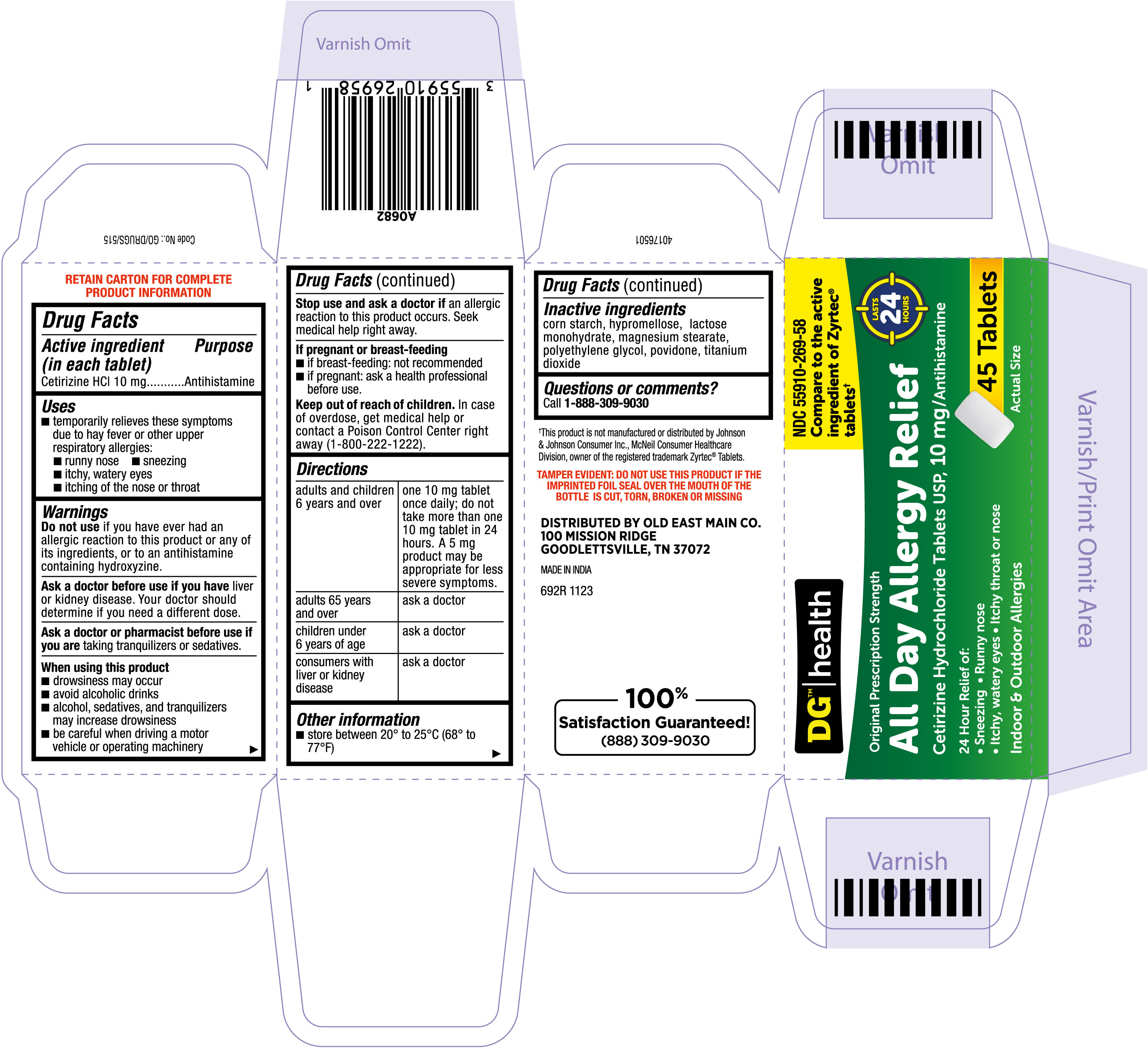
ALL DAY ALLERGY RELIEF- cetirizine hydrochloride tablet
All Day Allergy Relief by
Drug Labeling and Warnings
All Day Allergy Relief by is a Otc medication manufactured, distributed, or labeled by DOLGENCORP, INC., TIME CAP LABORATORIES, INC., MARKSANS PHARMA LIMITED. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Drug Facts
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
- DO NOT USE
- ASK DOCTOR
- ASK DOCTOR/PHARMACIST
- WHEN USING
- STOP USE
- PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
-
DOSAGE & ADMINISTRATION
Directions
adults and children 6 years and over one 10 mg tablet once daily; do not take more than one 10 mg tablet in 24 hours. A 5 mg product may be appropriate for less severe symptoms. adults 65 years and over ask a doctor
children under 6 years of age ask a doctor
consumers with liver or kidney disease ask a doctor
- OTHER SAFETY INFORMATION
- INACTIVE INGREDIENT
- QUESTIONS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ALL DAY ALLERGY RELIEF
cetirizine hydrochloride tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 55910-269 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CETIRIZINE HYDROCHLORIDE (UNII: 64O047KTOA) (CETIRIZINE - UNII:YO7261ME24) CETIRIZINE HYDROCHLORIDE 10 mg Inactive Ingredients Ingredient Name Strength HYPROMELLOSES (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POVIDONE (UNII: FZ989GH94E) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) STARCH, CORN (UNII: O8232NY3SJ) Product Characteristics Color white (White to off white) Score no score Shape RECTANGLE (Rounded-off, rectangular shaped tablet) Size 9mm Flavor Imprint Code J;220 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 55910-269-58 1 in 1 CARTON 12/20/2023 1 45 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA078933 12/20/2023 Labeler - DOLGENCORP, INC. (068331990) Registrant - TIME CAP LABORATORIES, INC. (037052099) Establishment Name Address ID/FEI Business Operations MARKSANS PHARMA LIMITED 925822975 manufacture(55910-269)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.