STELFONTA 1 MG/ML- tigilanol tiglate injection solution
STELFONTA 1 mg/mL by
Drug Labeling and Warnings
STELFONTA 1 mg/mL by is a Animal medication manufactured, distributed, or labeled by Virbac AH Inc, QBiotics Group LTD. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- STELFONTA® (tigilanol tiglate injection)
-
BOXED WARNING
(What is this?)
WARNING: SEVERE WOUND FORMATION IN HUMANS;
EXTENSIVE WOUND FORMATION, MAST CELL DEGRANULATION, AND
DEATH IN DOGS DUE TO MAST CELL DEGRANULATIONHuman Safety
- Accidental self-injection of STELFONTA® may cause severe wound formation. To decrease the risk of accidental self-injection, sedation of the dog may be necessary (see Dosage and Administration, Human Warnings and Adverse Reactions).
Dog Safety
- Always administer a corticosteroid (e.g. prednisone or prednisolone), an H1 receptor blocking agent (e.g. diphenhydramine), and an H2 receptor blocking agent (e.g. famotidine) when treating with STELFONTA to decrease the potential for severe systemic adverse reactions, including death, from mast cell degranulation (see Contraindications and Dosage and Administration).
- Do not inject STELFONTA into subcutaneous mast cell tumors located above the elbow or hock (e.g. on the body, head, or neck). This may result in accumulation of necrotic debris in the subcutaneous space increasing the risk of systemic adverse reactions, including death, from mast cell degranulation (see Contraindications, Warnings and Adverse Events).
- Treatment with STELFONTA has been associated with cellulitis and severe tissue sloughing extending away from the treated site resulting in extensive wounds that require additional treatment and prolonged recovery times (see Warnings, Precautions and Adverse Events).
- CAUTION
-
DESCRIPTION
The active ingredient for tigilanol tiglate injection is a phorbol ester that activates alpha, beta I, beta II, and gamma isoforms of protein kinase C. The chemical name is (4S,5S,6R,7S,8R,9R,10S,11R,12R,13S,14R)-12-(2E)-2-methylbut-2-enoatyl-13- [(2S)-2-methylbutyroyl]-6,7-epoxy-4,5,9,12,13,20-hexahydroxy-1-tigliaen-3-one. The molecular formula is C30H42O10 and its molecular weight is 562.65 g mol-1. Each mL of STELFONTA contains 1 mg tigilanol tiglate and sterile water for injection (60% v/v), propylene glycol (40% v/v), sodium acetate (<0.1% w/v), and glacial acetic acid (<0.1% w/v).
The chemical structure for tigilanol tiglate is:

- INDICATION
-
DOSAGE AND ADMINISTRATION
ALWAYS PROVIDE THE CLIENT INFORMATION SHEET TO THE DOG OWNER
BEFORE DOSE ADMINISTRATION. Carefully consider the potential benefits and risks of STELFONTA before deciding to use STELFONTA.It is crucial to follow the dosing and administration instructions to use the product safely and effectively (see Boxed Warning, Animal Warnings, Precautions, Clinical Pharmacology).
Concomitant medications
Administer the following medications to decrease the potential for severe systemic adverse reactions from mast cell degranulation (see Effectiveness). Do not underdose concomitant medications and confirm that the dog owner administered the medications as prescribed prior to the date of STELFONTA treatment.
- Corticosteroid (oral prednisone or prednisolone at anti-inflammatory dose): Start medication 2 days prior to STELFONTA treatment at a dose of 0.5 mg/kg orally every 12 hours for 7 days (2 days prior, the day of treatment, and 4 days after treatment), then 0.5 mg/kg orally every 24 hours for an additional 3 days (10 days total).
- H1 receptor blocking agent (oral diphenhydramine): Start medication on the day of STELFONTA treatment at a dose of 2 mg/kg orally every 12 hours and continue for a total of 8 days (the day of treatment and 7 days after treatment).
- H2 receptor blocking agent (oral famotidine): Start medication on the day of STELFONTA treatment at a dose of 0.5 mg/kg orally every 12 hours and continue for a total of 8 days (the day of treatment and 7 days after treatment).
- Fill out the medication schedule (drug name, dose, route of administration, date) on the Client Information Sheet to help the dog owner administer these medicaitons correctly.
Consider administering analgesic medicaitons prior to, during, and after treatment with STELFONTA.
