DESCOVY- emtricitabine and tenofovir alafenamide tablet
DESCOVY by
Drug Labeling and Warnings
DESCOVY by is a Prescription medication manufactured, distributed, or labeled by Gilead Sciences, Inc., Shanghai STA Pharmaceuticals Product Co., Ltd.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use DESCOVY safely and effectively. See full prescribing information for DESCOVY.
DESCOVY® (emtricitabine and tenofovir alafenamide) tablets, for oral use
Initial U.S. Approval: 2015WARNING: POST-TREATMENT ACUTE EXACERBATION OF HEPATITIS B and RISK OF DRUG RESISTANCE WITH USE OF DESCOVY FOR HIV-1 PRE-EXPOSURE PROPHYLAXIS (PrEP) IN UNDIAGNOSED EARLY HIV-1 INFECTION
See full prescribing information for complete boxed warning.
Severe acute exacerbations of hepatitis B (HBV) have been reported in HBV-infected individuals who have discontinued products containing emtricitabine (FTC) and/or tenofovir disoproxil fumarate (TDF), and may occur with discontinuation of DESCOVY. Hepatic function should be monitored closely in these individuals. If appropriate, anti-hepatitis B therapy may be warranted. (5.1)
DESCOVY used for HIV-1 PrEP must only be prescribed to individuals confirmed to be HIV-negative immediately prior to initiating and at least every 3 months during use. Drug-resistant HIV-1 variants have been identified with use of FTC/TDF for HIV-1 PrEP following undetected acute HIV-1 infection. Do not initiate DESCOVY for HIV-1 PrEP if signs or symptoms of acute HIV-1 infection are present unless negative infection status is confirmed. (5.2)
RECENT MAJOR CHANGES
INDICATIONS AND USAGE
HIV-1 Treatment (1.1):
DESCOVY is a two-drug combination of emtricitabine (FTC) and tenofovir alafenamide (TAF), both HIV nucleoside analog reverse transcriptase inhibitors (NRTIs), and is indicated:
- in combination with other antiretroviral agents for the treatment of HIV-1 infection in adults and pediatric patients weighing at least 35 kg.
- in combination with other antiretroviral agents other than protease inhibitors that require a CYP3A inhibitor for the treatment of HIV-1 infection in pediatric patients weighing at least 25 kg and less than 35 kg.
HIV-1 PrEP (1.2):
DESCOVY is indicated in at-risk adults and adolescents weighing at least 35 kg for pre-exposure prophylaxis (PrEP) to reduce the risk of HIV-1 infection from sexual acquisition, excluding individuals at risk from receptive vaginal sex. Individuals must have a negative HIV-1 test immediately prior to initiating DESCOVY for HIV-1 PrEP.
Limitations of Use (1.2):
The indication does not include use of DESCOVY in individuals at risk of HIV-1 from receptive vaginal sex because effectiveness in this population has not been evaluated.
DOSAGE AND ADMINISTRATION
- Testing: Prior to or when initiating DESCOVY, test for hepatitis B virus infection. Prior to or when initiating DESCOVY, and during use on a clinically appropriate schedule, assess serum creatinine, estimated creatinine clearance, urine glucose, and urine protein in all individuals. In individuals with chronic kidney disease, also assess serum phosphorus. (2.1)
- HIV-1 Screening: Screen all individuals for HIV-1 infection immediately prior to initiating DESCOVY for HIV-1 PrEP and at least once every 3 months while taking DESCOVY, and upon diagnosis of any other sexually transmitted infections (STIs). (2.2)
- Recommended dosage:
- Renal impairment: DESCOVY is not recommended in individuals with estimated creatinine clearance of 15 to below 30 mL per minute, or below 15 mL per minute who are not receiving chronic hemodialysis. (2.5)
DOSAGE FORMS AND STRENGTHS
Tablets: 200 mg of FTC and 25 mg of TAF (3)
CONTRAINDICATIONS
DESCOVY for HIV-1 PrEP is contraindicated in individuals with unknown or positive HIV-1 status. (4)
WARNINGS AND PRECAUTIONS
- Comprehensive management to reduce the risk of sexually transmitted infections (STIs), including HIV-1, when DESCOVY is used for HIV-1 PrEP: Counsel on adherence to daily dosing and safer sex practices, including condoms, to reduce the risk of STIs. (5.2)
- Management to reduce the risk of acquiring HIV-1 drug resistance when DESCOVY is used for HIV-1 PrEP: refer to full prescribing information for additional detail. (5.2)
- Immune reconstitution syndrome during treatment of HIV-1 infection: May necessitate further evaluation and treatment. (5.3)
- New onset or worsening renal impairment: Assess serum creatinine, estimated creatinine clearance, urine glucose, and urine protein when initiating DESCOVY and during use on a clinically appropriate schedule in all individuals. Also assess serum phosphorus in individuals with chronic kidney disease. (5.4)
- Lactic acidosis/severe hepatomegaly with steatosis: Discontinue DESCOVY in individuals who develop symptoms or laboratory findings suggestive of lactic acidosis or pronounced hepatotoxicity. (5.5)
ADVERSE REACTIONS
- In HIV-1 infected patients, the most common adverse reaction (incidence greater than or equal to 10%, all grades) was nausea. (6.1)
- In HIV-1 uninfected adults in a PrEP trial, the most common adverse reaction (incidence greater than or equal to 5%, all grades) was diarrhea. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Gilead Sciences, Inc. at 1-800-GILEAD-5 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
USE IN SPECIFIC POPULATIONS
- Lactation: Mothers infected with HIV-1 should be instructed not to breastfeed, due to the potential for HIV transmission. (8.2)
- Pediatrics:
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 1/2020
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: POST-TREATMENT ACUTE EXACERBATION OF HEPATITIS B and RISK OF DRUG RESISTANCE WITH USE OF DESCOVY FOR HIV-1 PRE-EXPOSURE PROPHYLAXIS (PrEP) IN UNDIAGNOSED EARLY HIV-1 INFECTION
1 INDICATIONS AND USAGE
1.1 Treatment of HIV-1 Infection
1.2 HIV-1 Pre-Exposure Prophylaxis (PrEP)
2 DOSAGE AND ADMINISTRATION
2.1 Testing When Initiating and During Use of DESCOVY for Treatment of HIV-1 Infection or for HIV-1 PrEP
2.2 HIV-1 Screening for Individuals Receiving DESCOVY for HIV-1 PrEP
2.3 Recommended Dosage for Treatment of HIV-1 Infection in Adults and Pediatric Patients Weighing at Least 25 kg
2.4 Recommended Dosage for HIV-1 PrEP in Adults and Adolescents Weighing at Least 35 kg
2.5 Not Recommended in Individuals with Severe Renal Impairment for Treatment of HIV-1 Infection or for HIV-1 PrEP
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Severe Acute Exacerbation of Hepatitis B in Individuals with HBV Infection
5.2 Comprehensive Management to Reduce the Risk of Sexually Transmitted Infections, Including HIV-1, and Development of HIV-1 Resistance When DESCOVY Is Used for HIV-1 PrEP
5.3 Immune Reconstitution Syndrome
5.4 New Onset or Worsening Renal Impairment
5.5 Lactic Acidosis/Severe Hepatomegaly with Steatosis
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Potential for Other Drugs to Affect One or More Components of DESCOVY
7.2 Drugs Affecting Renal Function
7.3 Established and Other Potentially Significant Interactions
7.4 Drugs without Clinically Significant Interactions with DESCOVY
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
8.7 Hepatic Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
12.4 Microbiology
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
13.2 Animal Toxicology and/or Pharmacology
14 CLINICAL STUDIES
14.1 Overview of Clinical Trials
14.2 Clinical Trial Results for Treatment of HIV-1
14.3 Clinical Trial Results for HIV-1 PrEP
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: POST-TREATMENT ACUTE EXACERBATION OF HEPATITIS B and RISK OF DRUG RESISTANCE WITH USE OF DESCOVY FOR HIV-1 PRE-EXPOSURE PROPHYLAXIS (PrEP) IN UNDIAGNOSED EARLY HIV-1 INFECTION
Severe acute exacerbations of hepatitis B (HBV) have been reported in HBV-infected individuals who have discontinued products containing emtricitabine (FTC) and/or tenofovir disoproxil fumarate (TDF) and may occur with discontinuation of DESCOVY.
Hepatic function should be monitored closely with both clinical and laboratory follow-up for at least several months in individuals who are infected with HBV and discontinue DESCOVY. If appropriate, anti-hepatitis B therapy may be warranted [see Warnings and Precautions (5.1)].
DESCOVY used for HIV-1 PrEP must only be prescribed to individuals confirmed to be HIV-negative immediately prior to initiating and at least every 3 months during use. Drug-resistant HIV-1 variants have been identified with use of FTC/TDF for HIV-1 PrEP following undetected acute HIV-1 infection. Do not initiate DESCOVY for HIV-1 PrEP if signs or symptoms of acute HIV-1 infection are present unless negative infection status is confirmed [see Warnings and Precautions (5.2)].
-
1 INDICATIONS AND USAGE
1.1 Treatment of HIV-1 Infection
DESCOVY is indicated, in combination with other antiretroviral agents, for the treatment of HIV-1 infection in adults and pediatric patients weighing at least 35 kg.
DESCOVY is indicated, in combination with other antiretroviral agents other than protease inhibitors that require a CYP3A inhibitor, for the treatment of HIV-1 infection in pediatric patients weighing at least 25 kg and less than 35 kg.
1.2 HIV-1 Pre-Exposure Prophylaxis (PrEP)
DESCOVY is indicated in at-risk adults and adolescents weighing at least 35 kg for pre-exposure prophylaxis (PrEP) to reduce the risk of HIV-1 infection from sexual acquisition, excluding individuals at risk from receptive vaginal sex. Individuals must have a negative HIV-1 test immediately prior to initiating DESCOVY for HIV-1 PrEP [see Dosage and Administration (2.2) and Warnings and Precautions (5.2)].
Limitations of Use:
The indication does not include use of DESCOVY in individuals at risk of HIV-1 from receptive vaginal sex because effectiveness in this population has not been evaluated [see Clinical Studies (14.3)].
-
2 DOSAGE AND ADMINISTRATION
2.1 Testing When Initiating and During Use of DESCOVY for Treatment of HIV-1 Infection or for HIV-1 PrEP
Prior to or when initiating DESCOVY, test individuals for hepatitis B virus infection [see Warnings and Precautions (5.1)].
Prior to or when initiating DESCOVY, and during use of DESCOVY on a clinically appropriate schedule, assess serum creatinine, estimated creatinine clearance, urine glucose, and urine protein in all individuals. In individuals with chronic kidney disease, also assess serum phosphorus [see Warnings and Precautions (5.4)].
2.2 HIV-1 Screening for Individuals Receiving DESCOVY for HIV-1 PrEP
Screen all individuals for HIV-1 infection immediately prior to initiating DESCOVY for HIV-1 PrEP and at least once every 3 months while taking DESCOVY, and upon diagnosis of any other sexually transmitted infections (STIs) [see Indications and Usage (1.2), Contraindications (4), and Warnings and Precautions (5.2)].
If recent (<1 month) exposures to HIV-1 are suspected or clinical symptoms consistent with acute HIV-1 infection are present, use a test approved or cleared by the FDA as an aid in the diagnosis of acute or primary HIV-1 infection [see Warnings and Precautions (5.2), Use in Specific Populations (8.4), and Clinical Studies (14.3)].
2.3 Recommended Dosage for Treatment of HIV-1 Infection in Adults and Pediatric Patients Weighing at Least 25 kg
DESCOVY is a two-drug fixed dose combination product containing 200 mg of emtricitabine (FTC) and 25 mg of tenofovir alafenamide (TAF). The recommended dosage of DESCOVY for treatment of HIV-1 is one tablet taken orally once daily with or without food in:
- adults and pediatric patients with body weight at least 25 kg and creatinine clearance greater than or equal to 30 mL per minute; or
- adults with creatinine clearance below 15 mL per minute who are receiving chronic hemodialysis. On days of hemodialysis, administer the daily dose of DESCOVY after completion of hemodialysis treatment [see Use in Specific Populations (8.6) and Clinical Pharmacology (12.3)].
