Duramycin-100Oxytetracycline Hydrochloride Injection
Duramycin 100 by
Drug Labeling and Warnings
Duramycin 100 by is a Animal medication manufactured, distributed, or labeled by Durvet, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
DURAMYCIN 100- oxytetracycline hydrochloride injection, solution
Durvet, Inc.
----------
Duramycin-100
Oxytetracycline Hydrochloride Injection
ANTIBIOTIC
Each mL contains:
100 mg Oxytetracycline HCl
For use in Beef Cattle, Beef Calves, Non-lactating Dairy Cattle and Dairy Calves Only
DESCRIPTION
Duramycin-100 [oxytetracycline hydrochloride] is a sterile ready-to-use preparation containing 100 mg/mL oxytetracycline HCl, for administration of the broad spectrum antibiotic, oxytetracycline, by injection.
ANTIBIOTIC ACTION OF OXYTETRACYCLINE
Oxytetracycline is effective against a wide range of gram-negative and gram-positive organisms that are pathogenic for cattle. The antibiotic is primarily bacteriostatic in effect, and is believed to exert its antimicrobial action by the inhibition of microbial protein synthesis. The antibiotic activity of oxytetracycline is not appreciably diminished in the presence of body fluids, serum or exudates. Since the drugs in the tetracycline class have similar antimicrobial spectra, organisms can develop cross resistance among them. Oxytetracycline is concentrated by the liver in the bile and excreted in the urine and feces at high concentrations and in a biologically active form.
WARNING
Discontinue treatment with Duramycin-100 at least 22 days prior to slaughter of the animal. Not for use in lactating dairy animals.
A withdrawal period has not been established for this product in pre-ruminating calves. Do not use in calves to be processed for veal.
CAUTION
Rapid intravenous administration may result in animal collapse. Oxytetracycline should be administered intravenously slowly over a period of at least 5 minutes.
If no improvement occurs within 24 to 48 hours, consult a veterinarian. Do not use the drug for more than 4 consecutive days. Use beyond 4 days or doses higher than maximum recommended dose may result in antibiotic tissue residues beyond the withdrawal period.
PRECAUTIONS
The improper or accidental injection of the drug outside of the vein will cause local tissue imitation manifested by temporary swelling and discoloration at the injection site.
Shortly after injection, treated animals may have a transient hemoglobinuria (darkened urine).
Consult with your veterinarian prior to administering this product in order to determine the proper treatment required in the event of an adverse reaction. At the first sign of any adverse reaction, discontinue use of product and seek the advice of your veterinarian. Some of the reactions may be attributed either to anaphylaxis (an allergic reaction) or to cardiovascular collapse of unknown cause.
Because bacteriostatic drugs interfere with the bactericidal action of penicillin, do not give oxytetracyeline hydrochloride in conjunction with penicillin.
As with other antibiotics, use of this drug may result in over-growth of non-susceptible organisms. If any unusual symptoms occur or in the absence of a favorable response following treatment, discontinue use immediately and call a veterinarian.
ADVERSE REACTIONS
Reports of adverse reactions associated with oxytetracycline administration include injection site swelling, restlessness, ataxia, trembling, swelling of eyelids, ears, muzzle, anus and vulva (or scrotum and sheath in males), respiratory abnormalities (labored breathing), frothing at the mouth, collapse and possibly death. Some of these reactions may be attributed either to anaphylaxis (an allergic reaction) or to cardiovascular collapse of unknown cause.
GENERAL INDICATIONS FOR USE
A great many of the pathogens involved in cattle diseases are known to be susceptible to oxytetracycline hydrochloride therapy. Many strains of organisms, however, have shown resistance to oxytetracycline. In the case of certain coliforms, streptococci and staphylococci, it may be advisable to conduct culture and sensitivity testing to determine susceptibility of the infecting organism to oxytetracycline. In this manner, the likelihood of successful treatment with Duramycin-100 solution can be determined in advance.