Dosing Instructions
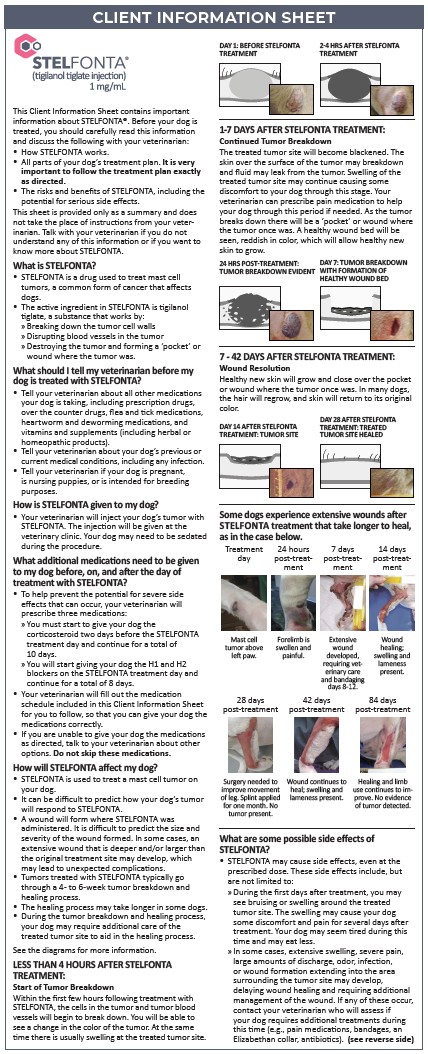
Administer STELFONTA as an intratumoral injection at a dose of 0.5 mL per cm3 of tumor volume, as determined by the following calculations:
STEP 1. Calculate Tumor Volume:
- Measure the tumor dimensions (Length, Width and Height) with calipers on the day of STELFONTA injection.
- Determine the Tumor Volume using the midified ellipsoid formula to account for the tumor shape (cube volume x 1/2) as below:
- Confirm the Tumor Volume does not exceed 10 cm3.
- Do not use STELFONTA if tumor volume is >10 cm3.
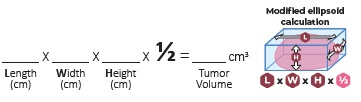
STEP 2. Calculate the mL of STELFONTA to inject:
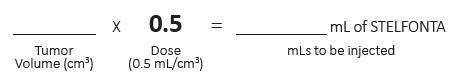
- Confirm the dose of STELFONTA does not exceed 0.25 mL/kg body weight of body weight and do not use if the calculate dose exceeds this.
- Do not exceed 5 mL per dog, regardless of tumor volume or body weight.
- The minimum dose of STELFONTA is 0.1 mL, regardless of tumor volume or body weight. If the calculated dose is <0.1 mL, administer 0.1 mL.
- Confirm the calculate dose of STELFONTA using the online dosing calculator at www.stelfonta.com/calculator (or scan the QR code below).
Administration of STELFONTA:
Sedation may be necessary to safely and accurately administer STELFONTA to decrease the chance of accidental self-injection. Wear gloves, eye protection, and lab coat or gown in the preparation and administration of STELFONTA. Care should be taken to restrict injections to the tumor only. STELFONTA should not be injected into the margins, beyond the periphery, or deep to the tumor.
- Shave the tumor site. Avoid manipulation of the tumor.
- Draw the calculated volume of STELFONTA into a sterile Luer-lock syringe with a 23 gauge needle.
- Identify an appropriate injection point on the edge of the tumor. See Figure 1. Insertion of the needle depends on the tumor’s location, form, and appearance. If a tumor protrudes above the surface of the skin, insert the needle at an oblique angle of approximately 45°.
- Insert and embed the needle in the tumor through a single injection site and draw the syringe plunger back slightly to ensure STELFONTA is not injected into a blood vessel. While applying even pressure on the syringe plunger move the needle back and forth in a fanning manner to inject STELFONTA into the tumor. See Figure 1. The drug should fully perfuse the entire tumor.
- When the total dose of STELFONTA has been administered, pause to allow tissue dispersion before removing the needle from the tumor. Pull back on the syringe plunger to create a small negative pressure before removing the needle to minimize leakage from the injection site.
- After the needle is withdrawn, apply light pressure for 30 seconds over the needle exit hole using a gloved finger. If leakage does occur, rinse injection site with saline to wash STELFONTA from the skin surface. Do not readminister.
- To minimize risk of accidental self-injection, do not recap the needle. Disposeof the needle and syringe.
-
CONTRAINDICATIONS
Do not inject STELFONTA into subcutaneous mast cell tumors located above the elbow or hock (e.g. on the body, head, or neck). This may result in accumulation of necrotic debris in the subcutaneous space increasing the risk of systemic adverse reactions, including death, from mast cell degranulation (see Adverse Reactions).
-
WARNINGS
Human Safety Warnings
NOT FOR USE IN HUMANS. KEEP THIS AND ALL MEDICATIONS OUT OF REACH OF CHILDREN.
Caution is required during treatment to avoid accidental self-injection. Dogs undergoing treatment with STELFONTA should be adequately restrained and sedation used if necessary. Use a Luer-lock syringe to administer STELFONTA. Do not recap the needle. Accidental self-injection may result in local inflammatory reactions, including swelling, redness and severe wound formation. In case of accidental self-injection, immediately rinse the area with water, seek medical advice immediately, and show the package insert to the physician.
Wear personal protective equipment consisting of disposable gloves, protective eye wear, and a lab coat or gown when handling STELFONTA. STELFONTA is an irritant and accidental exposure to skin, eye, or by ingestion should be avoided. In case of dermal or ocular exposure, repeatedly wash the exposed skin or eye with water. If wearing contacts, rinse the eyes first then remove contacts and continue to rinse with water. If symptoms such as local signs of redness and swelling occur, or if there has been ingestion, seek the advice of a physician and show them the package insert.