For specific dosing recommendations for coadministered third agents, refer to their respective prescribing information [see Drug Interactions (7)]. The safety and effectiveness of DESCOVY coadministered with an HIV-1 protease inhibitor that is administered with either ritonavir or cobicistat have not been established in:
- pediatric subjects weighing less than 35 kg; or
- adult subjects with creatinine clearance below 15 mL per minute, with or without hemodialysis.
2.4 Recommended Dosage for HIV-1 PrEP in Adults and Adolescents Weighing at Least 35 kg
The dosage of DESCOVY for HIV-1 PrEP is one tablet (containing 200 mg of FTC and 25 mg of TAF) once daily taken orally with or without food in HIV-1 uninfected:
- adults and adolescents weighing at least 35 kg and with a creatinine clearance greater than or equal to 30 mL per minute; or
- adults with creatinine clearance below 15 mL per minute who are receiving chronic hemodialysis. On days of hemodialysis, administer the daily dose of DESCOVY after completion of hemodialysis treatment [see Indications and Usage (1.2) and Clinical Pharmacology (12.3)].
2.5 Not Recommended in Individuals with Severe Renal Impairment for Treatment of HIV-1 Infection or for HIV-1 PrEP
DESCOVY is not recommended in individuals with:
- severe renal impairment (estimated creatinine clearance of 15 to below 30 mL per minute); or
- end stage renal disease (ESRD; estimated creatinine clearance below 15 mL per minute) who are not receiving chronic hemodialysis [see Dosage and Administration (2.3, 2.4) and Use in Specific Populations (8.6)].
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
DESCOVY for HIV-1 PrEP is contraindicated in individuals with unknown or positive HIV-1 status [see Warnings and Precautions (5.2)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Severe Acute Exacerbation of Hepatitis B in Individuals with HBV Infection
All individuals should be tested for the presence of hepatitis B virus (HBV) before or when initiating DESCOVY [see Dosage and Administration (2.1)].
Severe acute exacerbations of hepatitis B (e.g., liver decompensation and liver failure) have been reported in HBV-infected individuals who have discontinued products containing FTC and/or tenofovir disoproxil fumarate (TDF) and may occur with discontinuation of DESCOVY. Individuals infected with HBV who discontinue DESCOVY should be closely monitored with both clinical and laboratory follow-up for at least several months after stopping treatment. If appropriate, anti-hepatitis B therapy may be warranted, especially in individuals with advanced liver disease or cirrhosis, since post-treatment exacerbation of hepatitis may lead to hepatic decompensation and liver failure. HBV-uninfected individuals should be offered vaccination.
5.2 Comprehensive Management to Reduce the Risk of Sexually Transmitted Infections, Including HIV-1, and Development of HIV-1 Resistance When DESCOVY Is Used for HIV-1 PrEP
Use DESCOVY for HIV-1 PrEP to reduce the risk of HIV-1 infection as part of a comprehensive prevention strategy, including adherence to daily administration and safer sex practices, including condoms, to reduce the risk of sexually transmitted infections (STIs). The time from initiation of DESCOVY for HIV-1 PrEP to maximal protection against HIV-1 infection is unknown.
Risk for HIV-1 acquisition includes behavioral, biological, or epidemiologic factors including but not limited to condomless sex, past or current STIs, self-identified HIV risk, having sexual partners of unknown HIV-1 viremic status, or sexual activity in a high prevalence area or network.
Counsel individuals on the use of other prevention measures (e.g., consistent and correct condom use, knowledge of partner(s)' HIV-1 status, including viral suppression status, regular testing for STIs that can facilitate HIV-1 transmission). Inform uninfected individuals about and support their efforts in reducing sexual risk behavior.
Use DESCOVY to reduce the risk of acquiring HIV-1 only in individuals confirmed to be HIV-1 negative. HIV-1 resistance substitutions may emerge in individuals with undetected HIV-1 infection who are taking only DESCOVY, because DESCOVY alone does not constitute a complete regimen for HIV-1 treatment [see Microbiology (12.4)]; therefore, care should be taken to minimize the risk of initiating or continuing DESCOVY before confirming the individual is HIV-1 negative.
- Some HIV-1 tests only detect anti-HIV antibodies and may not identify HIV-1 during the acute stage of infection. Prior to initiating DESCOVY for HIV-1 PrEP, ask seronegative individuals about recent (in past month) potential exposure events (e.g., condomless sex or condom breaking during sex with a partner of unknown HIV-1 status or unknown viremic status, or a recent STI), and evaluate for current or recent signs or symptoms consistent with acute HIV-1 infection (e.g., fever, fatigue, myalgia, skin rash).
- If recent (<1 month) exposures to HIV-1 are suspected or clinical symptoms consistent with acute HIV-1 infection are present, use a test approved or cleared by the FDA as an aid in the diagnosis of acute or primary HIV-1 infection.
While using DESCOVY for HIV-1 PrEP, HIV-1 testing should be repeated at least every 3 months, and upon diagnosis of any other STIs.
- If an HIV-1 test indicates possible HIV-1 infection, or if symptoms consistent with acute HIV-1 infection develop following a potential exposure event, convert the HIV-1 PrEP regimen to an HIV treatment regimen until negative infection status is confirmed using a test approved or cleared by the FDA as an aid in the diagnosis of acute or primary HIV-1 infection.
Counsel HIV-1 uninfected individuals to strictly adhere to the once daily DESCOVY dosing schedule. The effectiveness of DESCOVY in reducing the risk of acquiring HIV-1 is strongly correlated with adherence, as demonstrated by measurable drug levels in a clinical trial of DESCOVY for HIV-1 PrEP. Some individuals, such as adolescents, may benefit from more frequent visits and counseling to support adherence [see Use in Specific Populations (8.4), Microbiology (12.4), and Clinical Studies (14.3)].
5.3 Immune Reconstitution Syndrome
Immune reconstitution syndrome has been reported in HIV-1 infected patients treated with combination antiretroviral therapy, including FTC, a component of DESCOVY. During the initial phase of combination antiretroviral treatment, HIV-1 infected patients whose immune system responds may develop an inflammatory response to indolent or residual opportunistic infections (such as Mycobacterium avium infection, cytomegalovirus, Pneumocystis jirovecii pneumonia [PCP], or tuberculosis), which may necessitate further evaluation and treatment.
Autoimmune disorders (such as Graves' disease, polymyositis, Guillain-Barré syndrome, and autoimmune hepatitis) have also been reported to occur in the setting of immune reconstitution; however, the time to onset is more variable, and can occur many months after initiation of treatment.
5.4 New Onset or Worsening Renal Impairment
Renal impairment, including cases of acute renal failure and Fanconi syndrome (renal tubular injury with severe hypophosphatemia), has been reported with the use of tenofovir prodrugs in both animal toxicology studies and human trials. In clinical trials of FTC+TAF with cobicistat (COBI) plus elvitegravir (EVG) in HIV-1 infected patients there have been no cases of Fanconi syndrome or Proximal Renal Tubulopathy (PRT). In clinical trials of FTC+TAF with EVG+COBI in treatment-naïve subjects and in virally suppressed subjects switched to FTC+TAF with EVG+COBI with estimated creatinine clearance greater than 50 mL per minute, renal serious adverse events or discontinuations due to renal adverse reactions were encountered in less than 1% of participants treated with FTC+TAF with EVG+COBI. In a study of virally suppressed subjects with baseline estimated creatinine clearance between 30 and 69 mL per minute treated with FTC+TAF with EVG+COBI for a median duration of 43 weeks, FTC+TAF with EVG+COBI was permanently discontinued due to worsening renal function in two of 80 (3%) subjects with a baseline estimated creatinine clearance between 30 and 50 mL per minute [see Adverse Reactions (6.1)]. DESCOVY is not recommended in individuals with estimated creatinine clearance of 15 to below 30 mL per minute, or in individuals with estimated creatinine clearance below 15 mL per minute who are not receiving chronic hemodialysis.
Individuals taking tenofovir prodrugs who have impaired renal function and those taking nephrotoxic agents including non-steroidal anti-inflammatory drugs are at increased risk of developing renal-related adverse reactions.
Prior to or when initiating DESCOVY, and during treatment with DESCOVY on a clinically appropriate schedule, assess serum creatinine, estimated creatinine clearance, urine glucose, and urine protein in all individuals. In individuals with chronic kidney disease, also assess serum phosphorus. Discontinue DESCOVY in individuals who develop clinically significant decreases in renal function or evidence of Fanconi syndrome.
5.5 Lactic Acidosis/Severe Hepatomegaly with Steatosis
Lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been reported with the use of nucleoside analogs, including emtricitabine, a component of DESCOVY, and tenofovir DF, another prodrug of tenofovir, alone or in combination with other antiretrovirals. Treatment with DESCOVY should be suspended in any individual who develops clinical or laboratory findings suggestive of lactic acidosis or pronounced hepatotoxicity (which may include hepatomegaly and steatosis even in the absence of marked transaminase elevations).
-
6 ADVERSE REACTIONS
The following adverse reactions are discussed in other sections of the labeling:
- Severe Acute Exacerbations of Hepatitis B [see Warnings and Precautions (5.1)].
- Immune Reconstitution Syndrome [see Warnings and Precautions (5.3)].
- New Onset or Worsening Renal Impairment [see Warnings and Precautions (5.4)].
- Lactic Acidosis/Severe Hepatomegaly with Steatosis [see Warnings and Precautions (5.5)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug (or a drug given in various combinations with other concomitant therapy) cannot be directly compared to rates in the clinical trials of another drug (or drug given in the same or different combination therapy) and may not reflect the rates observed in practice.
Adverse Reactions in Clinical Trials of FTC+TAF with EVG+COBI in Treatment-Naïve Adults with HIV-1 Infection
In pooled 48-week trials of antiretroviral treatment-naïve HIV-1 infected adult subjects, the most common adverse reaction in subjects treated with FTC+TAF with EVG+COBI (N=866) (incidence greater than or equal to 10%, all grades) was nausea (10%). In this treatment group, 0.9% of subjects discontinued FTC+TAF with EVG+COBI due to adverse events during the 48-week treatment period [see Clinical Studies (14.2)]. The safety profile was similar in virologically-suppressed adults with HIV-1 infection who were switched to FTC+TAF with EVG+COBI (N=799). Antiretroviral treatment-naïve adult subjects treated with FTC+TAF with EVG+COBI experienced mean increases of 30 mg/dL of total cholesterol, 15 mg/dL of LDL cholesterol, 7 mg/dL of HDL cholesterol, and 29 mg/dL of triglycerides after 48 weeks of use.
Renal Laboratory Tests
In two 48-week trials in antiretroviral treatment-naïve HIV-1 infected adults treated with FTC+TAF with EVG+COBI (N=866) with a median baseline eGFR of 115 mL per minute, mean serum creatinine increased by 0.1 mg per dL from baseline to Week 48. Median urine protein-to-creatinine ratio (UPCR) was 44 mg per gram at baseline and at Week 48. In a 48-week trial in virologically-suppressed TDF-treated adults who switched to FTC+TAF with EVG+COBI (N=959) with a mean baseline eGFR of 112 mL per minute, mean serum creatinine was similar to baseline at Week 48; median UPCR was 61 mg per gram at baseline and 46 mg per gram at Week 48. In a 24-week trial in adults with renal impairment (baseline eGFR 30 to 69 mL per minute) who received FTC+TAF with EVG+COBI (N=248), mean serum creatinine was 1.5 mg per dL at both baseline and Week 24. Median UPCR was 161 mg per gram at baseline and 93 mg per gram at Week 24.
Bone Mineral Density Effects
In the pooled analysis of two 48-week trials of antiretroviral treatment-naïve HIV-1 infected adult subjects, bone mineral density (BMD) from baseline to Week 48 was assessed by dual-energy X-ray absorptiometry (DXA). Mean BMD decreased from baseline to Week 48 –1.30% with FTC+TAF with EVG+COBI at the lumbar spine and –0.66% at the total hip. BMD declines of 5% or greater at the lumbar spine were experienced by 10% of FTC+TAF with EVG+COBI subjects. BMD declines of 7% or greater at the femoral neck were experienced by 7% of FTC+TAF with EVG+COBI subjects. The long-term clinical significance of these BMD changes is not known.
