should be monitored and appropriate supportive care given.
Studies in monkeys and dogs showed that intravenous bortezomib doses as low as 2 times the recommended clinical dose on a mg/m 2 basis were associated with increases in heart rate, decreases in contractility, hypotension, and death. In dog studies, a slight increase in the corrected QT interval was observed at doses resulting in death. In monkeys, doses of 3 mg/m 2 and greater (approximately twice the recommended clinical dose) resulted in hypotension starting at 1 hour post-administration, with progression to death in 12 to 14 hours following drug administration.
-
11 DESCRIPTION
Bortezomib for Injection is an antineoplastic agent available for intravenous injection. Each single-dose vial contains 3.5 mg of bortezomib, 10.5 mg boric acid, 25 mg glycine as a sterile lyophilized powder.
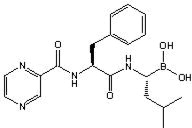
The chemical name for bortezomib, the monomeric boronic acid, is [(1R)-3-methyl-1- [[(2S)-1-oxo-3-phenyl-2-[(pyrazinylcarbonyl) amino]propyl]amino]butyl] boronic acid. Bortezomib has the following chemical structure:
The molecular weight of bortezomib is 384.24 and its molecular formula is C 19H 25BN 4O 4.
The solubility of bortezomib, as the monomeric boronic acid, in water is 3.3 to 3.8 mg/mL in a pH range of 2 to 6.5.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Bortezomib is a reversible inhibitor of the chymotrypsin-like activity of the 26S proteasome in mammalian cells. The 26S proteasome is a large protein complex that degrades ubiquitinated proteins. The ubiquitin proteasome pathway plays an essential role in regulating the intracellular concentration of specific proteins, thereby maintaining homeostasis within cells. Inhibition of the 26S proteasome prevents this targeted proteolysis, which can affect multiple signaling cascades within the cell. This disruption of normal homeostatic mechanisms can lead to cell death. Experiments have demonstrated that bortezomib is cytotoxic to a variety of cancer cell types in vitro. Bortezomib causes a delay in tumor growth in vivo in nonclinical tumor models, including multiple myeloma.
12.2 Pharmacodynamics
Following twice weekly administration of 1 mg/m 2 and 1.3 mg/m 2 bortezomib doses (n=12 per each dose level), the maximum inhibition of 20S proteasome activity (relative to baseline) in whole blood was observed 5 minutes after drug administration. Comparable maximum inhibition of 20S proteasome activity was observed between 1 and 1.3 mg/m 2 doses. Maximal inhibition ranged from 70% to 84% and from 73% to 83% for the 1 mg/m 2 and 1.3 mg/m 2 dose regimens, respectively.
12.3 Pharmacokinetics
Following intravenous administration of 1 mg/m 2 and 1.3 mg/m 2 doses, the mean maximum plasma concentrations of bortezomib (C max) after the first dose (Day 1) were 57 and 112 ng/mL, respectively. When administered twice weekly, the mean maximum observed plasma concentrations ranged from 67 to 106 ng/mL for the 1 mg/m 2 dose and 89 to 120 ng/mL for the 1.3 mg/m 2 dose.
Distribution:
The mean distribution volume of bortezomib ranged from approximately 498 to 1,884 L/m 2 following single- or multiple-dose administration of 1 mg/m 2 or 1.3 mg/m 2.
The binding of bortezomib to human plasma proteins averaged 83% over the concentration range of 100 to 1,000 ng/mL.
Elimination:
The mean elimination half-life of bortezomib after multiple dosing ranged from 40 hours to 193 hours after the 1 mg/m 2 dose and 76 hours to 108 hours after the 1.3 mg/m 2 dose. The mean total body clearances was 102 L/h and 112 L/h following the first dose for doses of 1 mg/m 2 and 1.3 mg/m 2, respectively, and ranged from 15 L/h to 32 L/h following subsequent doses for doses of 1 and 1.3 mg/m 2, respectively.