Limited data is available on the potential teratogenic effects of STELFONTA. Therefore, STELFONTA should not be administered by women who are pregnant or planning to become pregnant.
People with known hypersensitivity to tigilanol tiglate or to any of the excipients should avoid contact with STELFONTA.
Animal Safety Warnings
Dogs should be monitored during and for 5-7 days after intratumoral treatment with STELFONTA for signs of systemic mast cell degranulation such as vomiting, diarrhea, lethargy, anorexia/hyporexia, altered breathing, hypotension, urticaria, edema at or away from the treated site, or bruising at or away from the treated site. If signs are observed, appropriate treatment should be started immediately.
Always administer the concomitant medications (prednisone or prednisolone, diphenhydramine, and famotidine), as directed in the Dosage and Administration section, with STELFONTA in order to decrease the potential for severe systemic adverse reactions, including death, from mast cell degranulation (see Adverse Reactions).
Treatment with STELFONTA causes tumor necrosis which is part of the mechanism of action of the drug. Bruising, heat, pain, and swelling may begin at the site within 2 hours of treatment. By day 7 after treatment, wound formation including full thickness dermal necrosis with exudate, peripheral tissue edema, erythema, skin discoloration, tissue sloughing, and necrotic eschar may occur.
STELFONTA can induce a substantial local inflammatory reaction which may result in severe pain and swelling, bruising, cellulitis, extensive wound formation, and severe tissue sloughing extending away from the treated site. Consider administering analgesic medications prior to, during, and after treatment with STELFONTA in addition to the use of corticosteroids and both H1 and H2 receptor blocking agents.
Amputation of an extremity has been reported in some cases (see Post-Approval Experience).
Some dogs require wound care and pain management for an extended period.
Do not inject STELFONTA into normal subcutaneous tissue or adjacent tissues (e.g. beyond tumor margins) because severe edema, erythema and necrosis of the injected tissue may occur.
-
PRECAUTIONS
STELFONTA is not intended for the treatment of metastatic mast cell tumors.
The safe and effective use of STELFONTA has not been evaluated in dogs with a mast cell tumor volume >10 cm3.
Use STELFONTA with caution in tumors located within mucocutaneous regions (e.g., eyelids, vulva, prepuce, and anus) as tumor necrosis could cause a change in morphology of the mucocutaneous region resulting in loss of functional integrity.
Use STELFONTA with caution in mast cell tumors with significant ulceration as leakage of the drug from the ulcerated area may occur following treatment potentially reducing effectiveness.
Some discharge from the site following treatment is expected. Wear disposable gloves to clean the site with warm water as necessary.
After treatment with STELFONTA, dogs may require additional care of the treated site to aid in the healing process, especially if there is extensive wound formation (see Animal Safety Warnings and Post-Approval Experience).
Tongue lesions have been reported (see Post-Approval Experience). Do not allow the dog to lick the site for the first few days after treatment. Discourage excessive licking for the remainder of the healing period.
An Elizabethan collar or a non-constricting dry gauze bandage may be needed to prevent the dog from self-traumatizing the treated site.
After treatment with STELFONTA, separation from other household animals may be necessary to prevent grooming and trauma to the treated site.
STELFONTA has not been evaluated in dogs with signs of systemic disease due to the mast cell tumor(s).
The safe and effective use of STELFONTA has not been evaluated for sumultaneous treatment of more than one mast cell tumor.
The safe use of STELFONTA has not been evaluated in dogs with concurrent diseases that may result in delayed wound healing.
The safe use of STELFONTA under conditions of use has not been evaluated in dogs younger than 3.5 years old.
The safe use of STELFONTA has not been evaluated in dogs that are pregnant, lactating, or intended for breeding.
-
ADVERSE REACTIONS
Human Exposure
There was one human exposure during the field study where the veterinarian had a needle stick injury to the thumb at completion of tumor treatment and was injected with an unknown amount of STELFONTA. The incident resulted in pain and necrosis of the center of the thumb at the point of needle stick. The wound healed over a period of three months. See Pictures 1 and 2 below. A separate needle stick injury was reported with a maximum potential dose of 0.1 mL tigilanol tiglate into the distal extremity of the left index finger, resulting in a localized burning sensation, local inflammation, bruising, muscular pain up the left arm, and localized tissue necrosis. Muscular pain resolved in the first 12-24 hours and the wound healed in 8 weeks. There have been other needle stick injuries reported, with at least one injection into a thumb, with minimal (stinging, pain, and swelling) to no adverse events associated with these accidental self-injections.