In 799 virologically-suppressed TDF-treated adult subjects that switched to FTC+TAF with EVG+COBI, at Week 48 mean BMD increased (1.86% lumbar spine, 1.95% total hip). BMD declines of 5% or greater at the lumbar spine were experienced by 1% of FTC+TAF with EVG+COBI subjects. BMD declines of 7% or greater at the femoral neck were experienced by 1% of FTC+TAF with EVG+COBI subjects.
Adverse Reactions in a Clinical Trial of FTC+TAF with EVG+COBI in Virologically-Suppressed Adults with End Stage Renal Disease (ESRD) Receiving Chronic Hemodialysis
In a 48-week trial of virologically-suppressed HIV-1 infected adult subjects with end stage renal disease (ESRD) (estimated creatinine clearance of less than 15 mL/min) on chronic hemodialysis treated with FTC+TAF with EVG+COBI (N=55), the most commonly reported adverse reaction (adverse event assessed as causally related by investigator and all grades) was nausea (7%). Serious adverse events were reported in 53% of subjects and the most common serious adverse events were pneumonia (13%), fluid overload (7%), hyperkalemia (7%) and osteomyelitis (7%). Overall 5% of subjects permanently discontinued treatment due to an adverse event.
Adverse Reactions in Clinical Trials in Pediatric Subjects with HIV-1 Infection
In an open-label trial of antiretroviral treatment-naïve HIV-1 infected pediatric subjects between the ages of 12 to less than 18 years weighing at least 35 kg through 48 weeks (N=50; Cohort 1) and virologically-suppressed subjects between the ages of 6 to less than 12 years weighing at least 25 kg (N=23; Cohort 2) who received FTC+TAF with EVG+COBI through 24 weeks, with the exception of a decrease in the mean CD4+ cell count observed in cohort 2, the safety of this combination was similar to that of adults.
Bone Mineral Density Effects
Cohort 1: Treatment-naïve adolescents (12 to less than 18 years; at least 35 kg)
Among the subjects in cohort 1 receiving FTC+TAF with EVG+COBI, mean BMD increased from baseline to Week 48, +4.2% at the lumbar spine and +1.3% for the total body less head (TBLH). Mean changes from baseline BMD Z-scores were –0.07 for lumbar spine and –0.20 for TBLH at Week 48. One subject had significant (at least 4%) lumbar spine BMD loss at Week 48.
Cohort 2: Virologically-suppressed children (6 to less than 12 years; at least 25 kg)
Among the subjects in cohort 2 receiving FTC+TAF with EVG+COBI, mean BMD increased from baseline to Week 24, +2.9% at the lumbar spine and +1.7% for TBLH. Mean changes from baseline BMD Z-scores were -0.06 for lumbar spine and -0.18 for TBLH at Week 24. Two subjects had significant (at least 4%) lumbar spine BMD loss at Week 24.
Change from Baseline in CD4+ cell counts
Cohort 2: Virologically-suppressed children (6 to less than 12 years; at least 25 kg)
Cohort 2 evaluated pediatric subjects (N=23) who were virologically-suppressed and who switched from their antiretroviral regimen to FTC+TAF with EVG+COBI. Although all subjects had HIV-1 RNA < 50 copies/mL, there was a decrease from baseline in CD4+ cell count at Week 24. The mean baseline and mean change from baseline in CD4+ cell count and in CD4% from Week 2 to Week 24 are presented in Table 1. All subjects maintained their CD4+ cell counts above 400 cells/mm3 [see Use in Specific Populations (8.4)].
Table 1 Mean Change in CD4+ Count and Percentage from Baseline to Week 24 in Virologically-Suppressed Pediatric Patients from 6 to <12 Years Who Switched to FTC+TAF with EVG+COBI Baseline Mean Change from Baseline Week 2 Week 4 Week 12 Week 24 - * Mean (SD)
CD4+ Cell Count (cells/mm3) 966 (201.7)* -162 -125 -162 -150 CD4% 40 (5.3)* +0.5% -0.1% -0.8% -1.5% Adverse Reactions from Clinical Trial Experience in HIV-1 Uninfected Individuals Taking DESCOVY for HIV-1 PrEP
The safety profile of DESCOVY for HIV-1 PrEP was comparable to that observed in clinical trials of HIV-infected subjects based on a double-blind, randomized, active-controlled trial (DISCOVER) in which a total of 5,387 HIV-1 uninfected adult men and transgender women who have sex with men received DESCOVY (N=2,694) or TRUVADA (N=2,693) once daily for HIV-1 PrEP [see Clinical Studies (14.3)]. Median duration of exposure was 86 and 87 weeks, respectively. The most common adverse reaction in participants who received DESCOVY (incidence greater than or equal to 5%, all grades) was diarrhea (5%). Table 2 provides a list of the most common adverse reactions that occurred in 2% or more of participants in either treatment group. The proportion of participants who discontinued treatment with DESCOVY or TRUVADA due to adverse events, regardless of severity, was 1.3% and 1.8%, respectively.
Table 2 Adverse Reactions (All Grades) Reported in ≥2% in Either Arm in the DISCOVER Trial of HIV-1 Uninfected Participants DESCOVY
(N=2,694)TRUVADA
(N=2,693)- * Includes the following terms: abdominal pain, abdominal pain upper, abdominal pain lower, gastrointestinal pain, and abdominal discomfort
Diarrhea 5% 6% Nausea 4% 5% Headache 2% 2% Fatigue 2% 3% Abdominal pain* 2% 3% Renal Laboratory Tests
Changes from baseline to Week 48 in renal laboratory data are presented in Table 3. The long-term clinical significance of these renal laboratory changes on adverse reaction frequencies between DESCOVY and TRUVADA is not known.
Table 3 Laboratory Assessments of Renal Function Reported in HIV-1 Uninfected Participants Receiving DESCOVY or TRUVADA in the DISCOVER Trial DESCOVY
(N=2,694)TRUVADA
(N=2,693)eGFRCG=estimated Glomerular Filtration Rate by Cockcroft-Gault; UPCR=urine protein/creatinine ratio - * Mean (SD).
- † Median (Q1, Q3).
- ‡ Based on N who had normal UPCR (≤ 200 mg/g) at baseline.
Serum Creatinine (mg/dL)*
Change at Week 48–0.01 (0.107) 0.01 (0.111) eGFRCG (mL/min)†
Change at Week 481.8 (–7.2, 11.1) –2.3 (–10.8, 7.2) Percentage of Participants who Developed UPCR >200 mg/g‡
At Week 480.7% 1.5% Bone Mineral Density Effects
In the DISCOVER trial, mean increases from baseline to Week 48 of 0.5% at the lumbar spine (N=159) and 0.2% at the total hip (N=158) were observed in participants receiving DESCOVY, compared to mean decreases of 1.1% at the lumbar spine (N=160) and 1.0% at the total hip (N=158) in participants receiving TRUVADA. BMD declines of 5% or greater at the lumbar spine and 7% or greater at the total hip were experienced by 4% and 1% of participants, respectively, in both treatment groups at Week 48. The long-term clinical significance of these BMD changes is not known.
Serum Lipids
Changes from baseline to Week 48 in total cholesterol, HDL-cholesterol, LDL-cholesterol, triglycerides, and total cholesterol to HDL ratio are presented in Table 4.
Table 4 Fasting Lipid Values, Mean Change from Baseline, Reported in HIV-1 Uninfected Participants Receiving DESCOVY or TRUVADA in the DISCOVER Trial* DESCOVY
(N=2,694)TRUVADA
(N=2,693)Baseline Week 48 Baseline Week 48 mg/dL Change† mg/dL Change† - * Excludes subjects who received lipid lowering agents during the treatment period.
- † The baseline and change from baseline are for subjects with both baseline and Week 48 values.
- ‡ N=1,098
- § N=1,124
- ¶ N=1,079
- # N=1,107
Total Cholesterol (fasted) 176 ‡ 0 ‡ 176 § -12 § HDL-Cholesterol (fasted) 51 ‡ -2 ‡ 51 § -5 § LDL-Cholesterol (fasted) 103 ¶ 0 ¶ 103 # -7 # Triglycerides (fasted) 109 ‡ +9 ‡ 111 § -1 § Total Cholesterol to HDL ratio 3.7 ‡ 0.2 ‡ 3.7 § 0.1 § 6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of products containing TAF. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Skin and Subcutaneous Tissue Disorders
Angioedema, urticaria, and rash
-
7 DRUG INTERACTIONS
7.1 Potential for Other Drugs to Affect One or More Components of DESCOVY
TAF, a component of DESCOVY, is a substrate of P-gp, BCRP, OATP1B1, and OATP1B3. Drugs that strongly affect P-gp and BCRP activity may lead to changes in TAF absorption (see Table 5). Drugs that induce P-gp activity are expected to decrease the absorption of TAF, resulting in decreased plasma concentration of TAF, which may lead to loss of therapeutic effect of DESCOVY and development of resistance. Coadministration of DESCOVY with other drugs that inhibit P-gp and BCRP may increase the absorption and plasma concentration of TAF. TAF is not an inhibitor of CYP1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, or UGT1A1. TAF is a weak inhibitor of CYP3A in vitro. TAF is not an inhibitor or inducer of CYP3A in vivo.
7.2 Drugs Affecting Renal Function
Because FTC and tenofovir are primarily excreted by the kidneys by a combination of glomerular filtration and active tubular secretion, coadministration of DESCOVY with drugs that reduce renal function or compete for active tubular secretion may increase concentrations of FTC, tenofovir, and other renally eliminated drugs and this may increase the risk of adverse reactions. Some examples of drugs that are eliminated by active tubular secretion include, but are not limited to, acyclovir, cidofovir, ganciclovir, valacyclovir, valganciclovir, aminoglycosides (e.g., gentamicin), and high-dose or multiple NSAIDs [see Warnings and Precautions (5.4)].
7.3 Established and Other Potentially Significant Interactions
Table 5 provides a listing of established or potentially clinically significant drug interactions with recommended steps to prevent or manage the drug interaction (the table is not all inclusive). The drug interactions described are based on studies conducted with either DESCOVY, the components of DESCOVY (emtricitabine and tenofovir alafenamide) as individual agents, or are predicted drug interactions that may occur with DESCOVY. For magnitude of interaction, see Clinical Pharmacology (12.3).
Table 5 Established and Other Potentially Significant* Drug Interactions Concomitant Drug Class: Drug Name Effect on Concentration† Clinical Comment - * This table is not all inclusive.
- † ↓=Decrease
Antiretroviral Agents: Protease Inhibitors (PI) tipranavir/ritonavir ↓ TAF Coadministration with DESCOVY is not recommended. Other Agents Anticonvulsants:
carbamazepine
oxcarbazepine
phenobarbital
phenytoin↓ TAF Consider alternative anticonvulsant. Antimycobacterials:
rifabutin
rifampin
rifapentine↓ TAF Coadministration of DESCOVY with rifabutin, rifampin, or rifapentine is not recommended. Herbal Products:
St. John's wort
(Hypericum perforatum)↓ TAF Coadministration of DESCOVY with St. John's wort is not recommended. 7.4 Drugs without Clinically Significant Interactions with DESCOVY
Based on drug interaction studies conducted with the components of DESCOVY, no clinically significant drug interactions have been either observed or are expected when DESCOVY is combined with the following antiretroviral agents: atazanavir with ritonavir or cobicistat, darunavir with ritonavir or cobicistat, dolutegravir, efavirenz, ledipasvir, lopinavir/ritonavir, maraviroc, nevirapine, raltegravir, rilpivirine, and sofosbuvir. No clinically significant drug interactions have been either observed or are expected when DESCOVY is combined with the following drugs: buprenorphine, itraconazole, ketoconazole, lorazepam, methadone, midazolam, naloxone, norbuprenorphine, norgestimate/ethinyl estradiol, and sertraline.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors pregnancy outcomes in individuals exposed to DESCOVY during pregnancy. Healthcare providers are encouraged to register patients by calling the Antiretroviral Pregnancy Registry (APR) at 1-800-258-4263.