DISEASES FOR WHICH Duramycin-100 IS INDICATED
| Disease | Causative organism(s) which show sensitivity to Duramycin-100 |
| Bacterial Pneumonia and Shipping Fever complex associated with Pasteurella spp. | Pasteurella spp |
| Bacterial Enteritis (scours) | Escherichia coli |
| Necrotic Pododermatitis (Foot Rot) | Fusobacterium necrophorum |
| Calf Diphtheria | Fusobacterium necrophorum |
| Wooden Tongue | Actinobacillus lignieresi |
| Wound Infections; | Caused by oxytetracyeline-susceptible strains of streptococcal and staphylococcal organisms. |
| Acute Metritis; | |
| Traumatic Injury | |
RECOMMENDED DAILY DOSAGES
Treat at the first clinical signs of disease.
The intravenous injection of 3 to 5 mg of oxytetracyeline hydrochloride per pound of body weight per day (3 to 5 mL per 100 lbs body weight) is the recommended dosage.
Severe foot-rot and severe forms of the indicated diseases should be treated with 5 mg per pound of body weight. Surgical procedures may be indicated in some terms of foot-rot or other conditions.
In disease treatment, the daily dose of Duramycin-100 should be continued 24 to 48 hours following remission of disease symptoms; however, not to exceed a total of 4 consecutive days.
DIRECTIONS FOR MAKING AN INTRAVENOUS INJECTION IN CATTLE
Equipment Recommended
- Choke rope - a rope or cord about 5 feet long, with a loop in one end, to be used as a tourniquet.
- Syringe and needles; gravity flow intravenous set. (See Fig. 1.)
- Use new, very sharp hypodermic needles, 16-gauge, 1½ to 2 inches long. Dull needles will not work. Extra needles should be available in case the one being used becomes clogged.
- Scissors or clippers.
- 70% rubbing alcohol compound or other equally effective antiseptic for disinfecting the skin.
- The medication to be given.
PREPARATION OF EQUIPMENT
Thoroughly clean the needles, syringe and intravenous set and disinfect them by boiling in water for twenty minutes or by immersing in a suitable chemical disinfectant such as 70% alcohol for a period of not less than 30 minutes. Warm the bottle of medication to approximately body temperature and keep warm until used.
It is recommended that the correct dose be diluted in water for injection, sodium chloride injection or other suitable vehicle immediately prior to administration. Doses up to 50 mL may be diluted in 250 mL. Larger doses may be diluted in 500 mL of one of the diluents. Adverse reactions may be minimized and the drug dose can be better regulated by this method of administration.
Avoid touching the needle with the hands at all times.
In case of the syringe method of administration, disinfect the vial cap by wiping with 70% alcohol or other suitable antiseptic. Touching a sterile needle only by the hub, attach it to the syringe and push the plunger down the barrel to empty it of air. Puncture the rubber cap of the vial and withdraw the plunger upward in the syringe to draw up a volume of Duramycin-100, 100 mg/mL of about 5 mL more than is needed for injection. Withdraw from the vial and, pointing the needle upward, remove all air bubbles from the syringe by pushing the plunger upward to the volume required.
If the injection cannot be made immediately, the tip of the needle may be covered with cotton soaked in 70% alcohol to prevent contamination.
PREPARATION OF THE ANIMAL FOR INJECTION
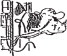
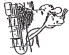
- Approximate location of vein. The jugular vein runs in the jugular groove on each side of the neck from the angle of the jaw to just above the brisket and slightly above and to the side of the windpipe. (See Figure 2 and 3.)
- Method of restraint - A stanchion or chute is ideal for restraining the animal. With a halter, rope or cattle leader (nose tongs), pull the animal's head around the side of the stanchion, cattle chute or post in such a manner as to form a bow in the neck (see Figure 4), then snub the head securely to prevent movement. By forming the bow in the neck, the outside curvature of the bow tends to expose the jugular vein and make it easily accessible. Caution: Avoid a tight rope or halter around the throat or upper neck which might impede blood flow. Animals that are down present no problem as far as restraint is concerned.