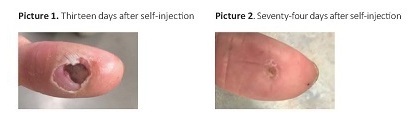
Field Study
In a well-controlled, multi-center, randomized, double-masked field study evaluating the effectiveness and safety of STELFONTA for the treatment of cutaneous and subcutaneous mast cell tumors in dogs, 117 dogs treated with STELFONTA and 42 dogs receiving sham treatment (untreated control) were evaluated for safety. Eighty-one dogs were treated with STELFONTA on Day 0. Thirty-six previously untreated control dogs were treated with STELFONTA on Day 30. In addition, 18 dogs treated with STELFONTA on Day 0 had the same tumor re-treated with STELFONTA on Day 30 due to incomplete response. The most common adverse reactions included wound formation, injection site pain, lameness in the treated limb, vomiting, diarrhea, and hypoalbuminemia. Wound formation, vomiting, and diarrhea were mainly observed within the first 7 to 10 days after treatment. Injection site pain and lameness in the treated leg were mainly observed within the first 2 days after treatment. Hypoalbuminemia was mainly observed within the first 28 days after treatment. All dogs received concomitant medications as noted in the Effectiveness section. The adverse reactions during the study are summarized in Table 1 below.
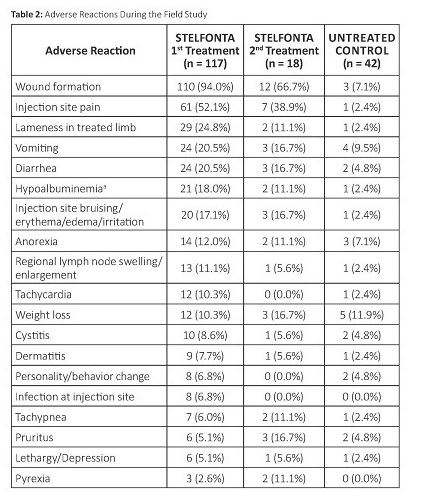
a There was a statistically significant decrease in albumin and albumin/globulin ratios at Day 7 in the STELFONTA group compared to the control group. The hypoalbuminemia ranged from 2.0 to 2.6 g/dL (reference range 2.7-3.9 g/dL). Note: If an animal experienced the same adverse reaction more than once, onlythe highest grade was tabulated.
Adverse reactions were graded using the Veterinary Co-operative Oncology Group – Common Terminology Criteria for Adverse Events (VCOG-CTCAE).1 Most adverse reactions were Grade 1 (mild) or 2 (moderate). Grade 3 (severe) and 4 (life-threatening) adverse reactions in dogs treated with STELFONTA included: lameness in the treated limb (6 dogs), injection site pain (4 dogs), wound formation (3 dogs), lethargy/depression (3 dogs), anorexia (2 dogs), infection at injection site (1 dog), pruritis (1 dog), and tachycardia (1 dog).
Adverse reactions associated with use of the required concomitant corticosteroids were similarly reported in STELFONTA and untreated control
dogs and included elevated alkaline phosphatase, polyuria, and polydipsia.Wound Formation
Tumor observations were conducted at 2, 4, 8, and 24 hours and 4 days after treatment. The 81 dogs treated with STELFONTA on Day 0 were reported most frequently with swelling, bruising, pain and heat at all tumor observation timepoints. The following were reported at 24 hours post treatment:
- Swelling: 97.5% (79/81 dogs)
- Bruising: 91.4% (74/81 dogs)
- Pain: 69.1% (56/81 dogs)
- Heat: 53.1% (43/81 dogs)
At 24 hours post treatment, intact skin was reported in 71.6% (58/81 dogs) of STELFONTA treated dogs. On Day 4 intact skin was reported in 17.3% (14/81 dogs) of STELFONTA treated dogs. On Day 4, the following observations were reported with the highest frequency:
- Necrosis: 55.6% (45/81 dogs)
- Crater pockets: 37.0% (30/81 dogs)
- Exudate: 37.0% (30/81 dogs)
- Eschar: 28.4% (23/81 dogs)
- Ulceration: 11.1% (9/81 dogs)
A wound healing assessment was performed on the effectiveness dataset which included 80 dogs in the STELFONTA group and 38 dogs in the untreated control group. Wounds developed in 92.5% (74/80) of STELFONTA treated dogs and 2.6% (1/38) of untreated control dogs by Day 7. On Day 28, the presence of wounds was 40% (32/80) in the STELFONTA group and 2.6% (1/38) in the untreated control group. On Day 42 and Day 84, the presence of wounds was 27.1% (16/59) and 1.8% (1/57), respectively, in the STELFONTA group.
Exudate from the treated site including serous, serosanguinous, sanguineous, seropurulent, and purulent discharges were seen mainly on Day 7 and to a lesser extent on Day 14. Sloughing of the treated site was observed from Day 7 to Day 42, with decreasing frequency after Day 7. Peripheral pitting or non-pitting edema and erythema of the surrounding area were observed from Day 7 to Day 28, with decreasing intensity and frequency after Day 7. Necrotic eschar and epithelialization of the treated site was observed from Day 7 to Day 84, with decreasing frequency after Day 14. Granulation or hyper-granulation of the treated site was observed from Day 7 to Day 84, with decreasing frequency after Day 14.
The average wound size at Day 7 for a STELFONTA treated dog was 3.3 cm x 2.4 cm (original average tumor size 1.9 x 1.6 x 0.9 cm). On Day 28, the average wound size was 2.0 x 1.4 cm.