Risk Summary
Available data from the APR show no increase in the risk of overall major birth defects for emtricitabine (FTC) compared with the background rate for major birth defects of 2.7% in a U.S. reference population of the Metropolitan Atlanta Congenital Defects Program (MACDP). There are insufficient tenofovir alafenamide (TAF) data from the APR to adequately assess the risk of major birth defects. The rate of miscarriage for individual drugs is not reported in the APR. The estimated background rate of miscarriage in the clinically recognized pregnancies in the U.S. general population is 15–20%.
In animal studies, no adverse developmental effects were observed when the components of DESCOVY were administered separately during the period of organogenesis at exposures 60 and 108 times (mice and rabbits, respectively) the FTC exposure and at exposure equal to or 53 times (rats and rabbits, respectively) the TAF exposure at the recommended daily dose of DESCOVY (see Data). Likewise, no adverse developmental effects were seen when FTC was administered to mice through lactation at exposures up to approximately 60 times the exposure at the recommended daily dose of DESCOVY. No adverse effects were observed in the offspring when TDF was administered through lactation at tenofovir exposures of approximately 14 times the exposure at the recommended daily dosage of DESCOVY.
Data
Human Data
Emtricitabine: Based on prospective reports to the APR through January 2019 of over 4,450 exposures to FTC-containing regimens during pregnancy (including over 3,150 exposed in the first trimester and over 1,300 exposed in the second/third trimester), there was no difference between FTC and overall birth defects compared with the background birth defect rate of 2.7% in the U.S. reference population of the MACDP. The prevalence of birth defects in live births was 2.4% (95% CI: 1.9% to 3.0%) with first trimester exposure to FTC-containing regimens and 2.3% (95% CI: 1.5% to 3.2%) with the second/third trimester exposure to FTC-containing regimens.
Tenofovir Alafenamide: Based on prospective reports to the APR of over 220 exposures to TAF-containing regimens during pregnancy (including over 160 exposed in the first trimester and over 60 exposed in the second/third trimester), there have been 6 birth defects with first trimester exposure to TAF-containing regimens.
Methodologic limitations of the APR include the use of MACDP as the external comparator group. The MACDP population is not disease-specific, evaluates women and infants from a limited geographic area, and does not include outcomes for births that occurred at <20 weeks gestation.
Additionally, published observational studies on emtricitabine and tenofovir exposure in pregnancy have not shown an increased risk for major malformations.
Animal Data
Emtricitabine: FTC was administered orally to pregnant mice (250, 500, or 1000 mg/kg/day) and rabbits (100, 300, or 1000 mg/kg/day) through organogenesis (on gestation days 6 through 15, and 7 through 19, respectively). No significant toxicological effects were observed in embryo-fetal toxicity studies performed with FTC in mice at exposures (area under the curve [AUC]) approximately 60 times higher and in rabbits at approximately 108 times higher than human exposures at the recommended daily dose. In a pre/postnatal development study with FTC, mice were administered doses up to 1000 mg/kg/day; no significant adverse effects directly related to drug were observed in the offspring exposed daily from before birth (in utero) through sexual maturity at daily exposures (AUC) of approximately 60-fold higher than human exposures at the recommended daily dose.
Tenofovir Alafenamide: TAF was administered orally to pregnant rats (25, 100, or 250 mg/kg/day) and rabbits (10, 30, or 100 mg/kg/day) through organogenesis (on gestation days 6 through 17, and 7 through 20, respectively). No adverse embryo-fetal effects were observed in rats and rabbits at TAF exposures approximately similar to (rats) and 53 (rabbits) times higher than the exposure in humans at the recommended daily dose of DESCOVY. TAF is rapidly converted to tenofovir; the observed tenofovir exposures in rats and rabbits were 59 (rats) and 93 (rabbits) times higher than human tenofovir exposures at the recommended daily dose. Since TAF is rapidly converted to tenofovir and a lower tenofovir exposure in rats and mice was observed after TAF administration compared to tenofovir disoproxil fumarate (TDF, another prodrug for tenofovir) administration, a pre/postnatal development study in rats was conducted only with TDF. Doses up to 600 mg/kg/day were administered through lactation; no adverse effects were observed in the offspring on gestation day 7 (and lactation day 20) at tenofovir exposures of approximately 14 (21) times higher than the exposures in humans at the recommended daily dose of DESCOVY.
8.2 Lactation
Risk Summary
The Centers for Disease Control and Prevention recommend that HIV-infected mothers not breastfeed their infants, to avoid risking postnatal transmission of HIV-1.
Based on limited data, FTC has been shown to be present in human breast milk; it is not known if TAF is present in human breast milk. Tenofovir has been shown to be present in the milk of lactating rats and rhesus monkeys after administration of TDF (see Data). It is not known if TAF is present in animal milk.
It is not known if DESCOVY affects milk production or has effects on the breastfed child.
Because of the potential for: 1) HIV transmission (in HIV-negative infants); 2) developing viral resistance (in HIV-positive infants); and 3) adverse reactions in a breastfed infant similar to those seen in adults, instruct mothers not to breastfeed if they are taking DESCOVY for the treatment of HIV-1 (see Data).
Data
Animal Data
Tenofovir Alafenamide: Studies in rats and monkeys have demonstrated that tenofovir is secreted in milk. Tenofovir was excreted into the milk of lactating rats following oral administration of TDF (up to 600 mg/kg/day) at up to approximately 24% of the median plasma concentration in the highest dosed animals at lactation day 11. Tenofovir was excreted into the milk of lactating monkeys following a single subcutaneous (30 mg/kg) dose of tenofovir at concentrations up to approximately 4% of plasma concentration, resulting in exposure (AUC) of approximately 20% of plasma exposure.
8.4 Pediatric Use
Treatment of HIV-1 Infection
The safety and effectiveness of DESCOVY, in combination with other antiretroviral agents, for the treatment of HIV-1 infection was established in pediatric patients with body weight greater than or equal to 25 kg [see Indication and Usage (1.1) and Dosage and Administration (2.3)].
Use of DESCOVY in pediatric patients between the ages of 12 to less than 18 years weighing at least 35 kg is supported by adequate and well controlled studies of FTC+TAF with EVG+COBI in adults and by an open-label trial in antiretroviral treatment-naïve HIV-1 infected pediatric subjects ages 12 to less than 18 years and weighing at least 35 kg (N=50; cohort 1). The safety and efficacy of FTC+TAF with EVG+COBI in these pediatric subjects was similar to that of HIV-1 infected adults on this regimen [see Clinical Pharmacology (12.3) and Clinical Studies (14.2)].
Use of DESCOVY in pediatric patients weighing at least 25 kg is supported by adequate and well controlled studies of FTC+TAF with EVG+COBI in adults and by an open-label trial in virologically-suppressed pediatric subjects between the ages of 6 to less than 12 years weighing at least 25 kg, in which subjects were switched from their antiretroviral regimen to FTC+TAF with EVG+COBI (N=23; cohort 2). The safety in these subjects through 24 weeks of FTC+TAF with EVG+COBI was similar to that of HIV-1 infected adults on this regimen, with the exception of a decrease in mean change from baseline in CD4+ cell count [see Adverse Reactions (6.1), Clinical Pharmacology (12.3) and Clinical Studies (14.2)].
Safety and effectiveness of DESCOVY coadministered with an HIV-1 protease inhibitor that is administered with either ritonavir or cobicistat have not been established in pediatric subjects weighing less than 35 kg [see Dosage and Administration (2.3)].
Safety and effectiveness of DESCOVY for treatment of HIV-1 infection in pediatric patients less than 25 kg have not been established.
HIV-1 PrEP
Safety and effectiveness of DESCOVY for HIV-1 PrEP in at-risk adolescents weighing at least 35 kg, excluding individuals at risk from receptive vaginal sex, is supported by data from an adequate and well-controlled trial of DESCOVY for HIV-1 PrEP in adults with additional data from safety and pharmacokinetic studies in previously conducted trials with the individual drug products, FTC and TAF, with EVG+COBI, in HIV-1 infected adults and pediatric subjects [see Dosage and Administration (2.4), Adverse Reactions (6.1), Clinical Pharmacology (12.3 and 12.4), and Clinical Studies (14)].
While using DESCOVY for HIV-1 PrEP, HIV-1 testing should be repeated at least every 3 months, and upon diagnosis of any other STIs. Previous studies in at-risk adolescents indicated waning adherence to a daily oral PrEP regimen once visits were switched from monthly to quarterly visits. Adolescents may therefore benefit from more frequent visits and counseling [see Warnings and Precautions (5.2)].
Safety and effectiveness of DESCOVY for HIV-1 PrEP in pediatric patients less than 35 kg have not been established.
8.5 Geriatric Use
In clinical trials of an FTC+TAF-containing regimen for treatment of HIV-1, 80 of the 97 subjects enrolled aged 65 years and over received FTC+TAF and EVG+COBI. No differences in safety or efficacy have been observed between elderly subjects and adults between 18 and less than 65 years of age.
8.6 Renal Impairment
No dosage adjustment of DESCOVY is recommended in individuals with estimated creatinine clearance greater than or equal to 30 mL per minute, or in adults with ESRD (estimated creatinine clearance below 15 mL per minute) who are receiving chronic hemodialysis. On days of hemodialysis, administer the daily dose of DESCOVY after completion of hemodialysis treatment.
Safety and effectiveness of DESCOVY coadministered with an HIV-1 protease inhibitor that is administered with either ritonavir or cobicistat have not been established in patients with ESRD [see Dosage and Administration (2.3)].
DESCOVY is not recommended in individuals with severe renal impairment (estimated creatinine clearance of 15 to below 30 mL per minute), or in individuals with ESRD who are not receiving chronic hemodialysis, as the safety of DESCOVY has not been established in these populations [see Dosage and Administration (2.5) and Clinical Studies (14.2)].
8.7 Hepatic Impairment
No dosage adjustment of DESCOVY is recommended in individuals with mild (Child-Pugh Class A) or moderate (Child-Pugh Class B) hepatic impairment. DESCOVY has not been studied in individuals with severe hepatic impairment (Child-Pugh Class C) [see Clinical Pharmacology (12.3)].
-
10 OVERDOSAGE
No data are available on overdose of DESCOVY in patients. If overdose occurs, monitor the individual for evidence of toxicity. Treatment of overdose with DESCOVY consists of general supportive measures including monitoring of vital signs as well as observation of the clinical status of the individual.
Emtricitabine (FTC): Limited clinical experience is available at doses higher than the recommended dose of FTC in DESCOVY. In one clinical pharmacology study, single doses of FTC 1200 mg (6 times the FTC dose in DESCOVY) were administered to 11 subjects. No severe adverse reactions were reported. The effects of higher doses are not known.
Hemodialysis treatment removes approximately 30% of the FTC dose over a 3-hour dialysis period starting within 1.5 hours of FTC dosing (blood flow rate of 400 mL per minute and a dialysate flow rate of 600 mL per minute). It is not known whether FTC can be removed by peritoneal dialysis.
Tenofovir Alafenamide (TAF): Limited clinical experience is available at doses higher than the recommended dose of TAF. A single dose of 125 mg TAF (5 times the TAF dose in 200/25 mg DESCOVY) was administered to 48 healthy subjects; no serious adverse reactions were reported. The effects of higher doses are unknown. Tenofovir is efficiently removed by hemodialysis with an extraction coefficient of approximately 54%.
-
11 DESCRIPTION
DESCOVY (emtricitabine and tenofovir alafenamide) is a fixed dose combination tablet containing emtricitabine (FTC) and tenofovir alafenamide (TAF) for oral administration.
- FTC, a synthetic nucleoside analog of cytidine, is an HIV nucleoside analog reverse transcriptase inhibitor (HIV NRTI).
- TAF, an HIV NRTI, is converted in vivo to tenofovir, an acyclic nucleoside phosphonate (nucleotide) analog of adenosine 5'-monophosphate.