Metabolism: The major metabolic pathway is deboronation to form two deboronated metabolites that subsequently undergo hydroxylation to several metabolites. Deboronated bortezomib metabolites are inactive as 26S proteasome inhibitors. Pooled plasma data from 8 patients at 10 min and 30 min after dosing indicate that the plasma levels of metabolites are low compared to the parent drug.
In vitro studies indicate that bortezomib is primarily oxidatively metabolized via cytochrome P450 enzymes 3A4, 2C19, and 1A2.
Excretion: The pathways of elimination of bortezomib have not been characterized in humans.
Specific Populations:
Age: Analyses of data after the first dose of Cycle 1 (Day 1) in patients who had received intravenous doses of 1 mg/m 2 and 1.3 mg/m 2 showed that both dose-normalized AUC and C max tend to be less in younger patients. Patients less than 65 years of age had about 25% lower mean dose-normalized AUC and C max than those greater than or equal to 65 years of age .
Sex: Sex has no clinically important effect on bortezomib exposure.
Hepatic Impairment: Mild hepatic impairment had no clinically important effect on dose-normalized AUC or C max. The dose-normalized mean AUC was increased by approximately 60% in patients with moderate hepatic impairment (defined as total bilirubin greater than 1.5 to 3 times the upper limit of normal and any AST) or severe hepatic impairment (defined as total bilirubin greater than 3 times the upper limit of normal and any AST) [ see Dosage and Administration ( 2.6) and Use in Specific Populations ( 8.7) ].
Renal Impairment: Dose-normalized AUC and C max was comparable for patients with creatinine clearance (CLcr) from 59 mL/min/1.73 m 2 to less than 20 mL/min/1.73 m 2 compared to patients with CLcr greater than or equal to 60 mL/min/1.73 m 2 [ see Use in Specific Populations ( 8.6) ].
Effect of Other Drugs on Bortezomib: The coadministration of omeprazole, a strong inhibitor of CYP2C19, had no effect on the exposure of bortezomib.
The coadministration of ketoconazole, a strong CYP3A4 inhibitor, increased the exposure of bortezomib by 35%.
The coadministration of rifampin, a strong CYP3A4 inducer, is expected to decrease the exposure of bortezomib by at least 45%. Decreases greater than 45% may occur, as the drug interaction trial was not designed to evaluate the maximum effect of rifampin on bortezomib exposure.
Effect of Bortezomib on Other Drugs: Bortezomib inhibits CYP2C19 activity in vitro and the coadministration of Bortezomib for Injection with sensitive or narrow therapeutic CYP2C19 substrates may increase their exposure. Bortezomib did not inhibit CYP1A2, 2C9, 2D6, or 3A4 in vitro.
Bortezomib did not induce the CYP3A4 or 1A2 activity in vitro.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies have not been conducted with bortezomib.
Bortezomib showed clastogenic activity (structural chromosomal aberrations) in the in vitro chromosomal aberration assay using Chinese hamster ovary cells. Bortezomib was not genotoxic when tested in the in vitro mutagenicity assay (Ames test) and in vivo micronucleus assay in mice.
Fertility studies with bortezomib were not performed but evaluation of reproductive tissues has been performed in the general toxicity studies. In the 6-month rat toxicity study, degenerative effects in the ovary were observed at doses ≥ 0.3 mg/m 2 (one-fourth of the recommended clinical dose), and degenerative changes in the testes occurred at 1.2 mg/m 2.
13.2 Animal Toxicology and/or Pharmacology
Cardiovascular Toxicity: Studies in monkeys showed that administration of dosages approximately twice the recommended clinical dose resulted in heart rate elevations, followed by profound progressive hypotension, bradycardia, and death 12 to 14 hours post dose. Doses ≥ 1.2 mg/m 2 induced dose-proportional changes in cardiac parameters. Bortezomib has been shown to distribute to most tissues in the body, including the myocardium. In a repeated dosing toxicity study in the monkey, myocardial hemorrhage, inflammation, and necrosis were also observed.
