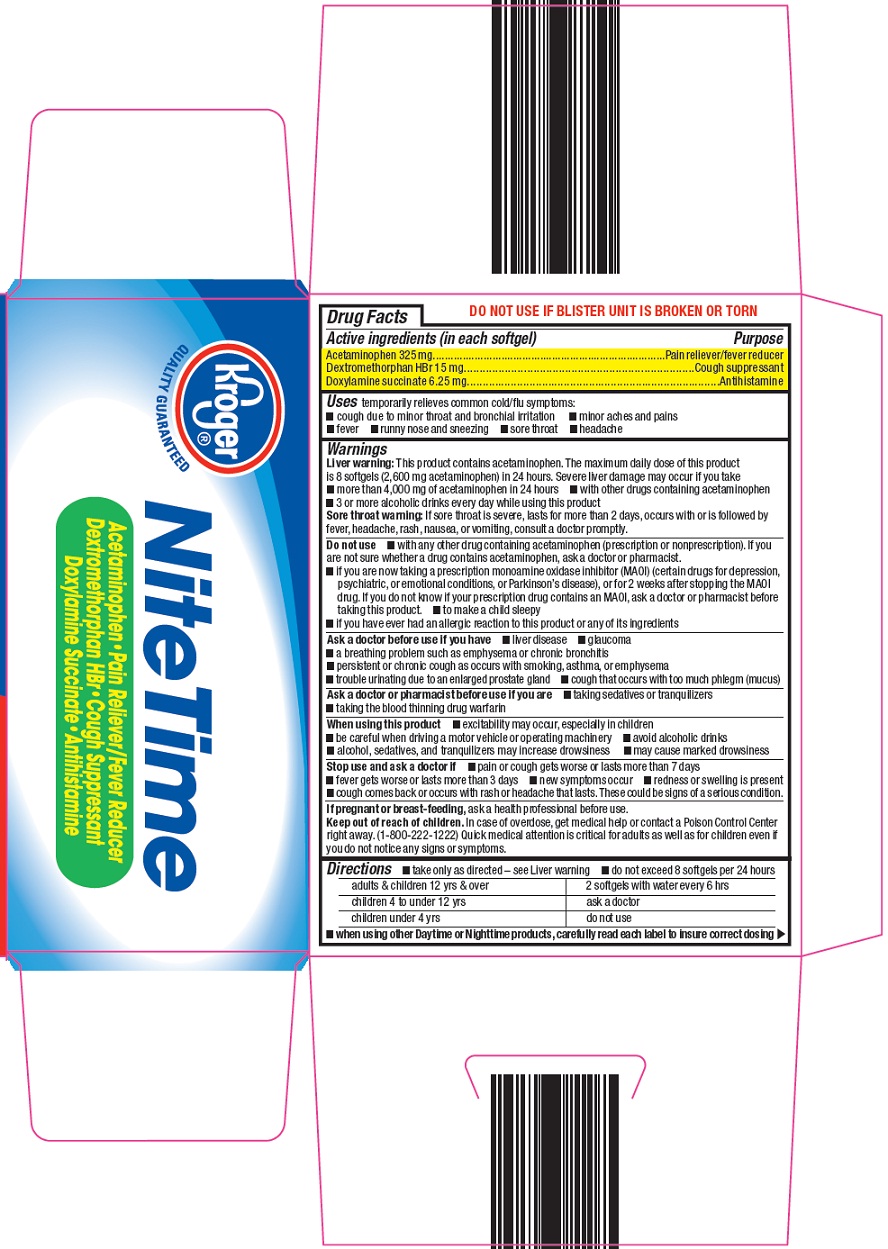
NITE TIME MULTI SYMPTOM COLD/FLU- acetaminophen, dextromethorphan hydrobromide, doxylamine succinate capsule, liquid filled
Nite Time by
Drug Labeling and Warnings
Nite Time by is a Otc medication manufactured, distributed, or labeled by Kroger Company. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Active ingredient (in each softgel)
- Purpose
- Uses
-
Warnings
Liver warning: This product contains acetaminophen. The maximum daily dose of this product is 8 softgels (2,600 mg acetaminophen) in 24 hours. Severe liver damage may occur if you take
- more than 4,000 mg of acetaminophen in 24 hours
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Sore throat warning: If sore throat is severe, lasts for more than 2 days, occurs with or is followed by fever, headache, rash, nausea, or vomiting, consult a doctor promptly.