Each 200/25 mg tablet contains 200 mg of FTC and 25 mg of TAF (equivalent to 28 mg of tenofovir alafenamide fumarate) and the following inactive ingredients: croscarmellose sodium, magnesium stearate, and microcrystalline cellulose. The tablets are film-coated with a coating material containing indigo carmine aluminum lake, polyethylene glycol, polyvinyl alcohol, talc, and titanium dioxide.
Emtricitabine: The chemical name of FTC is 4-amino-5-fluoro-1-(2R-hydroxymethyl-1,3-oxathiolan-5S-yl)-(1H)-pyrimidin-2-one. FTC is the (-)enantiomer of a thio analog of cytidine, which differs from other cytidine analogs in that it has a fluorine in the 5 position.
FTC has a molecular formula of C8H10FN3O3S and a molecular weight of 247.24 and has the following structural formula:
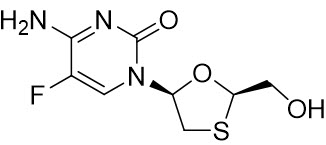
FTC is a white to off-white powder with a solubility of approximately 112 mg per mL in water at 25 °C.
Tenofovir Alafenamide: The chemical name of tenofovir alafenamide fumarate drug substance is L-alanine, N-[(S)-[[(1R)-2-(6-amino-9H-purin-9-yl)-1-methylethoxy]methyl]phenoxyphosphinyl]-, 1-methylethyl ester, (2E)-2-butenedioate (2:1).
Tenofovir alafenamide fumarate has an empirical formula of C21H29O5N6P∙½(C4H4O4) and a formula weight of 534.50 and has the following structural formula:
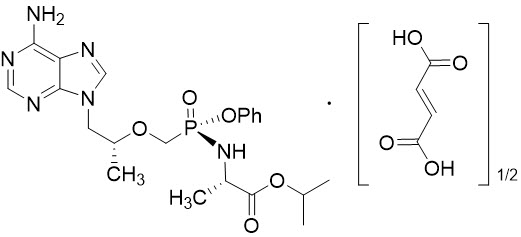
Tenofovir alafenamide fumarate is a white to off-white or tan powder with a solubility of 4.7 mg per mL in water at 20 °C.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
DESCOVY is a fixed dose combination of antiretroviral drugs emtricitabine (FTC) and tenofovir alafenamide (TAF) [see Microbiology (12.4)].
12.2 Pharmacodynamics
Cardiac Electrophysiology
In a thorough QT/QTc study in 48 healthy subjects, TAF at the recommended dose or at a dose approximately 5 times the recommended dose, did not affect the QT/QTc interval and did not prolong the PR interval. The effect of the other component of DESCOVY, FTC, or the combination of FTC and TAF on the QT interval is not known.
12.3 Pharmacokinetics
Absorption, Distribution, Metabolism, and Excretion
The pharmacokinetic (PK) properties of the components of DESCOVY are provided in Table 6. The multiple dose PK parameters of FTC and TAF and its metabolite tenofovir are provided in Table 7. HIV status has no effect on the pharmacokinetics of FTC and TAF in adults.
Table 6 Pharmacokinetic Properties of the Components of DESCOVY Emtricitabine Tenofovir Alafenamide PBMCs=peripheral blood mononuclear cells; CES1=carboxylesterase 1 - * Values refer to geometric mean ratio [High-fat meal/ fasting] in PK parameters and (90% confidence interval). High-calorie/high-fat meal = ~800 kcal, 50% fat.
- † In vivo, TAF is hydrolyzed within cells to form tenofovir (major metabolite), which is phosphorylated to the active metabolite, tenofovir diphosphate. In vitro studies have shown that TAF is metabolized to tenofovir by cathepsin A in PBMCs and macrophages; and by CES1 in hepatocytes. Upon coadministration with the moderate CYP3A inducer probe efavirenz, TAF exposure was unaffected.
- ‡ t1/2 values refer to median terminal plasma half-life. Note that the pharmacologically active metabolite, tenofovir diphosphate, has a half-life of 150–180 hours within PBMCs.
- § Dosing in mass balance studies: FTC (single dose administration of [14C] emtricitabine after multiple dosing of emtricitabine for 10 days); TAF (single dose administration of [14C] tenofovir alafenamide).
Absorption Tmax (h) 3 1 Effect of high fat meal (relative to fasting)* AUC Ratio = 0.91 (0.89, 0.93)
Cmax Ratio = 0.74 (0.69, 0.78)AUC Ratio = 1.75 (1.64, 1.88)
Cmax Ratio= 0.85 (0.75, 0.95)Distribution % Bound to human plasma proteins <4 ~80 Source of protein binding data In vitro Ex vivo Blood-to-plasma ratio 0.6 1.0 Metabolism Metabolism Not significantly metabolized Cathepsin A† (PBMCs)
CES1 (hepatocytes)
CYP3A (minimal)Elimination Major route of elimination Glomerular filtration and active tubular secretion Metabolism (>80% of oral dose) t1/2 (h)‡ 10 0.51 % Of dose excreted in urine§ 70 <1 % Of dose excreted in feces§ 13.7 31.7 Table 7 Multiple Dose PK Parameters of Emtricitabine, Tenofovir Alafenamide and its Metabolite Tenofovir Following Oral Administration with Food in HIV-Infected Adults Parameter
Mean (CV%)Emtricitabine* Tenofovir Alafenamide† Tenofovir‡ CV=Coefficient of Variation; NA=Not Applicable - * From Intensive PK analysis in a phase 2 trial in HIV-infected adults treated with FTC+TAF and EVG+COBI.
- † From Population PK analysis in two trials of treatment-naïve adults with HIV-1 infection treated with FTC+TAF with EVG+COBI (N=539).
- ‡ From Population PK analysis in two trials of treatment-naïve adults with HIV-1 infection treated with FTC+TAF with EVG+COBI (N=841).
Cmax
(microgram per mL)2.1 (20.2) 0.16 (51.1) 0.02 (26.1) AUCtau
(microgram∙hour per mL)11.7 (16.6) 0.21 (71.8) 0.29 (27.4) Ctrough
(microgram per mL)0.10 (46.7) NA 0.01 (28.5) Specific Populations
Geriatric Patients
Pharmacokinetics of FTC and TAF have not been fully evaluated in the elderly (65 years of age and older). Population pharmacokinetics analysis of HIV-infected subjects in Phase 2 and Phase 3 trials of FTC+TAF and EVG+COBI showed that age did not have a clinically relevant effect on exposures of TAF up to 75 years of age [see Use in Specific Populations (8.5)].
Pediatric Patients
Treatment of HIV-1 Infection: Mean exposures of TAF in 24 pediatric subjects aged 12 to less than 18 years who received FTC+TAF with EVG+COBI were decreased (23% for AUC) and FTC exposures were similar compared to exposures achieved in treatment-naïve adults following administration of this dosage regimen. The TAF exposure differences are not thought to be clinically significant based on exposure-response relationships (Table 8).
Table 8 Multiple Dose PK Parameters of Emtricitabine, Tenofovir Alafenamide, and its Metabolite Tenofovir Following Oral Administration of FTC+TAF with EVG+COBI in HIV-Infected Pediatric Subjects Aged 12 to less than 18 Years* Parameter
Mean (CV%)Emtricitabine Tenofovir Alafenamide Tenofovir CV = Coefficient of Variation; NA = Not Applicable - * From Intensive PK analysis in a trial in treatment-naïve pediatric subjects with HIV-1 infection (N=24).
- † N=23
Cmax
(microgram per mL)2.3
(22.5)0.17
(64.4)0.02
(23.7)AUCtau
(microgram∙hour per mL)14.4
(23.9)0.20†
(50.0)0.29†
(18.8)Ctrough
(microgram per mL)0.10†
(38.9)NA 0.01
(21.4)Exposures of FTC and TAF achieved in 23 pediatric subjects between the ages of 6 to less than 12 years and weighing at least 25 kg (55 lbs) who received FTC+TAF with EVG+COBI were higher (20% to 80% for AUC) than exposures achieved in adults following the administration of this dosage regimen; however, the increase was not considered clinically significant (Table 9) [see Use in Specific Populations (8.4)].
Table 9 Multiple Dose PK Parameters of Emtricitabine, Tenofovir Alafenamide and its Metabolite Tenofovir Following Oral Administration of FTC+TAF with EVG+COBI in HIV-Infected Pediatric Subjects Aged 6 to less than 12 Years* Parameter
Mean (CV%)Emtricitabine Tenofovir Alafenamide Tenofovir CV = Coefficient of Variation; NA = Not Applicable - * From Intensive PK analysis in a trial in virologically-suppressed pediatric subjects with HIV-1 infection (N=23).
- † N=22
Cmax
(microgram per mL)3.4
(27.0)0.31
(61.2)0.03
(20.8)AUCtau
(microgram∙hour per mL)20.6†
(18.9)0.33
(44.8)0.44
(20.9)Ctrough
(microgram per mL)0.11
(24.1)NA 0.02
(24.9)HIV-1 PrEP: The pharmacokinetic data for FTC and TAF following administration of DESCOVY in HIV-1 uninfected adolescents weighing 35 kg and above are not available. The dosage recommendations of DESCOVY for HIV-1 PrEP in this population are based on known pharmacokinetic information in HIV-infected adolescents taking FTC and TAF for treatment [see Use in Specific Populations (8.4)].
Race and Gender
Based on population pharmacokinetic analyses, there are no clinically meaningful differences based on race or gender.
Patients with Renal Impairment
The pharmacokinetics of FTC+TAF combined with EVG+COBI in HIV-1 infected subjects with renal impairment (eGFR 30 to 69 mL per minute by Cockcroft-Gault method), and in HIV-1 infected subjects with ESRD (eGFR less than 15 mL per minute by Cockcroft-Gault method) receiving chronic hemodialysis were evaluated in subsets of virologically-suppressed subjects in open-label trials. The pharmacokinetics of TAF were similar among healthy subjects, subjects with mild or moderate renal impairment, and subjects with ESRD receiving chronic hemodialysis; increases in FTC and TFV exposures in subjects with renal impairment were not considered clinically relevant (Table 10).
Table 10 Pharmacokinetics of the Components of DESCOVY and a Metabolite of TAF (Tenofovir) in HIV-Infected Adults with Renal Impairment Compared to Subjects with Normal Renal Function AUCtau (microgram∙hour per mL)
Mean (CV%)Estimated Creatinine Clearance* ≥90 mL per minute (N=18)† 60–89 mL per minute (N=11)‡ 30–59 mL per minute (N=18)§ <15 mL per minute (N=12)¶ - * By Cockcroft-Gault method.
- † From a phase 2 trial in HIV-infected adults with normal renal function treated with FTC+TAF with EVG+COBI.
- ‡ These subjects had an eGFR ranging from 60 to 69 mL per minute.
- § From a phase 3 trial in HIV-1 infected adults with renal impairment treated with FTC+TAF with EVG+COBI.
- ¶ From a phase 3 trial in HIV-1 infected adults with ESRD receiving chronic hemodialysis treated with FTC+TAF with EVG+COBI; PK assessed prior to hemodialysis following 3 consecutive daily doses of FTC+TAF with EVG+COBI.
- # N = 11.
- Þ N = 10.
Emtricitabine 11.4 (11.9) 17.6 (18.2) 23.0 (23.6) 62.9 (48.0)# Tenofovir 0.32 (14.9) 0.46 (31.5) 0.61 (28.4) 8.72 (39.4)Þ Patients with Hepatic Impairment
Emtricitabine: The pharmacokinetics of FTC has not been studied in subjects with hepatic impairment; however, FTC is not significantly metabolized by liver enzymes, so the impact of hepatic impairment should be limited.
Tenofovir Alafenamide: Clinically relevant changes in tenofovir pharmacokinetics in subjects with hepatic impairment were not observed in subjects with mild to moderate (Child-Pugh Class A and B) hepatic impairment [see Use in Specific Populations (8.7)].
Drug Interaction Studies
The effects of coadministered drugs on the exposure of TAF are shown in Table 11 and the effects of DESCOVY or its components on the exposure of coadministered drugs are shown in Table 12 [these studies were conducted with DESCOVY or the components of DESCOVY (FTC or TAF) administered alone]. For information regarding clinical recommendations, see Drug Interactions (7).