Chronic Administration: In animal studies at a dose and schedule similar to that recommended for patients (twice weekly dosing for 2 weeks followed by 1-week rest), toxicities observed included severe anemia and thrombocytopenia, and gastrointestinal, neurological and lymphoid system toxicities. Neurotoxic effects of bortezomib in animal studies included axonal swelling and degeneration in peripheral nerves, dorsal spinal roots, and tracts of the spinal cord. Additionally, multifocal hemorrhage and necrosis in the brain, eye, and heart were observed.
-
14 CLINICAL STUDIES
14.1 Multiple Myeloma
Randomized, Open-Label Clinical Study in Patients with Previously Untreated Multiple Myeloma:
A prospective, international, randomized (1:1), open-label clinical study of 682 patients was conducted to determine whether bortezomib administered intravenously (1.3 mg/m 2) in combination with melphalan
(9 mg/m 2) and prednisone (60 mg/m 2) resulted in improvement in time to progression (TTP) when compared to melphalan (9 mg/m 2) and prednisone (60 mg/m 2) in patients with previously untreated multiple myeloma. Treatment was administered for a maximum of 9 cycles (approximately 54 weeks) and was discontinued early for disease progression or unacceptable toxicity. Antiviral prophylaxis was recommended for patients on the bortezomib study arm.
The median age of the patients in the study was 71 years (48;91), 50% were male, 88% were Caucasian and the median Karnofsky performance status score for the patients was 80 (60;100). Patients had IgG/IgA/Light chain myeloma in 63%/25%/8% instances, a median hemoglobin of 105 g/L (64;165), and a median platelet count of 221,500/microliter (33,000;587,000).
Efficacy results for the trial are presented in Table 10. At a pre-specified interim analysis (with median follow-up of 16.3 months), the combination of bortezomib, melphalan and prednisone therapy resulted in significantly superior results for time to progression, progression-free survival, overall survival and response rate. Further enrollment was halted, and patients receiving melphalan and prednisone were offered bortezomib in addition. A later, pre-specified analysis of overall survival (with median follow-up of 36.7 months with a hazard ratio of 0.65, 95% CI: 0.51, 0.84) resulted in a statistically significant survival benefit for the bortezomib, melphalan and prednisone treatment arm despite subsequent therapies including bortezomib based regimens. In an updated analysis of overall survival based on 387 deaths (median follow-up 60.1 months), the median overall survival for the bortezomib, melphalan and prednisone treatment arm was 56.4 months and for the melphalan and prednisone treatment arm was 43.1 months, with a hazard ratio of 0.695 (95% CI: 0.57, 0.85).
Table 10: Summary of Efficacy Analyses in the Previously Untreated Multiple Myeloma Study Note: All results are based on the analysis performed at a median follow-up duration of 16.3 months except for the overall survival analysis.