The largest total wound for a STELFONTA treated dog was reported seven days after treatment. The treated tumor was located on the left caudal stifle and the original tumor size measured 2.4 x 2.1 x 1.4 cm. The wound area initially consisted of three individual wounds recorded on the treated limb (both medial and lateral sides): 7.5 x 4.5 cm, 7.0 x 3.5 cm, and 11.5 x 7.0 cm. The wounds had reduced to 3.5 x 1.4 cm, 3.9 x 1.5 cm, and 9.7 x 4.3 cm 28 days after treatment, and 0.5 x 0.7 cm and 2.5 x 2.9 cm 42 days after treatment and were no longer present at 84 days after treatment.
One dog treated with STELFONTA was reported with an extensive wound formation (wound size 25.0 x 9.5 cm) with severe tissue slough (Grade 3) nine days after treatment of a mast cell tumor on the left metacarpal area (original tumor size 2.5 x 1.9 x 1.3 cm). The wound extended proximally up the leg to the shoulder and required bandaging of the leg and antibiotics. Scar contracture formed, requiring treatment under sedation to release the scar tissue. Clinical pathology abnormalities included elevated band neutrophils, anemia, and hypoalbuminemia. The wound had not fully healed by the end of the study 89 days after treatment. See pictures below comparing progression of this extensive
wound formation versus commonly observed wound progression.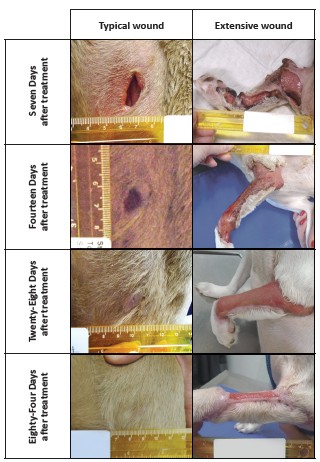
One dog treated with STELFONTA was reported with a bacterial infection and cellulitis in the right rear leg 9 days after treatment of a mast cell tumor on the right rear paw. There was bruising of the upper thigh and necrotic skin on the caudal right thigh and cranial aspect of the hock. Bloody discharge under the necrotic tissue revealed rod bacteria and toxic neutrophils. The dog was treated with intravenous fluids and antibiotics.
Systemic Mast Cell Degranulation and Death
Two dogs from two separate pilot studies died from a suspected mast cell degranulation reaction. Both dogs were treated with STELFONTA for a subcutaneous mast cell tumor located above the hock and did not receive the concomitant medications as prescribed.
In a pilot field study, one dog with a large (10 cm³) subcutaneous mast cell tumor on the right hip was treated with STELFONTA. The dog had a partial Response Evaluation Criteria in Solid Tumors Guideline (RECIST)2 response to the initial STELFONTA injection and was re-treated with STELFONTA, 30 days following the initial injection. The patient did not receive any of the recommended concomitant medications of prednisolone, chlorpheniramine and famotidine from 24 hours after the second STELFONTA injection. On Day 2 following the second STELFONTA injection, the dog became anorexic, painful, and lethargic and had marked swelling of the right hind limb extending to the chest with hemorrhagic, ruptured blisters near the hock joint. Blood work showed anemia, hypoproteinemia, liver enzyme elevations, and white blood cell changes (leukocytosis, neutrophilia, monocytosis, and thrombocytopenia). The dog was hospitalized, received a blood transfusion, and was administered intravenous fluids, prednisolone, chlorpheniramine and tramadol. Pitting edema progressed to the neck by four days following treatment. Despite supportive care, the dog died five days following treatment likely due to degranulation of the mast cell tumor and internal necrotic discharge of the tumor.
In a separate pilot field study, one dog with a moderate (2.53 cm3) subcutaneous mast cell tumor on the left caudal hindlimb was treated with STELFONTA. The dog was treated with chlorpheniramine and meloxicam on treatment day (Day 0) and Day 1 only. The dog did not receive further concomitant medication. On Day 3 the dog was lethargic and there was significant edema at the injection site. While intravenous fluid and antibiotic therapy was initiated on Day 3, the dog rapidly deteriorated and died on the following day likely due to degranulation of the mast cell tumor. Pathology findings included widespread cellulitis, panniculitis (likely of bacterial origin), and septic peritonitis.
Post-Approval Experience (2024)
The following adverse events are based on post-approval adverse drug experience reporting for STELFONTA. Not all adverse events are reported to FDA/CVM. It is not always possible to reliably estimate the adverse event frequency or establish a causal relationship to product exposure using these data.
The following adverse events reported in dogs are listed in decreasing order of reporting frequency:
Injection site reactions (wound formation, swelling, pain, necrosis, skin sloughing, bleeding, bruising and erythema). These injection site reactions varied in severity and ranged from localized to extending away from the injection site.
Other signs reported include anorexia, lameness, lethargy, diarrhea, vomiting, fever, tachypnea/dyspnea, and ulceration or necrosis of tongue.
In some cases, when STELFONTA was used to treat a mast cell tumor on an extremity, the entire extremity became swollen, painful, and developed tissue sloughing. Some of these cases resulted in amputation.