Table 11 Drug Interactions: Changes in TAF Pharmacokinetic Parameters in the Presence of Coadministered Drug(s)* Coadministered Drug Coadministered Drug(s) Dosage
(once daily)
(mg)Tenofovir Alafenamide Dosage
(once daily)
(mg)N Mean Ratio of TAF PK Parameters (90% CI);
No effect = 1.00Cmax AUC Cmin NC=Not Calculated - * All interaction studies conducted in healthy volunteers.
- † Study conducted with DESCOVY (FTC/TAF).
- ‡ Study conducted with FTC+TAF with EVG+COBI.
Atazanavir 300 (+100 ritonavir) 10 10 1.77
(1.28, 2.44)1.91
(1.55, 2.35)NC Cobicistat 150 8 12 2.83
(2.20, 3.65)2.65
(2.29, 3.07)NC Darunavir 800 (+150 cobicistat) 25† 11 0.93
(0.72, 1.21)0.98
(0.80, 1.19)NC Darunavir 800 (+100 ritonavir) 10 10 1.42
(0.96, 2.09)1.06
(0.84, 1.35)NC Dolutegravir 50 10 10 1.24
(0.88, 1.74)1.19
(0.96, 1.48)NC Efavirenz 600 40† 11 0.78
(0.58, 1.05)0.86
(0.72, 1.02)NC Lopinavir 800 (+200 ritonavir) 10 10 2.19
(1.72, 2.79)1.47
(1.17, 1.85)NC Rilpivirine 25 25 17 1.01
(0.84, 1.22)1.01
(0.94, 1.09)NC Sertraline 50 (dosed as a single dose) 10‡ 19 1.00
(0.86, 1.16)0.96
(0.89, 1.03)NC Table 12 Drug Interactions: Changes in PK Parameters for Coadministered Drug in the Presence of DESCOVY or the Individual Components* Coadministered Drug Coadministered Drug Dosage (once daily)
(mg)Tenofovir Alafenamide Dosage
(once daily)
(mg)N Mean Ratio of Coadministered Drug PK Parameters (90% CI);
No effect = 1.00Cmax AUC Cmin NC=Not Calculated - * All interaction studies conducted in healthy volunteers.
- † Study conducted with DESCOVY (FTC/TAF).
- ‡ A sensitive CYP3A4 substrate.
- § Study conducted with FTC+TAF with EVG+COBI.
Atazanavir 300 +100 ritonavir 10 10 0.98
(0.89, 1.07)0.99
(0.96, 1.01)1.00
(0.96, 1.04)Darunavir 800 +150 cobicistat 25† 11 1.02
(0.96, 1.09)0.99
(0.92, 1.07)0.97
(0.82, 1.15)Darunavir 800 +100 ritonavir 10 10 0.99
(0.91, 1.08)1.01
(0.96, 1.06)1.13
(0.95, 1.34)Dolutegravir 50 mg 10 10 1.15
(1.04, 1.27)1.02
(0.97, 1.08)1.05
(0.97, 1.13)Lopinavir 800 +200 ritonavir 10 10 1.00
(0.95, 1.06)1.00
(0.92, 1.09)0.98
(0.85, 1.12)Midazolam‡ 2.5 (single dose, orally) 25 18 1.02
(0.92, 1.13)1.13
(1.04, 1.23)NC 1 (single dose, intravenous) 0.99
(0.89, 1.11)1.08
(1.04, 1.14)NC Rilpivirine 25 25 16 0.93
(0.87, 0.99)1.01
(0.96, 1.06)1.13
(1.04, 1.23)Sertraline 50 (single dose) 10§ 19 1.14
(0.94, 1.38)0.93
(0.77, 1.13)NC 12.4 Microbiology
Mechanism of Action
Emtricitabine: FTC, a synthetic nucleoside analog of cytidine, is phosphorylated by cellular enzymes to form emtricitabine 5'-triphosphate. Emtricitabine 5'-triphosphate inhibits the activity of the HIV-1 reverse transcriptase by competing with the natural substrate deoxycytidine 5'-triphosphate and by being incorporated into nascent viral DNA which results in chain termination. Emtricitabine 5′-triphosphate is a weak inhibitor of mammalian DNA polymerases α, β, Ɛ, and mitochondrial DNA polymerase γ.
Tenofovir Alafenamide: TAF is a phosphonoamidate prodrug of tenofovir (2'-deoxyadenosine monophosphate analog). Plasma exposure to TAF allows for permeation into cells and then TAF is intracellularly converted to tenofovir through hydrolysis by cathepsin A. Tenofovir is subsequently phosphorylated by cellular kinases to the active metabolite tenofovir diphosphate. Tenofovir diphosphate inhibits HIV-1 replication through incorporation into viral DNA by the HIV reverse transcriptase, which results in DNA chain-termination.
Tenofovir has activity against HIV-1. Cell culture studies have shown that both tenofovir and FTC can be fully phosphorylated when combined in cells. Tenofovir diphosphate is a weak inhibitor of mammalian DNA polymerases that include mitochondrial DNA polymerase γ and there is no evidence of toxicity to mitochondria in cell culture.
Antiviral Activity in Cell Culture
Emtricitabine: The antiviral activity of FTC against laboratory and clinical isolates of HIV-1 was assessed in T lymphoblastoid cell lines, the MAGI-CCR5 cell line, and primary peripheral blood mononuclear cells. The EC50 values for FTC were in the range of 1.3–640 nM. FTC displayed antiviral activity in cell culture against HIV-1 clades A, B, C, D, E, F, and G (EC50 values ranged from 7–75 nM) and showed strain specific activity against HIV-2 (EC50 values ranged from 7–1,500 nM).
In a study of FTC with a broad panel of representatives from the major classes of approved anti-HIV agents (NRTIs, non-nucleoside reverse transcriptase inhibitors [NNRTIs], integrase strand transfer inhibitors [INSTIs], and PIs) no antagonism was observed for these combinations.
Tenofovir Alafenamide: The antiviral activity of TAF against laboratory and clinical isolates of HIV-1 subtype B was assessed in lymphoblastoid cell lines, PBMCs, primary monocyte/macrophage cells and CD4+-T lymphocytes. The EC50 values for TAF ranged from 2.0 to 14.7 nM.
TAF displayed antiviral activity in cell culture against all HIV-1 groups (M, N, O), including sub-types A, B, C, D, E, F, and G (EC50 values ranged from 0.10 to 12.0 nM) and strain specific activity against HIV-2 (EC50 values ranged from 0.91 to 2.63 nM).
In a study of TAF with a broad panel of representatives from the major classes of approved anti-HIV agents (NRTIs, NNRTIs, INSTIs, and PIs) no antagonism was observed for these combinations.
Prophylactic Activity in a Nonhuman Primate Model of HIV-1 Transmission
Emtricitabine and Tenofovir Alafenamide: The prophylactic activity of the combination of oral FTC and TAF was evaluated in a controlled study of macaques administered once weekly intra-rectal inoculations of chimeric simian/human immunodeficiency type 1 virus (SHIV) for up to 19 weeks (n=6). All 6 macaques that received FTC and TAF at doses resulting in PBMC exposures consistent with those achieved in humans administered a dose of FTC/TAF 200/25 mg remained SHIV uninfected.
Resistance
In Cell Culture
In Clinical Trials
Treatment of HIV-1
The resistance profile of DESCOVY in combination with other antiretroviral agents for the treatment of HIV-1 infection is based on studies of FTC+TAF with EVG+COBI in the treatment of HIV-1 infection. In a pooled analysis of antiretroviral-naïve subjects, genotyping was performed on plasma HIV-1 isolates from all subjects with HIV-1 RNA greater than 400 copies per mL at confirmed virologic failure, at Week 48, or at time of early study drug discontinuation. Genotypic resistance developed in 7 of 14 evaluable subjects. The resistance-associated substitutions that emerged were M184V/I (N=7) and K65R (N=1). Three subjects had virus with emergent R, H, or E at the polymorphic Q207 residue in reverse transcriptase.
One subject was identified with emergent resistance to FTC (M184M/I) out of 4 virologic failure subjects in a clinical study of virologically-suppressed subjects who switched from a regimen containing FTC+TDF to FTC+TAF with EVG+COBI (N=799).
HIV-1 PrEP
In the DISCOVER trial of HIV-1 uninfected men and transgender women who have sex with men and who are at risk of HIV-1 infection receiving DESCOVY or TRUVADA for HIV-1 PrEP, genotyping was performed on participants found to be infected during the trial who had HIV-1 RNA ≥400 copies/mL (6 of 7 participants receiving DESCOVY and 13 of 15 participants receiving TRUVADA). The development of FTC resistance-associated substitutions, M184I and/or M184V, was observed in 4 HIV-1 infected participants in the TRUVADA group who had suspected baseline infections.
Cross-Resistance
Emtricitabine: FTC-resistant viruses with the M184V or I substitution were cross-resistant to lamivudine, but retained sensitivity to didanosine, stavudine, tenofovir, and zidovudine.
Viruses harboring substitutions conferring reduced susceptibility to stavudine and zidovudine-thymidine analog substitutions (M41L, D67N, K70R, L210W, T215Y/F, K219Q/E) or didanosine (L74V) remained sensitive to FTC. HIV-1 containing the K103N substitution or other substitutions associated with resistance to NNRTIs was susceptible to FTC.
Tenofovir Alafenamide: Tenofovir resistance substitutions K65R and K70E result in reduced susceptibility to abacavir, didanosine, emtricitabine, lamivudine, and tenofovir.
HIV-1 with multiple thymidine analog substitutions (M41L, D67N, K70R, L210W, T215F/Y, K219Q/E/N/R), or multinucleoside resistant HIV-1 with a T69S double insertion mutation or with a Q151M substitution complex including K65R, showed reduced susceptibility to TAF in cell culture.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Emtricitabine
In long-term carcinogenicity studies of FTC, no drug-related increases in tumor incidence were found in mice at doses up to 750 mg per kg per day (23 times the human systemic exposure at the recommended dose of 200 mg per day in DESCOVY) or in rats at doses up to 600 mg per kg per day (28 times the human systemic exposure at the recommended dose in DESCOVY).
FTC was not genotoxic in the reverse mutation bacterial test (Ames test), mouse lymphoma or mouse micronucleus assays.
FTC did not affect fertility in male rats at approximately 140 times or in male and female mice at approximately 60 times higher exposures (AUC) than in humans given the recommended 200 mg daily dosage in DESCOVY. Fertility was normal in the offspring of mice exposed daily from before birth (in utero) through sexual maturity at daily exposures (AUC) of approximately 60 times higher than human exposures at the recommended 200 mg daily dosage in DESCOVY.
Tenofovir Alafenamide
Since TAF is rapidly converted to tenofovir and a lower tenofovir exposure in rats and mice was observed after TAF administration compared to TDF administration, carcinogenicity studies were conducted only with TDF. Long-term oral carcinogenicity studies of TDF in mice and rats were carried out at exposures up to approximately 10 times (mice) and 4 times (rats) those observed in humans at the recommended dose of TDF (300 mg) for HIV-1 infection. The tenofovir exposure in these studies was approximately 167 times (mice) and 55 times (rat) those observed in humans after administration of the daily recommended dose of DESCOVY. At the high dose in female mice, liver adenomas were increased at tenofovir exposures approximately 10 times (300 mg TDF) and 167 times (DESCOVY) the exposure observed in humans. In rats, the study was negative for carcinogenic findings.
TAF was not genotoxic in the reverse mutation bacterial test (Ames test), mouse lymphoma or rat micronucleus assays.
There were no effects on fertility, mating performance, or early embryonic development when TAF was administered to male rats at a dose equivalent to 62 times (25 mg TAF) the human dose based on body surface area comparisons for 28 days prior to mating and to female rats for 14 days prior to mating through Day 7 of gestation.