a Kaplan-Meier estimate
b Hazard ratio estimate is based on a Cox proportional-hazard model adjusted for stratification factors: beta2-microglobulin, albumin, and region. A hazard ratio less than 1 indicates an advantage for bortezomib, melphalan and prednisone
c p-value based on the stratified log-rank test adjusted for stratification factors: beta2-microglobulin, albumin, and region
d EBMT criteria
e p-value for Response Rate (CR + PR) from the Cochran-Mantel-Haenszel chi-square test adjusted for the stratification factors
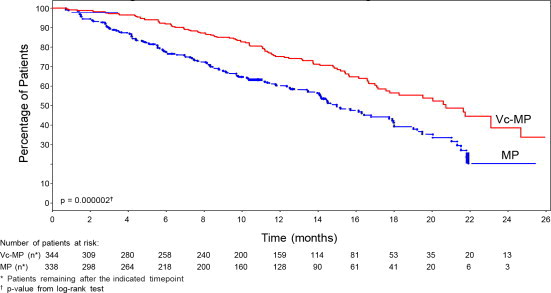
TTP was statistically significantly longer on the bortezomib, melphalan and prednisone arm (see Figure 1). (median follow-up 16.3 months)
Efficacy Endpoint Bortezomib,
Melphalan and
Prednisone
n=344Melphalan and
Prednisone
n=338Time to Progression Events n (%) 101 (29) 152 (45) Median a (months) 20.7 15 (95% CI) (17.6, 24.7) (14.1, 17.9) Hazard ratio b 0.54 (95% CI) (0.42, 0.70) p-value c 0.000002 Progression-free Survival Events n (%) 135 (39) 190 (56) Median a (months) 18.3 14 (95% CI) (16.6, 21.7) (11.1, 15) Hazard ratio b 0.61 (95% CI) (0.49, 0.76) p-value c 0.00001 Response Rate CR d n (%) 102 (30) 12 (4) PR d n (%) 136 (40) 103 (30) nCR n (%) 5 (1) 0 CR + PR d n (%) 238 (69) 115 (34) p-value e <10 -10 Overall Survival at median follow up of 36.7 months Events (deaths) n (%) 109 (32) 148 (44) Median a (months) Not Reached 43.1 (95% CI) (46.2, NR) (34.8, NR) Hazard ratio b 0.65 (95% CI) (0.51, 0.84) p-value c 0.00084 Figure 1: Time to Progression
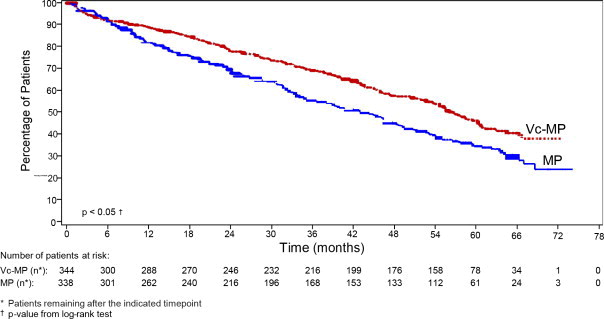
Bortezomib, Melphalan and Prednisone versus Melphalan and PrednisoneOverall survival was statistically significantly longer on the bortezomib, melphalan and prednisone arm (see Figure 2). (median follow-up 60.1 months)
Figure 2: Overall Survival
Bortezomib, Melphalan and Prednisone versus Melphalan and PrednisoneRandomized, Clinical Study in Relapsed Multiple Myeloma of Bortezomib versus Dexamethasone
A prospective phase 3, international, randomized (1:1), stratified, open-label clinical study enrolling 669
patients was designed to determine whether bortezomib resulted in improvement in time to progression (TTP) compared to high-dose dexamethasone in patients with progressive multiple myeloma following 1 to 3 prior therapies. Patients considered to be refractory to prior high-dose dexamethasone were excluded as were those with baseline Grade ≥ 2 peripheral neuropathy or platelet counts < 50,000/μL. A total of 627 patients were evaluable for response.
Stratification factors were based on the number of lines of prior therapy the patient had previously received (1 previous line versus more than 1 line of therapy), time of progression relative to prior treatment (progression during or within 6 months of stopping their most recent therapy versus relapse > 6 months after receiving their most recent therapy), and screening β 2-microglobulin levels (≤ 2.5 mg/L versus > 2.5 mg/L).
Baseline patient and disease characteristics are summarized in Table 11.
