ALLERGY RELIEF- loratadine capsule, liquid filled
ALLERGY RELIEF by
Drug Labeling and Warnings
ALLERGY RELIEF by is a Otc medication manufactured, distributed, or labeled by CARDINAL HEALTH 110, LLC. DBA LEADER. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- SPL UNCLASSIFIED SECTION
- Active Ingredient (in each capsule)
- Purpose
- Uses
-
Warnings
Do not useif you have ever had an allergic reaction to this product or any of its ingredients.
Ask a doctor before use if you haveliver or kidney disease. Your doctor should determine if you need a different dose.
When using this productdo not take more than directed. Taking more than directed may cause drowsiness.
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
PRINCIPAL DISPLAY PANEL
LEADER™
NDC: 70000-0755-1
Non-Drowsy* | 24 Hour
Allergy Relief
Loratadine, 10 mg | Antihistamine
Indoor and Outdoor Allergies
For Relief of:
Sneezing, Runny Nose
Itchy, Watery Eyes
Itchy Throat or Nose
Actual Size
COMPARE TO CLARITIN® LIQUI-GELS® active ingredient†
100% Money Back Guarantee
10 LIQUID-FILLED CAPSULES
*When taken as directed. See Drug Facts Panel
100% Money Back Guarantee
TAMPER EVIDENT: DO NOT USE IF
IMPRINTED SAFETY SEAL UNDER
CAP IS BROKEN OR MISSING†This product is not manufactured or distributed by
Bayer Healthcare LLC, distributor of Claritin®, or Catalent Pharma Soulutions, Inc.,
owner of the registered trademark Liqui-Gels®.CIN 6104293 Rev. 02/26
DISTRIBUTED BY CARDINAL HEALTH
DUBLIN, OHIO 43017
www.myleader.com 1-800-200-6313
Essential to Care™ since 1979
Made in India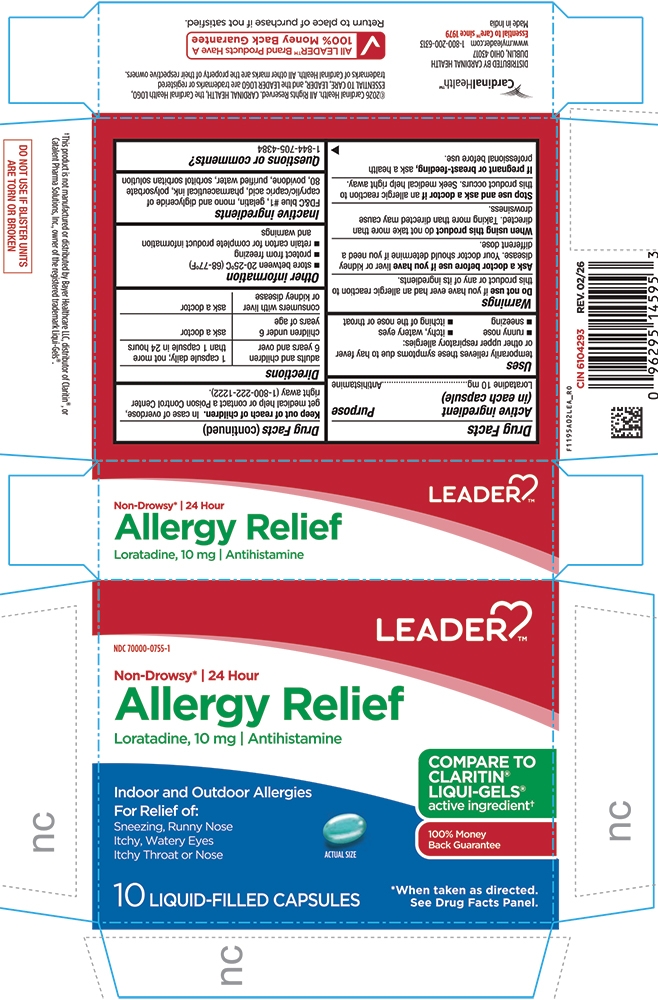
-
INGREDIENTS AND APPEARANCE
ALLERGY RELIEF
loratadine capsule, liquid filledProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 70000-0755 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LORATADINE (UNII: 7AJO3BO7QN) (LORATADINE - UNII:7AJO3BO7QN) LORATADINE 10 mg Inactive Ingredients Ingredient Name Strength CAPRIC ACID (UNII: 4G9EDB6V73) POVIDONE K30 (UNII: U725QWY32X) WATER (UNII: 059QF0KO0R) SORBITOL (UNII: 506T60A25R) POLYSORBATE 80 (UNII: 6OZP39ZG8H) SORBITAN (UNII: 6O92ICV9RU) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) GELATIN (UNII: 2G86QN327L) Product Characteristics Color blue (Light Blue) Score no score Shape OVAL (oval shaped) Size 3mm Flavor Imprint Code 21 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 70000-0755-1 10 in 1 CARTON 03/18/2026 1 1 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA206214 03/18/2026 Labeler - CARDINAL HEALTH 110, LLC. DBA LEADER (063997360)
Trademark Results [ALLERGY RELIEF]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 ALLERGY RELIEF 98236984 not registered Live/Pending |
Dmytro Kononenko 2023-10-24 |
 ALLERGY RELIEF 90457167 not registered Live/Pending |
American Textile Company, Inc. 2021-01-10 |
 ALLERGY RELIEF 78838437 3358249 Live/Registered |
Meshbesher Health Corporation 2006-03-16 |
 ALLERGY RELIEF 76619855 3066888 Live/Registered |
AMERICAN TEXTILE COMPANY 2004-11-09 |
 ALLERGY RELIEF 74668018 not registered Dead/Abandoned |
NaturaLife Corporation 1995-05-01 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.