13.2 Animal Toxicology and/or Pharmacology
Minimal to slight infiltration of mononuclear cells in the posterior uvea was observed in dogs with similar severity after three- and nine-month administration of TAF; reversibility was seen after a three-month recovery period. No eye toxicity was observed in the dog at systemic exposures of 5 (TAF) and 15 (tenofovir) times the exposure seen in humans with the recommended daily TAF dose in DESCOVY.
-
14 CLINICAL STUDIES
14.1 Overview of Clinical Trials
The efficacy and safety of DESCOVY have been evaluated in the trials summarized in Table 13.
Table 13 Trials Conducted with FTC+TAF-Containing Products for HIV-1 Treatment and DESCOVY for HIV-1 PrEP Trial Population Study Arms (N) Timepoint - * Randomized, double-blind, active-controlled study.
- † Administered as GENVOYA®.
- ‡ Administered as STRIBILD®.
- § Randomized, open-label, active controlled trial.
- ¶ HIV-1 RNA less than 50 copies per mL.
- # Open label trial
- Þ Estimated creatinine clearance between 30 and 69 mL per minute by Cockcroft-Gault method.
- ß End stage renal disease (estimated creatinine clearance of less than 15 mL per minute by Cockcroft-Gault method).
- à Exposure in the DESCOVY group.
Study 104 *
(NCT01780506)
Study 111 *
(NCT01797445)HIV-1 infected treatment-naïve adults FTC+TAF with EVG+COBI † (866)
FTC+TDF with EVG+COBI ‡ (867)48 Weeks Study 109 §
(NCT01815736)HIV-1 infected virologically-suppressed ¶ adults FTC+TAF with EVG+COBI † (799)
ATRIPLA® or TRUVADA®+atazanavir+cobicistat or ritonavir or FTC+TDF with EVG+COBI ‡ (397)48 Weeks Study 112 #
(NCT01818596)HIV-1 infected virologically-suppressed ¶ adults with renal impairment Þ FTC+TAF with EVG+COBI † (242) 24 Weeks Study 1825 #
(NCT02600819)HIV-1 infected virologically-suppressed ¶ adults with ESRD ß receiving chronic hemodialysis FTC+TAF with EVG+COBI † (55) 48 Weeks Study 106 #
(Cohort 1)
(NCT01854775)HIV-1 infected treatment-naïve adolescents between the ages of 12 to less than 18 years (at least 35 kg) FTC+TAF with EVG+COBI † (50) 48 Weeks Study 106 #
(Cohort 2)
(NCT01854775)HIV-1 infected, virologically suppressed children between the ages of 6 to less than 12 years (at least 25 kg) FTC+TAF with EVG+COBI † (23) 24 Weeks DISCOVER *
(NCT02842086)HIV-1 uninfected men or transgender women who have sex with men DESCOVY (2,670)
TRUVADA® (2,665)4,370 person-yearsà 14.2 Clinical Trial Results for Treatment of HIV-1
In trials of FTC+TAF with EVG+COBI in HIV-1 infected adults as initial therapy in those with no antiretroviral treatment history (N=866) and to replace a stable antiretroviral regimen in those who were virologically-suppressed for at least 6 months with no known resistance substitutions (N=799), 92% and 96% of patients in the two populations, respectively, had HIV-1 RNA less than 50 copies per mL at Week 48.
An open-label, single arm trial of FTC+TAF with EVG+COBI enrolled 50 treatment-naïve HIV-1 infected adolescents aged 12 to less than 18 years weighing at least 35 kg (cohort 1) and 23 virologically suppressed children aged 6 to less than 12 years weighing at least 25 kg (cohort 2). In cohort 1, the virologic response rate (i.e., HIV-1 RNA less than 50 copies per mL) was 92% (46/50) and the mean increase from baseline in CD4+ cell count was 224 cells per mm3 at Week 48. In cohort 2, 100% of subjects remained virologically suppressed at Week 24. From a mean (SD) baseline CD4+ cell count of 966 (201.7), the mean change from baseline in CD4+ cell count was -150 cells/mm3 and the mean (SD) change in CD4% was -1.5% (3.7%) at Week 24. All subjects maintained CD4+ cell counts above 400 cells/mm3 [see Adverse Reactions (6.1) and Use in Specific Populations (8.4)].
In a trial in 248 HIV-1 infected adults with estimated creatinine clearance greater than 30 mL per minute but less than 70 mL per minute, 95% (235/248) of the combined population of treatment-naïve subjects began on FTC+TAF with EVG+COBI (N=6) and those previously virologically-suppressed on other regimens and switched to FTC+TAF with EVG+COBI (N=242) had HIV-1 RNA less than 50 copies per mL at Week 24.
In a trial in 55 HIV-1 infected virologically-suppressed adults with ESRD (estimated creatinine clearance of less than 15 mL per minute) receiving chronic hemodialysis for at least 6 months who switched to FTC+TAF with EVG+COBI, 82% (45/55) maintained HIV-1 RNA less than 50 copies per mL at Week 48. Two subjects had HIV-1 RNA ≥ 50 copies per mL by Week 48, 7 discontinued due to AE or other reasons while suppressed, and 1 did not have an HIV-1 RNA measurement at Week 48.
14.3 Clinical Trial Results for HIV-1 PrEP
The efficacy and safety of DESCOVY to reduce the risk of acquiring HIV-1 infection were evaluated in a randomized, double-blind multinational trial (DISCOVER) in HIV-seronegative men (N=5,262) or transgender women (N=73) who have sex with men and are at risk of HIV-1 infection, comparing once daily DESCOVY (N=2,670) to TRUVADA (FTC/TDF 200 mg/300 mg; N=2,665). Evidence of risk behavior at entry into the trial included at least one of the following: two or more unique condomless anal sex partners in the past 12 weeks or a diagnosis of rectal gonorrhea/chlamydia or syphilis in the past 24 weeks. The median age of participants was 34 years (range, 18–76); 84% were White, 9% Black/Mixed Black, 4% Asian, and 24% Hispanic/Latino. At baseline, 897 participants (17%) reported receiving TRUVADA for PrEP.
At weeks 4, 12, and every 12 weeks thereafter, all participants received local standard of care HIV-1 prevention services, including HIV-1 testing, evaluation of adherence, safety evaluations, risk-reduction counseling, condoms, management of sexually transmitted infections, and assessment of sexual behavior.
Trial participants maintained a high risk of sexual HIV-1 acquisition, with high rates of rectal gonorrhea (DESCOVY, 24%; TRUVADA, 25%), rectal chlamydia (DESCOVY, 30%; TRUVADA, 31%), and syphilis (14% in both treatment groups) during the trial.
The primary outcome was the incidence of documented HIV-1 infection per 100 person-years in participants randomized to DESCOVY and TRUVADA (with a minimum follow-up of 48 weeks and at least 50% of participants having 96 weeks of follow-up). DESCOVY was non-inferior to TRUVADA in reducing the risk of acquiring HIV-1 infection (Table 14). The results were similar across the subgroups of age, race, gender identity, and baseline TRUVADA for PrEP use.
Table 14 HIV-1 Infection Results in DISCOVER Trial – Full Analysis Set DESCOVY
(N=2,670)TRUVADA
(N=2,665)Rate Ratio
(95% CI)4,370 person-years 4,386 person-years CI = Confidence interval. HIV-1 infections, n 7 15 Rate of HIV-1 infections per 100 person-years 0.16 0.34 0.468
(0.19, 1.15)Of the 22 participants diagnosed with HIV-1 infection in the trial, five had suspected baseline infection prior to study entry (DESCOVY, 1; TRUVADA, 4). In a case-control substudy of intracellular drug levels and estimated number of daily doses as measured by dried blood spot testing, median intracellular tenofovir diphosphate concentrations were substantially lower in participants infected with HIV-1 at the time of diagnosis compared with uninfected matched control participants. For both DESCOVY and TRUVADA, efficacy was therefore strongly correlated to adherence to daily dosing.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
DESCOVY 200 mg/25 mg tablets are blue, rectangular-shaped, and film-coated with "GSI" debossed on one side and "225" on the other side.
Each bottle contains 30 tablets (NDC: 61958-2002-1), a silica gel desiccant, polyester coil, and is closed with a child-resistant closure.
- Keep container tightly closed.
- Dispense only in original container.
Each blister pack contains 30 tablets (NDC: 61958-2002-2).
-
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Important Information for Uninfected Individuals Taking DESCOVY for HIV-1 PrEP
Advise HIV-1 uninfected individuals about the following [see Warnings and Precautions (5.2)]:
- The need to confirm that they are HIV-negative before starting to take DESCOVY to reduce the risk of acquiring HIV-1.
- That HIV-1 resistance substitutions may emerge in individuals with undetected HIV-1 infection who are taking DESCOVY, because DESCOVY alone does not constitute a complete regimen for HIV-1 treatment.
- The importance of taking DESCOVY on a regular dosing schedule and strict adherence to the recommended dosing schedule to reduce the risk of acquiring HIV-1. Uninfected individuals who miss doses are at greater risk of acquiring HIV-1 than those who do not miss doses.
- That DESCOVY does not prevent other sexually acquired infections and should be used as part of a complete prevention strategy including other prevention measures.
- To use condoms consistently and correctly to lower the chances of sexual contact with any body fluids such as semen, vaginal secretions, or blood.
- The importance of knowing their HIV-1 status and the HIV-1 status of their partner(s).
- The importance of virologic suppression in their partner(s) with HIV-1.
- The need to get tested regularly for HIV-1 (at least every 3 months, or more frequently for some individuals such as adolescents) and to ask their partner(s) to get tested as well.
- To report any symptoms of acute HIV-1 infection (flu-like symptoms) to their healthcare provider immediately.
- That the signs and symptoms of acute infection include fever, headache, fatigue, arthralgia, vomiting, myalgia, diarrhea, pharyngitis, rash, night sweats, and adenopathy (cervical and inguinal).
- To get tested for other sexually transmitted infections, such as syphilis, chlamydia, and gonorrhea, that may facilitate HIV-1 transmission.
- To assess their sexual risk behavior and get support to help reduce sexual risk behavior.
Post-treatment Acute Exacerbation of Hepatitis B in Patients with HBV Infection
Inform individuals that severe acute exacerbations of hepatitis B have been reported in patients who are infected with HBV and have discontinued products containing FTC and/or TDF and may likewise occur with discontinuation of DESCOVY [see Warnings and Precautions (5.1)]. Advise HBV-infected individuals to not discontinue DESCOVY without first informing their healthcare provider.
Immune Reconstitution Syndrome
Advise HIV-1 infected patients to inform their healthcare provider immediately of any symptoms of infection. In some patients with advanced HIV infection (AIDS), signs and symptoms of inflammation from previous infections may occur soon after anti-HIV treatment is started [see Warnings and Precautions (5.3)].
New Onset or Worsening Renal Impairment
Advise HIV-1 infected patients and uninfected individuals to avoid taking DESCOVY with concurrent or recent use of nephrotoxic agents. Renal impairment, including cases of acute renal failure, has been reported in association with the use of tenofovir prodrugs [see Warnings and Precautions (5.4)].
Lactic Acidosis and Severe Hepatomegaly
Lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been reported with use of drugs similar to DESCOVY. Advise HIV-1 infected patients and uninfected individuals that they should stop DESCOVY if they develop clinical symptoms suggestive of lactic acidosis or pronounced hepatotoxicity [see Warnings and Precautions (5.5)].
Dosage Recommendations for Treatment of HIV-1 Infection
Inform HIV-1 infected patients that it is important to take DESCOVY with other antiretroviral drugs for the treatment of HIV-1 on a regular dosing schedule with or without food and to avoid missing doses as it can result in development of resistance [see Dosage and Administration (2.3)].
Pregnancy Registry
Inform individuals using DESCOVY that there is an antiretroviral pregnancy registry to monitor fetal outcomes of pregnant women exposed to DESCOVY [see Use in Specific Populations (8.1)].
Lactation
Instruct mothers with HIV-1 infection not to breastfeed because of the risk of passing the HIV-1 virus to the baby [see Use in Specific Populations (8.2)].