Table 11: Summary of Baseline Patient and Disease Characteristics in the Relapsed Multiple Myeloma Study
Patient Characteristics
Bortezomib N=333Dexamethasone
N=336Median age in years (range) 62 (33, 84) 61 (27, 86) Gender: Male/female 56% / 44% 60% / 40% Race: Caucasian/Black/other 90% / 6% / 4% 88% / 7% / 5% Karnofsky performance status score ≤ 70 13% 17% Hemoglobin <100 g/L 32% 28% Platelet count < 75 x 10 9/L 6% 4% Disease Characteristics Type of myeloma (%): IgG/IgA/Light chain
Median β 2-microglobulin (mg/L)
Median albumin (g/L)
Creatinine clearance ≤ 30 mL/min [n (%)]60% / 23% / 12%
3.7
39
17 (5%)59% / 24% / 13%
3.6
39
11 (3%)Median Duration of Multiple Myeloma Since
Diagnosis (Years)3.5 3.1 Number of Prior Therapeutic Lines of Treatment Median 2 2 1 prior line 40% 35% >1 prior line 60% 65% Previous Therapy Any prior steroids, e.g., dexamethasone, VAD 98% 99% Any prior anthracyclines, e.g., VAD, mitoxantrone 77% 76% Any prior alkylating agents, e.g., MP, VBMCP 91% 92% Any prior thalidomide therapy 48% 50% Vinca alkaloids 74% 72% Prior stem cell transplant/other high-dose therapy 67% 68% Prior experimental or other types of therapy 3% 2% Patients in the bortezomib treatment group were to receive eight 3-week treatment cycles followed by three 5-week treatment cycles of bortezomib. Patients achieving a CR were treated for 4 cycles beyond first evidence of CR. Within each 3-week treatment cycle, bortezomib 1.3 mg/m 2/dose alone was administered by intravenous bolus twice weekly for 2 weeks on Days 1, 4, 8, and 11 followed by a 10-day rest period (Days 12 to 21). Within each 5-week treatment cycle, bortezomib 1.3 mg/m 2/dose alone was administered by intravenous bolus once weekly for 4 weeks on Days 1, 8, 15, and 22 followed by a 13-day rest period (Days 23 to 35) [see Dosage and Administration ( 2.2)] .
Patients in the dexamethasone treatment group were to receive four 5-week treatment cycles followed by five 4-week treatment cycles. Within each 5-week treatment cycle, dexamethasone 40 mg/day PO was administered once daily on Days 1 to 4, 9 to 12, and 17 to 20 followed by a 15-day rest period (Days 21 to 35). Within each 4-week treatment cycle, dexamethasone 40 mg/day PO was administered once daily on Days 1 to 4 followed by a 24-day rest period (Days 5 to 28). Patients with documented progressive disease on dexamethasone were offered bortezomib at a standard dose and schedule on a companion study. Following a preplanned interim analysis of time to progression, the dexamethasone arm was halted and all patients randomized to dexamethasone were offered bortezomib, regardless of disease status.
In the bortezomib arm, 34% of patients received at least one bortezomib dose in all 8 of the
3-week cycles of therapy, and 13% received at least one dose in all 11 cycles. The average number of bortezomib doses during the study was 22, with a range of 1 to 44. In the dexamethasone arm, 40% of patients received at least one dose in all 4 of the 5-week treatment cycles of therapy, and 6% received at least one dose in all 9 cycles.
The time to event analyses and response rates from the relapsed multiple myeloma study are presented in Table 12. Response and progression were assessed using the European Group for Blood and Marrow Transplantation (EBMT) criteria. Complete response (CR) required < 5% plasma cells in the marrow, 100% reduction in M- protein, and a negative immunofixation test (IF -). Partial response (PR) requires ≥ 50% reduction in serum myeloma protein and ≥ 90% reduction of urine myeloma protein on at least 2 occasions for a minimum of at least 6 weeks along with stable bone disease and normal calcium. Near complete response (nCR) was defined as meeting all the criteria for complete response including 100% reduction in M-protein by protein electrophoresis; however, M-protein was still detectable by immunofixation (IF +).