- Clip hair in area where injection is to be made (over the vein in the upper third of the neck). Clean and disinfect the skin with alcohol or other suitable antiseptic.
DOSAGE FOR INJECTION
Refer to the table below for proper dosage according to body weight of the animal.
| Weight of Animals, Lbs (Beef Cattle, Beef Calves, Non-Lactating Dairy Cattle, Dairy Calves) | Milligrams of Oxytetracycline Hydrochloride per 100 lbs of Body Weight per Day | Daily Dosage of Duramycin-100 |
| 50 lbs | 300 - 500 mg | 1.5 - 2.5 mL |
| 100 lbs | 300 - 500 mg | 3 - 5 mL |
| 200 lbs | 300 - 500 mg | 6 - 10 mL |
| 300 lbs | 300 - 500 mg | 9 - 15 mL |
| 400 lbs | 300 - 500 mg | 12 - 20 mL |
| 500 lbs | 300 - 500 mg | 15 - 25 mL |
| 600 lbs | 300 - 500 mg | 18 - 30 mL |
| 800 lbs | 300 - 500 mg | 24 - 40 mL |
| 1000 lbs | 300 - 500 mg | 30 - 50 mL |
| 1200 lbs | 300 - 500 mg | 36 - 60 mL |
| 1400 lbs | 300 - 500 mg | 42 - 70 mL |
CAUTION: If no improvement is noted within 24 to 48 hours consult a veterinarian.
For intravenous use only.
ENTERING THE VEIN AND MAKING THE INJECTION
- Raise the vein: this is accomplished by tying the choke rope tight around the neck, close to the shoulder. The rope should be tied in such a way that it will not come loose and so that it can be untied quickly by pulling the loose end. (See Figure 4.) In thick-necked animals, a block of wood placed in the jugular groove between the rope and the hide will help considerably in applying the desired pressure at the right point. The vein is a soft flexible tube through which blood flows back to the heart. Under ordinary conditions it cannot be seen or felt with the fingers. When the flow of blood is blocked at the base of the neck by the choke rope, the vein becomes enlarged and rigid because of the back pressure. If the choke rope is sufficiently tight, the vein stands out and can be easily seen and felt in the thick-necked animals. As a further check in identifying the vein, tap it with the fingers in front of the choke rope. Pulsations that can be seen or felt with the fingers in front of the point being tapped will confirm the fact that the vein is properly distended. It is impossible to put the needle into the vein unless it is distended. Experienced operators are able to raise the vein simply by hand pressure, but the use of a choke rope is more certain.
- Inserting the needle. This involves three distinct steps. First, insert the needle through the hide. Second, insert the needle into the vein. This may require two or three attempts before the vein is entered. The vein has a tendency to roll away from the point of the needle, especially if the needle is not sharp. The vein can be steadied with the thumb and finger of one hand. With the other hand, the needle point is placed directly over the vein, slanting it so that its direction is along the length of the vein, either toward the head or toward the heart. Properly positioned this way, a quick thrust of the needle will be followed by a spurt of blood through the needle, which indicates that the vein has been entered. Third, once in the vein, the needle should be inserted along the length of the vein all the way to the hub, exercising caution to see that the needle does not penetrate to the opposite side of the vein. Continuous steady flow of blood through the needle indicates that the needle is still in the vein. If blood does not flow continuously, the needle is out of the vein (or clogged) and another attempt must be made. If difficulty is encountered, it may be advisable to use the vein on the other side of the neck.
- While the needle is being placed in proper position in the vein, an assistant should get the medication ready so that the injection can be started without delay after the vein has been entered. Remove the rubber stopper from the bottle of intravenous solution, connect the intravenous tube to the neck of the bottle, invert the bottle and allow some of the solution to run through the tube to eliminate all air bubbles.