- SPL UNCLASSIFIED SECTION
-
MEDICATION GUIDE
Medication Guide
DESCOVY® (des-KOH-vee)
(emtricitabine and tenofovir alafenamide)
tabletsThis Medication Guide has been approved by the U.S. Food and Drug Administration Revised: 12/2019 Read this Medication Guide before you start taking DESCOVY and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or your treatment.
This Medication Guide provides information about two different ways that DESCOVY may be used. See the section "What is DESCOVY?" for detailed information about how DESCOVY may be used.What is the most important information I should know about DESCOVY?
DESCOVY can cause serious side effects, including:-
Worsening of hepatitis B virus infection (HBV). Your healthcare provider will test you for HBV infection before or when you start treatment with DESCOVY. If you have HBV infection and take DESCOVY, your HBV may get worse (flare-up) if you stop taking DESCOVY. A "flare-up" is when your HBV infection suddenly returns in a worse way than before.
- Do not run out of DESCOVY. Refill your prescription or talk to your healthcare provider before your DESCOVY is all gone.
- Do not stop taking DESCOVY without first talking to your healthcare provider.
- If you stop taking DESCOVY, your healthcare provider will need to check your health often and do blood tests regularly for several months to check your HBV infection, or give you a medicine to treat hepatitis B. Tell your healthcare provider about any new or unusual symptoms you may have after you stop taking DESCOVY.
Other important information for people who take DESCOVY to help reduce their risk of getting human immunodeficiency virus-1 (HIV-1) infection, also called pre-exposure prophylaxis or "PrEP":
Before taking DESCOVY to reduce your risk of getting HIV-1:- You must be HIV-1 negative to start DESCOVY. You must get tested to make sure that you do not already have HIV-1 infection.
- Do not take DESCOVY for HIV-1 PrEP unless you are confirmed to be HIV-1 negative.
- Some HIV-1 tests can miss HIV-1 infection in a person who has recently become infected. If you have flu-like symptoms, you could have recently become infected with HIV-1. Tell your healthcare provider if you had a flu-like illness within the last month before starting DESCOVY or at any time while taking DESCOVY. Symptoms of new HIV-1 infection include:
- tiredness
- fever
- joint or muscle aches
- headache
- sore throat
- vomiting or diarrhea
- rash
- night sweats
- enlarged lymph nodes in the neck or groin
While you are taking DESCOVY for HIV-1 PrEP: - DESCOVY does not prevent other sexually transmitted infections (STIs). Practice safer sex by using a latex or polyurethane condom to reduce the risk of getting STIs.
-
You must stay HIV-1 negative to keep taking DESCOVY for HIV-1 PrEP.
- Know your HIV-1 status and the HIV-1 status of your partners.
- Ask your partners with HIV-1 if they are taking HIV-1 medicines and have an undetectable viral load. An undetectable viral load is when the amount of virus in the blood is too low to be measured in a lab test. To maintain an undetectable viral load, your partners must keep taking HIV-1 medicines every day. Your risk of getting HIV-1 is lower if your partners with HIV-1 are taking effective treatment.
- Get tested for HIV-1 at least every 3 months or when your healthcare provider tells you.
- Get tested for other STIs such as syphilis, chlamydia, and gonorrhea. These infections make it easier for HIV-1 to infect you.
- If you think you were exposed to HIV-1, tell your healthcare provider right away. They may want to do more tests to be sure you are still HIV-1 negative.
- Get information and support to help reduce sexual risk behaviors.
- Do not miss any doses of DESCOVY. Missing doses increases your risk of getting HIV-1 infection.
- If you do become HIV-1 positive, you need more medicine than DESCOVY alone to treat HIV-1. DESCOVY by itself is not a complete treatment for HIV-1.
What is DESCOVY?
DESCOVY is a prescription medicine that may be used in two different ways. DESCOVY is used:- to treat HIV-1 infection
- in adults and children who weigh at least 77 pounds (35 kg) together with other HIV-1 medicines
- in children who weigh at least 55 pounds (25 kg) and less than 77 pounds (35 kg) together with certain other HIV-1 medicines. Your healthcare provider will determine which other HIV-1 medicines may be used with DESCOVY.
- for HIV-1 PrEP to reduce the risk of getting HIV-1 infection in adults and adolescents who weigh at least 77 pounds (35 kg). It is not known if DESCOVY is effective in reducing the risk of getting HIV-1 from certain types of sex.
- DESCOVY for PrEP is not for use in people born female (assigned female at birth) who are at risk of getting HIV-1 infection from vaginal sex, because its effectiveness has not been studied.
DESCOVY contains the prescription medicines emtricitabine and tenofovir alafenamide.
It is not known if DESCOVY for treatment of HIV-1 infection is safe and effective in children who weigh less than 55 pounds (25 kg).
It is not known if DESCOVY is safe and effective in reducing the risk of HIV-1 infection in people who weigh less than 77 pounds (35 kg).For people taking DESCOVY for HIV-1 PrEP:
Do not take DESCOVY for HIV-1 PrEP if:- you already have HIV-1 infection. If you are HIV-1 positive, you need to take other medicines with DESCOVY to treat HIV-1. DESCOVY by itself is not a complete treatment for HIV-1.
- you do not know your HIV-1 infection status. You may already be HIV-1 positive. You need to take other HIV-1 medicines with DESCOVY to treat HIV-1 infection.
What should I tell my healthcare provider before taking DESCOVY?
Before taking DESCOVY, tell your healthcare provider about all of your medical conditions, including if you:- have liver problems, including HBV infection
- have kidney problems
- are pregnant or plan to become pregnant. It is not known if DESCOVY can harm your unborn baby. Tell your healthcare provider if you become pregnant during treatment with DESCOVY.
Pregnancy Registry: There is a pregnancy registry for people who take DESCOVY during pregnancy. The purpose of this registry is to collect information about the health of you and your baby. Talk with your healthcare provider about how you can take part in this registry. - are breastfeeding or plan to breastfeed.
- Do not breastfeed if you take DESCOVY for treatment of HIV-1 because of the risk of passing HIV-1 to your baby.
- One of the ingredients in DESCOVY (emtricitabine) passes into your breast milk.
Some medicines may interact with DESCOVY. Keep a list of your medicines and show it to your healthcare provider and pharmacist when you get a new medicine.- You can ask your healthcare provider or pharmacist for a list of medicines that interact with DESCOVY.
- Do not start a new medicine without telling your healthcare provider. Your healthcare provider can tell you if it is safe to take DESCOVY with other medicines.
How should I take DESCOVY? - Take DESCOVY exactly as your healthcare provider tells you to take it. If you take DESCOVY to treat HIV-1 infection, you need to take DESCOVY with other HIV-1 medicines. Your healthcare provider will tell you what medicines to take and how to take them.
- Take DESCOVY 1 time each day with or without food.
- If you are on dialysis, take your daily dose of DESCOVY following dialysis.
- Do not change your dose or stop taking DESCOVY without first talking with your healthcare provider. Stay under a healthcare provider's care when taking DESCOVY. Do not miss a dose of DESCOVY.
- If you take too much DESCOVY, call your healthcare provider or go to the nearest hospital emergency room right away.
- When your DESCOVY supply starts to run low, get more from your healthcare provider or pharmacy.
- If you are taking DESCOVY for treatment of HIV-1, the amount of virus in your blood may increase if the medicine is stopped for even a short time. The virus may develop resistance to DESCOVY and become harder to treat.
- If you are taking DESCOVY for HIV-1 PrEP, missing doses increases your risk of getting HIV-1 infection.
What are the possible side effects of DESCOVY?
DESCOVY may cause serious side effects, including:- See "What is the most important information I should know about DESCOVY?"
- Changes in your immune system (Immune Reconstitution Syndrome) can happen when you start taking medicines to treat HIV-1 infection. Your immune system may get stronger and begin to fight infections that have been hidden in your body for a long time. Tell your healthcare provider right away if you start having any new symptoms after starting your HIV-1 medicine.
- New or worse kidney problems, including kidney failure. Your healthcare provider should do blood and urine tests to check your kidneys before you start and while taking DESCOVY. Your healthcare provider may tell you to stop taking DESCOVY if you develop new or worse kidney problems.
- Too much lactic acid in your blood (lactic acidosis). Too much lactic acid is a serious but rare medical emergency that can lead to death. Tell your healthcare provider right away if you get these symptoms: weakness or being more tired than usual, unusual muscle pain, being short of breath or fast breathing, stomach pain with nausea and vomiting, cold or blue hands and feet, feel dizzy or lightheaded, or a fast or abnormal heartbeat.
- Severe liver problems. In rare cases, severe liver problems can happen that can lead to death. Tell your healthcare provider right away if you get these symptoms: skin or the white part of your eyes turns yellow, dark "tea-colored" urine, light-colored stools, loss of appetite for several days or longer, nausea, or stomach-area pain.
The most common side effect of DESCOVY for HIV-1 PrEP is diarrhea.
These are not all of the possible side effects of DESCOVY.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.How should I store DESCOVY? - Store DESCOVY between 68°F to 77°F (20°C to 25°C).
- Keep DESCOVY in its original container.
- Keep the container tightly closed.
General information about the safe and effective use of DESCOVY.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use DESCOVY for a condition for which it was not prescribed. Do not give DESCOVY to other people, even if they have the same symptoms you have. It may harm them. You can ask your healthcare provider or pharmacist for information about DESCOVY that is written for health professionals.What are the ingredients in DESCOVY?
Active ingredients: emtricitabine and tenofovir alafenamide.
Inactive ingredients: croscarmellose sodium, magnesium stearate, and microcrystalline cellulose.
The tablets are film-coated with a coating material containing indigo carmine aluminum lake, polyethylene glycol, polyvinyl alcohol, talc, and titanium dioxide.
Manufactured and distributed by: Gilead Sciences, Inc. Foster City, CA 94404
DESCOVY is a trademark of Gilead Sciences, Inc., or its related companies. All other trademarks referenced herein are the property of their respective owners.
© 2020 Gilead Sciences, Inc. All rights reserved.
208215-GS-006
For more information, call 1-800-445-3235 or go to www.DESCOVY.com. -
Worsening of hepatitis B virus infection (HBV). Your healthcare provider will test you for HBV infection before or when you start treatment with DESCOVY. If you have HBV infection and take DESCOVY, your HBV may get worse (flare-up) if you stop taking DESCOVY. A "flare-up" is when your HBV infection suddenly returns in a worse way than before.
- PRINCIPAL DISPLAY PANEL - 200 mg/25 mg Tablet Bottle Label
-
PRINCIPAL DISPLAY PANEL - 30 Tablet Blister Pack
NDC: 61958-2002-2
30 tablet blister packDescovy®
(emtricitabine and
tenofovir alafenamide) tablets
200 mg / 25 mgRx only
Note to pharmacist:
Do not cover ALERT box with pharmacy label.ALERT: Find out about medicines that
should NOT be taken with Descovy
-
INGREDIENTS AND APPEARANCE
DESCOVY
emtricitabine and tenofovir alafenamide tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 61958-2002 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength emtricitabine (UNII: G70B4ETF4S) (emtricitabine - UNII:G70B4ETF4S) emtricitabine 200 mg tenofovir alafenamide fumarate (UNII: FWF6Q91TZO) (tenofovir anhydrous - UNII:W4HFE001U5) tenofovir alafenamide 25 mg Inactive Ingredients Ingredient Name Strength MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) MAGNESIUM STEARATE (UNII: 70097M6I30) WATER (UNII: 059QF0KO0R) POLYVINYL ALCOHOL, UNSPECIFIED (UNII: 532B59J990) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) POLYETHYLENE GLYCOL 3350 (UNII: G2M7P15E5P) TALC (UNII: 7SEV7J4R1U) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) Product Characteristics Color BLUE Score no score Shape OVAL (rectangular-shaped) Size 13mm Flavor Imprint Code GSI;255 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 61958-2002-1 30 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 04/04/2016 2 NDC: 61958-2002-2 30 in 1 BLISTER PACK; Type 0: Not a Combination Product 01/22/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA208215 04/04/2016 Labeler - Gilead Sciences, Inc. (185049848)
Trademark Results [DESCOVY]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 DESCOVY 86170326 4876632 Live/Registered |
Gilead Sciences Ireland UC 2014-01-20 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.