Table 12: Summary of Efficacy Analyses in the Relapsed Multiple Myeloma Study a Kaplan-Meier estimate
b Hazard ratio is based on Cox proportional-hazard model with the treatment as single independent variable. A hazard ratio less than
1 indicates an advantage for bortezomib
c p-value based on the stratified log-rank test including randomization stratification factors
d Precise p-value cannot be rendered
e Response population includes patients who had measurable disease at baseline and received at least 1 dose of study drug
f EBMT criteria; nCR meets all EBMT criteria for CR but has positive IF. Under EBMT criteria nCR is in the PR category
g In 2 patients, the IF was unknown
h p-value for Response Rate (CR + PR) from the Cochran-Mantel-Haenszel chi-square test adjusted for the stratification factors
Efficacy Endpoint
All Patients1 Prior Line of
Therapy> 1 Prior Line of
TherapyBortezomib Dex Bortezomib Dex Bortezomib Dex n=333 n=336 n=132 n=119 n=200 n=217 Time to Progression
Events n (%)
147 (44)
196 (58)
55 (42)
64 (54)
92 (46)
132 (61)Median a
(95% CI)6.2 mo
(4.9, 6.9)3.5 mo
(2.9, 4.2)7 mo
(6.2, 8.8)5.6 mo
(3.4, 6.3)4.9 mo
(4.2, 6.3)2.9 mo
(2.8, 3.5)Hazard ratio b
(95% CI)0.55
(0.44, 0.69)0.55
(0.38, 0.81)0.54
(0.41, 0.72)p-value c <0.0001 0.0019 <0.0001 Overall Survival
Events (deaths) n (%)
51 (15)
84 (25)
12 (9)
24 (20)
39 (20)
60 (28)Hazard ratio b
(95% CI)0.57
(0.40, 0.81)0.39
(0.19, 0.81)0.65
(0.43, 0.97)p-value c,d < 0.05 < 0.05 < 0.05 Response Rate
Population e n = 627
n=315
n=312
n=128
n=110
n=187
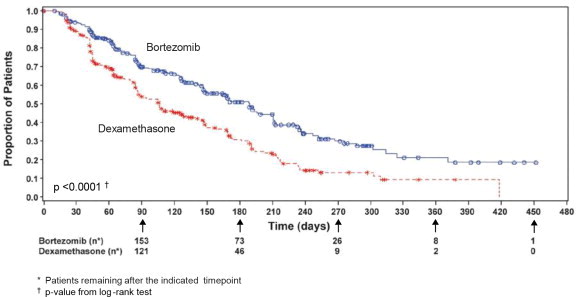
n=202CR f n (%) 20 (6) 2 (<1) 8 (6) 2 (2) 12 (6) 0 (0) PR f n(%) 101 (32) 54 (17) 49 (38) 27 (25) 52 (28) 27 (13) nCR f,g n(%) 21 (7) 3 (<1) 8 (6) 2 (2) 13 (7) 1 (<1) CR + PR f n (%) 121 (38) 56 (18) 57 (45) 29 (26) 64 (34) 27 (13) p-value h <0.0001 0.0035 <0.0001 TTP was statistically significantly longer on the bortezomib arm (see Figure 3).
Figure 3: Time to Progression
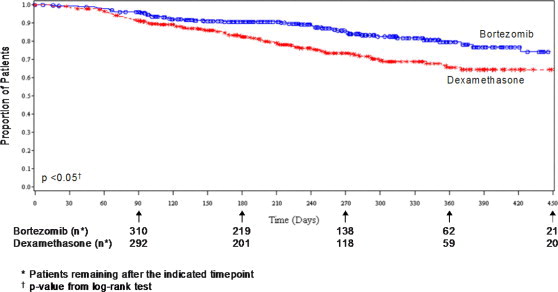
Bortezomib versus Dexamethasone (relapsed multiple myeloma study)As shown in Figure 4 bortezomib had a significant survival advantage relative to dexamethasone (p < 0.05). The median follow-up was 8.3 months.
Figure 4: Overall Survival
Bortezomib versus Dexamethasone (relapsed multiple myeloma study)For the 121 patients achieving a response (CR or PR) on the bortezomib arm, the median duration was 8 months (95% CI: 6.9, 11.5 months) compared to 5.6 months (95% CI: 4.8, 9.2 months) for the 56 responders on the dexamethasone arm. The response rate was significantly higher on the bortezomib arm regardless of β 2-microglobulin levels at baseline.
A Randomized Phase 2 Dose-Response Study in Relapsed Multiple Myeloma
An open-label, multicenter study randomized 54 patients with multiple myeloma who had progressed or relapsed on or after front-line therapy to receive bortezomib 1 mg/m 2 or 1.3 mg/m 2 intravenous bolus twice weekly for 2 weeks on Days 1, 4, 8, and 11 followed by a 10-day rest period (Days 12 to 21). The median duration of time between diagnosis of multiple myeloma and first dose of bortezomib on this trial was 2 years, and patients had received a median of 1 prior line of treatment (median of 3 prior therapies). A single complete response was seen at each dose. The overall response rates (CR + PR) were 30% (8/27) at 1 mg/m 2 and 38% (10/26) at 1.3 mg/m 2.