In some cases, death (including euthanasia) has been reported as an outcome of the adverse events reported above.CONTACT INFORMATION
To report suspected adverse drug experiences, to obtain a Safety Data Sheet (SDS), or for technical assistance, contact Virbac at 1-800-338-3659 or us.virbac.com.
For additional information about adverse drug experience reporting for animal drugs, contact the FDA at 1-888-FDA-VETS or www.fda.gov/reportanimalae.INFORMATION FOR DOG OWNERS
Owners should be given the Client Information Sheet (CIS) to read before STELFONTA is administered.
Owners should be advised to observe their dog for potential side effects, including signs of systemic mast cell degranulation, excessive pain and swelling, and excessive wound formation. Advise dog owners about the possibility of severe side effects, including amputation and death, when to contact a veterinarian, and how to care for the treated tumor site.
Some discharge from the site following treatment is expected. The site can be cleaned with warm water as necessary. Advise owners to wear disposable gloves when cleaning the area.
Discuss the importance of the concomitant medications and ensure that the owner is aware of the schedule of medications that should be administered. The owner should use the Medication Schedule on the CIS to keep track of the medications they have administered.
Discuss with owners that they should not allow the dog to lick the site for the first few days after treatment and they should discourage excessive licking for the remainder of the healing period.
An Elizabethan Collar may be utilized to prevent self-trauma of the treatment site. After treatment the owner may need to separate the dog from other household animals to prevent grooming and trauma to the treated site. -
CLINICAL PHARMACOLOGY
Mechanism of Action
In non-clinical pharmacology studies, tigilanol tiglate has been shown to have three inter-related effects that are responsible for its anti-tumor effectiveness. The first effect is to cause oncolysis of tumor cells that are in direct contact with tigilanol tiglate. The oncolysis occurs within the first hours following treatment and results from the disruption of mitochondrial functioning. Secondly, at the same time, tigilanol tiglate activates a protein kinase C (PKC) signaling cascade which propagates throughout the tumor, resulting in an acute inflammatory response with swelling and erythema extending to the tumor margins and immediate surroundings. This inflammatory response is normal and necessarily contributes to the activity of tigilanol tiglate by (a) restricting blood and oxygen supply to the tumor (causing localized hypoxia) and (b) recruiting and activating innate immune cells (principally neutrophils and macrophages), which then target the tumor and release reactive oxygen species, proteases, and cytokines that function in an antimicrobial role. This acute inflammatory response generally resolves within 48 to 96 hours. The third component of the antitumor activity of tigilanol tiglate is associated with direct effects of the drug in increased permeability of the tumor vasculature (via activation of the Beta-II isoform of PKC) leading to tumor vascular destruction. The resulting outcome is tumor destruction with a deficit or wound remaining where the tumor was located. Complete healing of the resulting wound following tumor destruction by STELFONTA is typically within 6 weeks.
Pharmacokinetics
Pharmacokinetic properties of STELFONTA were evaluated in a pilot study monitoring systemic levels following intratumoral injection, with a dose delivered according to the size of the mast cell tumor. A dose of 0.5 mg/cm3 (0.5 mL/cm3) was used in dogs with tumor volumes ranging from 0.1 to 6.8 cm3 resulting in doses ranging from 0.002 mg/kg to 0.145 mg/kg and total doses ranging from 0.05 mg to 3.4 mg per dog. A total of 6 cutaneous and 5 subcutaneous mast cell tumors were treated in 10 dogs (one dog had two tumors treated consecutively). The following range of pharmacokinetic parameters were determined for STELFONTA in plasma: 1) elimination half-life (t½): 2.85 to 36.87 hours; 2) maximum plasma concentration (Cmax): 0.356 ng/mL to 13.8 ng/mL; and 3) area under the plasma concentration time-curve to the last quantifiable plasma concentration (AUClast): 2.25 h*ng/mL to 31.24 h*ng/mL. There was no relationship between drug exposure (Cmax and AUClast) with tumor location (cutaneous or subcutaneous) or with total dose. In an evaluation of the pharmacokinetic data from the 5 dogs with cutaneous tumors, dose levels ranged from 0.002 mg/kg to 0.145 mg/kg. The highest Cmax was 11.1 ng/mL and the highest AUClast was 31.24 h*ng/mL at a dose of 0.125 mg/kg. For the other 5 dogs with subcutaneous tumors, doses ranged from 0.049 mg/kg to 0.094 mg/kg. The highest Cmax was 13.8 ng/mL and the highest AUClast was 30.81 h*ng/mL at a dose of 0.094 mg/kg.