- Making the injection. With needle in proper position as indicated by a continuous flow of blood, release the choke rope by a quick pull on the free end. This is essential - the medication cannot flow into the vein while the vein is blocked. Immediately connect the intravenous tube to the needle, and raise the bottle. The solution will flow by gravity. (See Figure 5.) Rapid injection may occasionally produce shock. Administer slowly. The animal should be observed at all times during the injection in order not to give the solution too fast. This may be determined by watching the respiration of the animal and feeling or listening to the heart beat. If the heart beat and respiration increase markedly, the rate of injection should be immediately stopped by pinching the tube until the animal recovers approximately to its previous respiration or heart beat rate, when the injection can be resumed at a slower rate. The rate of flow can be controlled by pinching the tube between the thumb and forefinger or by raising or lowering the bottle.
Bubbles entering the bottle through the air tube or valve indicate the rale at which the medication is flowing, if the flow should stop, this means that the needle has slipped out of the vein (or is clogged) and the operation will have to be repeated. If using the syringe technique, pull back gently on the plunger: if blood flows into the syringe, the needle is in proper position. Depress the plunger slowly. If there is any resistance to the depression of the plunger, stop and repeat insertion procedure. The resistance indicates that either the needle is clogged or it has slipped out of the vein. With either method of administration, syringe or gravity flow, watch for any swelling under the skin near the needle, which would indicate that the medication is not going into the vein. Should this occur, it is best to try the vein on the opposite side of the neck. Sudden movement of the animal, especially twisting of the neck or raising or lowering the head, may sometimes cause the needle to slip out of the vein. To prevent this, tape the needle hub to the skin of the neck to hold the needle in position. Whenever there is any doubt as to the position of the needle, this should be checked in the following manner: Pinch off the intravenous tube to stop flow, disconnect the tube from the needle and re-apply pressure to the vein. Free flow of blood through the needle indicates that it is in proper position and the injection can then be continued. If using the syringe, gently pull back on the plunger. Blood should flow into the syringe.
- Removing the needle. When the injection is complete, remove needle with a straight pull. Then apply pressure over the area of injection momentarily to control any bleeding through needle puncture, using cotton soaked in alcohol or other suitable antiseptic.
INSTRUCTIONS FOR CARE OF SICK ANIMALS
The use of antibiotics, as with most medications used in the management of diseases, is based on accurate diagnosis and adequate treatment. When properly used in the treatment of diseases caused by oxytetracycline-susceptible organisms, animals usually show a noticeable improvement within 24 to 48 hours. If improvement does not occur within this period of time, the diagnosis and treatment of animal diseases should be carried out by a veterinarian. The use of professional veterinary and laboratory services can reduce treatment costs, time and needless losses. Good management, housing, sanitation and nutrition are essential in the care of animals and in the successful treatment of disease.
PACKAGE INFORMATION
Duramycin-100 is available in 500 mL multidose vials containing 100 mg oxytetracycline hydrochloride per mL.
Store at controlled room temperature 20°-25°C (68°-77°F); excursions permitted 15°-30°C (59°-86°F). Protect from freezing.
For Use In Animals Only
RESTRICTED DRUG (California) - USE ONLY
AS DIRECTED
022215I02
Principal Display Panel - Duramycin 100 Carton Label
NDC: 30798-244-17
durvet
Duramycin-100
(Oxytetracycline
Hydrochloride Injection)
ANTIBIOTIC
Multiple Dose Sterile Vial (100 mg/mL)
Each mL contains 100 mg of oxytetracycline hydrochloride.
ANADA 200-452, Approved by FDA
For the treatment of disease in beef cattle,
beef calves, non-lactating dairy cattle,
and dairy calves.
NET CONTENTS: 500 mL
Principal Display Panel - Duramycin 100 Vial Label
NDC: 30798-244-17
durvet
Duramycin-100
(Oxytetracycline
Hydrochloride Injection)
ANTIBIOTIC
100 mg/mL Sterile
Restricted Drug (California)
Use Only as Directed
ANADA 200-452, Approved by FDA
NET CONTENTS: 500 mL
| DURAMYCIN 100
oxytetracycline hydrochloride injection, solution |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Durvet, Inc. (056387798) |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.