A Phase 2 Open-Label Extension Study in Relapsed Multiple Myeloma
Patients from the two phase 2 studies, who in the investigators' opinion would experience additional clinical
benefit, continued to receive bortezomib beyond 8 cycles on an extension study. Sixty-three (63) patients from the phase 2 multiple myeloma studies were enrolled and received a median of 7 additional cycles of bortezomib therapy for a total median of 14 cycles (range 7 to 32). The overall median dosing intensity was the same in both the parent protocol and extension study. Sixty-seven percent (67%) of patients initiated the extension study at the same or higher dose intensity at which they completed the parent protocol, and 89% of patients maintained the standard 3-week dosing schedule during the extension study. No new cumulative or new long-term toxicities were observed with prolonged bortezomib treatment [see Adverse Reactions ( 6.1)] .
14.2 Mantle Cell Lymphoma
A Phase 2 Single-arm Clinical Study in Relapsed Mantle Cell Lymphoma After Prior Therapy
The safety and efficacy of bortezomib in relapsed or refractory mantle cell lymphoma were evaluated in an
open-label, single-arm, multicenter study of 155 patients with progressive disease who had received at least 1 prior therapy. The median age of the patients was 65 years (42, 89), 81% were male, and 92% were Caucasian. Of the total, 75% had one or more extra-nodal sites of disease, and 77% were stage 4. In 91% of the patients, prior therapy included all of the following: an anthracycline or mitoxantrone, cyclophosphamide, and rituximab. A total of thirty seven percent (37%) of patients were refractory to their last prior therapy. An intravenous
bolus injection of bortezomib 1.3 mg/m 2/dose was administered twice weekly for 2 weeks on Days 1, 4, 8, and
11 followed by a 10-day rest period (Days 12 to 21) for a maximum of 17 treatment cycles. Patients achieving a CR or CRu were treated for 4 cycles beyond first evidence of CR or CRu. The study employed dose modifications for toxicity [see Dosage and Administration ( 2.4, 2.5)] .
Responses to bortezomib are shown in Table 13. Response rates to bortezomib were determined according to the International Workshop Response Criteria (IWRC) based on independent radiologic review of CT scans. The median number of cycles administered across all patients was 4; in responding patients the median number of cycles was 8. The median time to response was 40 days (range 31 to 204 days). The median duration of follow-up was more than 13 months.
Table 13: Response Outcomes in a Phase 2 Relapsed Mantle Cell Lymphoma Study Response Analyses (N = 155) N (%) 95% CI Overall Response Rate (IWRC) (CR + CRu + PR) 48 (31) (24, 39) Complete Response (CR + CRu) 12 (8) (4, 13) CR 10 (6) (3, 12) CRu 2 (1) (0, 5) Partial Response (PR) 36 (23) (17, 31) Duration of Response Median 95% CI CR + CRu + PR (N = 48) 9.3 months (5.4, 13.8) CR + CRu (N = 12) 15.4 months (13.4, 15.4) PR (N=36) 6.1 months (4.2, 9.3) - 15 REFERENCES
-
16 HOW SUPPLIED/STORAGE AND HANDLING
Bortezomib for Injection is supplied in a 10 mL vial containing 3.5 mg of bortezomib as a white to off-white cake or powder in a single-dose vial for reconstitution (after reconstitution the solution is clear and colorless).
Product No. NDC No. Strength 761210 63323-721-10 3.5 mg 10 mL single-dose vial, packaged individually. Unopened vials may be stored at 20°C to 25ºC (68°F to 77ºF); excursions permitted from 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Retain in original package to protect from light.
The vial stopper is not made with natural rubber latex.
Follow guidelines for handling and disposal for cytotoxic drugs, including the use of gloves and other protective clothing to prevent skin contact 1. ( 15)
-
17 PATIENT COUNSELING INFORMATION
Discuss the following with patients prior to treatment with Bortezomib for Injection:
Ability to Drive or Operate Machinery or Impairment of Mental Ability: Bortezomib for Injection may cause fatigue, dizziness, syncope, orthostatic/postural hypotension. Advise patients not to drive or operate machinery if they experience any of these symptoms [see Warnings and Precautions ( 5.2)].