-
EFFECTIVENESS
The effectiveness of STELFONTA was evaluated in a well-controlled, multi-center, randomized, double-masked, field study in client-owned dogs. Enrolled dogs had non-metastatic World Health Organization stages Ia (one tumor confined to the dermis, without regional lymph node involvement) and IIIa (multiple dermal tumors; large infiltrating tumors without regional lymph node involvement) mast cell tumors that were (i) cutaneous, or (ii) subcutaneous and located at or distal to the elbow or the hock). A total of 123 client-owned dogs with a mast cell tumor
measuring less than or equal to 10 cm3 were randomized to treatment with a single injection of STELFONTA (n=81) or untreated control (n=42). On the day of treatment, the average tumor volume was 1.7 cm3 (range 0.1 to 9.8 cm3).A total of 118 dogs were included in the effectiveness analysis; 80 dogs were in the STELFONTA group and 38 dogs were in the untreated control group. Response to treatment was evaluated using the RECIST2, where complete response (CR) is resolution of the target tumor, partial response (PR) is at least a 30% decrease in the longest diameter of target tumor, stable disease (SD) is a decrease of less than 30% or increase of less than 20% of the longest diameter of the target tumor, and progressive disease (PD) is greater than a 20% increase in the longest diameter of the target tumor.
The primary effectiveness variable compared CR rates of the target tumor between groups 28 days after treatment. At 28 days after treatment, a statistically significantly greater proportion of dogs in the STELFONTA treated group (60/80; 75%) achieved CR compared to dogs in the untreated control group (2/38; 5.3%) (p<0.0001). An objective tumor response (CR + PR) was observed in 64/80 (80%) of the STELFONTA treated dogs. Of the 60 dogs in the STELFONTA group that experienced CR at Day 28, response assessment was conducted for 59 dogs at Day 42 and for 57 dogs at Day 84. At Day 42, 59/59 (100%) were disease-free at the injection site, and at Day 84, 55/57 (96%) were disease-free at the injection site.
For all dogs, corticosteroids (prednisone or prednisolone) were initiated 2 days prior to treatment at a dose of 0.5 mg/kg orally twice daily and continued for 7 days total (2 days before, on the day of treatment and 4 days after treatment), then 0.5 mg/kg once daily for an additional 3 days. An H1 receptor blocking agent (diphenhydramine [2 mg/kg orally twice daily]) and H2 receptor blocking agent (famotidine [0.5 mg/kg orally twice daily]) were initiated on the day of treatment and continued for 7 days.
Other medications prescribed based on veterinary discretion included antibiotics, analgesics, and sedatives. The majority of antibiotics were used to treat injection site infections. The majority of analgesics were used to treat tumor pain and were mainly initiated on the day of or day after treatment. Sedatives were used for treatment administration, conducting diagnostics, anxiety, and temperament issues.
Quality of Life (QoL)3 was assessed by owners throughout the study and the mean scores for the QoL assessment was similar between the STELFONTA and untreated control groups at all time points.
Eighteen of the 20 STELFONTA treated dogs without CR received a second treatment. Twenty-eight days following the second treatment, CR was observed in 8/18 (44.4%) of these dogs. Forty-two days following the second treatment, CR was observed in 7/18 (38.9%) of treated dogs.
TARGET ANIMAL SAFETY
The margin of safety and toxicity of STELFONTA was evaluated in one laboratory safety study and one laboratory cardiovascular study utilizing final market formulation, and one pilot field study that used non-commercial formulation.
-
Laboratory Safety Study
In a 4-week laboratory safety study, 48 healthy Beagle dogs 6 to 8 months old were administered STELFONTA intravenously over a 15-minute infusion once a week for four weeks on Days 1, 8, 15, and 22, at doses of 0, 0.025, 0.05, or 0.075 mg/kg body weight (ranges between 0.02-0.036, 0.039-0.056, and 0.06-0.08 mg/kg, respectively due to dosing variability). Control dogs (0 mg/kg) received a vehicle control at a volume equal to the 0.075 mg/kg dose. The intravenous route was chosen for this study because subcutaneous injection was too toxic and intratumoral administration was not possible.
There were twelve dogs per group (6 male, 6 female). Four dogs/sex/group were necropsied two days following the last dose and two dogs/sex/group were necropsied following a 2-week recovery period.
All dogs survived the study, and there were no STELFONTA-related effects on body weight, body temperature, ophthalmic exam, electrocardiographic parameters, and organ weights.
The following were observed only in dogs in the groups administered STELFONTA: decreased food consumption from Days 22-29, vomiting/retching during infusion or immediately post-infusion, wound formation at the infusion site after the second or third dose, decrease in activity sporadically throughout the study, and elevations in alanine aminotransferase on Day 23. The following were observed in all groups, including vehicle control and increased in a dose dependent manner: limited use of the leg that received the infusion occurred soon after dosing, weakness after the first dose, salivation and infusion site edema and erythema increased in frequency and severity throughout the study, and tremors occurred immediately post-infusion and increased in severity with dose.
Vomiting, retching, or tremors were typically transient and resolved within 1 hour of dosing while salivation also typically resolved within 4 hours. Loose feces were observed in all groups in a non-dose dependent manner. Polydipsia occurred in the control, 0.05 and 0.075 mg/kg groups. Trending towards decreasing hematocrit (but still within reference intervals) was observed in all groups. One dog in the 0.05 mg/kg group was mildly anemic during recovery. Monocytosis and elevated fibrinogen were seen on Days 2 and 23 in a dosedependent manner.