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
- to make a child sleepy
- if you have ever had an allergic reaction to this product or any of its ingredients
Ask a doctor before use if you have
- liver disease
- glaucoma
- cough that occurs with too much phlegm (mucus)
- a breathing problem such as emphysema or chronic bronchitis
- persistent or chronic cough as occurs with smoking, asthma, or emphysema
- trouble urinating due to an enlarged prostate gland
Ask a doctor or pharmacist before use if you are
- taking sedatives or tranquilizers
- taking the blood thinning drug warfarin
When using this product
- excitability may occur, especially in children
- may cause marked drowsiness
- avoid alcoholic drinks
- be careful when driving a motor vehicle or operating machinery
- alcohol, sedatives, and tranquilizers may increase drowsiness
-
Directions
- take only as directed – see Liver warning
- do not exceed 8 softgels per 24 hours
adults & children 12 yrs & over
2 softgels with water every 6 hrs
children 4 to under 12 yrs
ask a doctor
children under 4 yrs
do not use
- when using other Daytime or Nighttime products, carefully read each label to insure correct dosing
- Inactive ingredients
- Questions or comments?
-
Principal Display Panel
COMPARE TO active ingredients of VICKS® MYQUIL® COLD & FLU LIQUICAPS®
See end panel
ALCOHOL FREE
SEE NEW WARNINGS AND DIRECTIONS
Multi-Symptom – Cold/Flu Relief
NiteTime
Acetaminophen – Pain Reliever/Fever Reducer
Dextromethorphan HBr – Cough Suppressant
Doxylamine Succinate – Antihistamine
Aches & Fever
Cough
Sneezing & Runny Nose
48 SOFTGELS
-
INGREDIENTS AND APPEARANCE
NITE TIME MULTI SYMPTOM COLD/FLU
acetaminophen, dextromethorphan hydrobromide, doxylamine succinate capsule, liquid filledProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 30142-977 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 325 mg DEXTROMETHORPHAN HYDROBROMIDE (UNII: 9D2RTI9KYH) (DEXTROMETHORPHAN - UNII:7355X3ROTS) DEXTROMETHORPHAN HYDROBROMIDE 15 mg DOXYLAMINE SUCCINATE (UNII: V9BI9B5YI2) (DOXYLAMINE - UNII:95QB77JKPL) DOXYLAMINE SUCCINATE 6.25 mg Inactive Ingredients Ingredient Name Strength D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) GELATIN, UNSPECIFIED (UNII: 2G86QN327L) GLYCERIN (UNII: PDC6A3C0OX) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) Product Characteristics Color GREEN Score no score Shape CAPSULE (OBLONG) Size 20mm Flavor Imprint Code L977 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 30142-977-60 2 in 1 CARTON 05/09/2006 1 10 in 1 BLISTER PACK; Type 0: Not a Combination Product 2 NDC: 30142-977-58 4 in 1 CARTON 05/09/2006 2 10 in 1 BLISTER PACK; Type 0: Not a Combination Product 3 NDC: 30142-977-62 2 in 1 CARTON 05/09/2006 3 12 in 1 BLISTER PACK; Type 0: Not a Combination Product 4 NDC: 30142-977-67 4 in 1 CARTON 05/09/2006 4 12 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 05/09/2006 Labeler - Kroger Company (006999528)
Trademark Results [Nite Time]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 NITE TIME 80985417 0985417 Dead/Cancelled |
Milton Bradley Company 0000-00-00 |
 NITE TIME 75309714 not registered Dead/Abandoned |
HEART FOODS COMPANY, INC. 1997-06-16 |
 NITE TIME 72227356 0811749 Dead/Cancelled |
SULLCRAFT MANUFACTURING COMPANY 1965-09-07 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.