Dehydration/Hypotension: Patients receiving Bortezomib for Injection therapy may experience vomiting and/or diarrhea. Advise patients how to avoid dehydration. Instruct patients to seek medical advice if they experience symptoms of dizziness, light headedness or fainting spells, or muscle cramps [see Warnings and Precautions ( 5.2)] .
Embryo-fetal Toxicity: Advise females of the potential risk to the fetus and to avoid pregnancy during treatment with Bortezomib for Injection. Advise female patients to use effective contraceptive measures to prevent pregnancy during treatment with Bortezomib for Injection and for 7 months following cessation of therapy. Advise male patients with female sexual partners of reproductive potential to use effective contraception during treatment with Bortezomib for Injection and for 4 months following cessation of therapy. Instruct patients to report pregnancy to their physicians immediately if they or their female partner becomes pregnant during treatment or within 6 months following treatment [see Warnings and Precautions ( 5.10)] .
Lactation: Advise patients to avoid breastfeeding while receiving Bortezomib for Injection and for 2 months after treatment [see Use in Specific Populations ( 8.2)] .
Concomitant Medications: Advise patients to speak with their physicians about any other medication they are currently taking.
Diabetic Patients: Advise patients to check their blood sugar frequently if using an oral antidiabetic medication and to notify their physicians of any changes in blood sugar level [see Use in Specific Populations ( 8.8)] .
Peripheral Neuropathy and Nervous System: Advise patients to contact their physicians if they experience new or worsening symptoms of peripheral neuropathy such as tingling, numbness, pain, a burning feeling in the feet or hands, or weakness in the arms or legs. Advise patients to contact their physicians if they experience symptoms possibly indicative of PRES [see Warnings and Precautions ( 5.5)] or PML, such as convulsion, persistent headache, reduced eyesight, blurred vision, confusion, lethargy, altered ability to think, or difficulty walking.
Cardiac: Advise patients to contact their physicians if they experience swelling of the feet, ankles, or legs or other heart-related problems [see Warnings and Precautions ( 5.3)] .
Respiratory: Advise patients to contact their physicians if they experience shortness of breath, cough, or other lung problems [see Warnings and Precautions ( 5.4)] .
Hepatic: Advise patients to contact their physicians if they experience jaundice or right upper quadrant abdominal pain [see Warnings and Precautions ( 5.9)] .
-
PRINCIPAL DISPLAY PANEL
Principal Display Panel - Bortezomib For Injection Vial Label
NDC: 63323-721-10 761210
BORTEZOMIB
FOR INJECTION3.5 mg per vial
FOR INTRAVENOUS USE ONLY
Single-Dose Vial.
Discard Unused Portion.
Rx only
-
PRINCIPAL DISPLAY PANEL
Principal Display Panel - Bortezomib For Injection Carton Label
NDC: 63323-721-10 761210
BORTEZOMIB
FOR INJECTION3.5 mg per vial
FOR INTRAVENOUS USE
ONLYSingle-Dose Vial.
Discard Unused Portion.
Rx only
-
INGREDIENTS AND APPEARANCE
BORTEZOMIB
bortezomib injection, powder, lyophilized, for solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 63323-721 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BORTEZOMIB (UNII: 69G8BD63PP) (BORTEZOMIB - UNII:69G8BD63PP) BORTEZOMIB 1 mg in 1 mL Inactive Ingredients Ingredient Name Strength BORIC ACID (UNII: R57ZHV85D4) GLYCINE (UNII: TE7660XO1C) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 63323-721-10 1 in 1 CARTON 11/06/2017 1 10 mL in 1 VIAL, SINGLE-DOSE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA205004 11/06/2017 Labeler - Fresenius Kabi USA, LLC (608775388) Establishment Name Address ID/FEI Business Operations Fresenius Kabi USA, LLC 840771732 manufacture(63323-721)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.