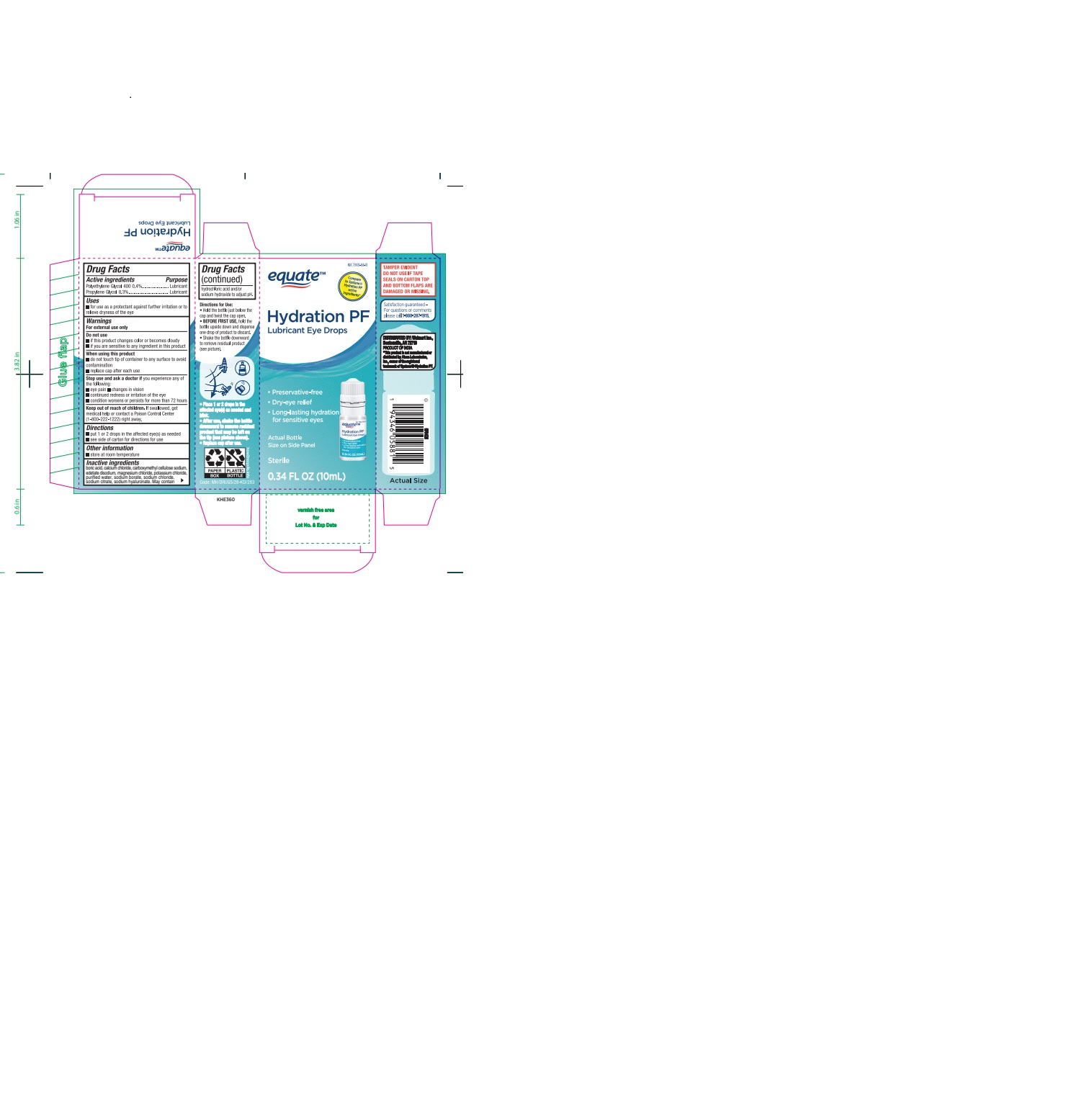
Hydration PF Lubricant Eye drops
HYDRATION PF by
Drug Labeling and Warnings
HYDRATION PF by is a Otc medication manufactured, distributed, or labeled by Walmart. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
HYDRATION PF- polyethylene glycol 400 and propylene glycol solution/ drops
Walmart
----------
Hydration PF Lubricant Eye drops
Do not use
- if this product changes color or becomes cloudy
- if you are sensitive to any ingredient in this product
When using this product
- do not touch tip of container to any surface to avoid contamination
- replace cap after each use
Stop use and ask a doctor ifyou experience any of the following:
- eye pain
- changes in vision
- continued redness or irritation of the eye
- condition worsens or persists for more than 72 hours
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
Directions
- put 1 or 2 drops in the affected eye(s) as needed.
- see side of carton for directions for use
| HYDRATION PF
polyethylene glycol 400 and propylene glycol solution/ drops |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Walmart (051957769) |
Revised: 1/2024
Document Id: 0fa8b3b7-572b-73df-e063-6394a90a7d63
Set id: 0450baa1-b361-df64-e063-6394a90a0965
Version: 2
Effective Time: 20240123
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.