Gross pathology findings at the infusion site included inflammation, redness, and thickening of the skin. Correlative histopathology findings of the infusion site included hemorrhage, edema, inflammation, mixed cell infiltration, fibrosis, and chronic organizing thrombosis. Only one of the recovery dogs had changes at the infusion site consisting of proliferation of the intima. One dog in the 0.075 mg/kg group had a severe wound, confirmed on histopathology as ulcerative inflammation and severe necrosis with bacteria present. Gross pathology findings also included red, mottled, firm, and enlarged lymph nodes in all dose groups, including recovery dogs, confirmed on histopathology as inflammation, lymphoid hypercellularity, hemorrhage, and sinus histiocytosis. Pituitary cysts were observed in 7 dogs in all STELFONTA treated groups. One dog each from the 0.075 mg/kg group was observed to have kidney tubular vacuolation, dilation of the ventricles of the brain, and chronic inflammation of both the left thigh skeletal muscle and left sciatic nerve.
Laboratory Cardiovascular Study
In a 12-day laboratory cardiovascular study, 4 healthy male conscious telemeterized Beagle dogs approximately 2-4 years old were administered STELFONTA as a single intravenous infusion. Treatment consisted of four groups: vehicle control and STELFONTA at doses of 0.01, 0.025 and 0.075 mg/kg body weight. All four dogs received all treatments with at least a 3-day wash-out period.
All dogs survived the study and there were no STELFONTA-related effects on body temperatures, blood pressure, or electrocardiograms. The following were observed only after administration of STELFONTA in all dose groups: salivation, vocalization, incoordination, tremors, red feces, and decreased feces output. Retching, vomiting, incoordination, and changes in activity levels (increased and decreased) occurred in the 0.075 mg/kg group only. Tachycardia was seen for the first 2.5 hours after the 0.075 mg/kg dose only. The following were observed after administration of control or STELFONTA: excessive panting, decreased appetite, and limited usage/swelling of leg or paw. All dogs lost weight during the study. Clinical signs resolved around 4 hours post dosing.
Pilot Field Study
In a 28-day unmasked field study, 10 client-owned dogs, 6-14 years old were administered tigilanol tiglate (non-commercial formulation) once as an intratumoral injection at a dose of 0.5 mg tigilanol tiglate per cubic centimeter (cm3) of tumor volume, not exceeding 0.25 mg/kg body weight (maximum dose of 5 mg). One dog was enrolled a second time to treat a second mast cell tumor after successful treatment of the first tumor. See pharmacokinetic results from this study under Clinical Pharmacology.
The most common observations after tigilanol tiglate administration were injection site reactions including necrosis, swelling (localized edema and edema extending well beyond the tumor injection site), pain, restlessness, inflammation, erythema, bleeding ulcerations, bruising/discoloration, sloughing of tissue, open wound, mild drainage, malodor, and presence of granulation tissue. Three dogs experienced dermatitis with or without skin necrosis in a region nearby but distinct from the tumor injection site. One dog experienced non-weight bearing lameness, muscle atrophy and enlarged popliteal lymph node. One dog vomited after administration. Three dogs required longer healing times beyond 28 days, with the longest requiring 5 months. Hypoalbuminemia was observed in 5 dogs with hypoproteinemia observed in 1 of these 5 dogs on Day 7 and was resolved by Day 28.
- STORAGE INFORMATION
- HOW SUPPLIED
-
REFERENCES
1. Veterinary Cooperative Oncology Group – common terminology criteria for adverse events (VCOG-CTCAE) following chemotherapy or biologic antineoplastic therapy in dogs and cats v1.1. Vet Compar Oncol. 20 Jul 2011.
2. Eisenhauer EA, Therase P, Bogaerts J, Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S, Mooney M, Rubinstein L, Shankar L, Dodd L, Kaplan R, Lacombe D, Verweij J. New response evaluation criteria in solid tumors: revised RECIST guideline (version 1.1), Eur J Cancer. 2009; 45(2):228-247.
3. Lynch S, Savary-Bataille K, Leeuw B, Argyle DJ. Development of a questionnaire assessing health-related quality-of-life in dogs and cats with cancer. Vet Compar Oncol. 2011; 9 (3):172-82.
Approved by FDA under NADA # 141-541
©2025 QBiotics Group of companies. All Rights Reserved.
STELFONTA is a registered trademark of QBiotics Group of
companies. Used under license.
Distributed by Virbac AH, Inc., P.O. Box 162059, Fort Worth, TX 76161
Tel. 1-800-338-3659PC5111C
Version date: September 2025A-IN-001.03
- Client Information Sheet
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
STELFONTA 1 MG/ML
tigilanol tiglate injection solutionProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC: 51311-993 Route of Administration intratumor Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TIGILANOL TIGLATE (UNII: R1ZJT87990) (TIGILANOL TIGLATE - UNII:R1ZJT87990) TIGILANOL TIGLATE 1 mg in 1 mg Product Characteristics Color white (white to off white) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 51311-993-02 1 mg in 1 VIAL, SINGLE-USE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NADA NADA141541 02/01/2021 Labeler - Virbac AH Inc (131568396) Registrant - QBiotics Group LTD (744550801)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.