FENTANYL patch, extended release
Fentanyl by
Drug Labeling and Warnings
Fentanyl by is a Prescription medication manufactured, distributed, or labeled by Dispensing Solutions Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
Full Prescribing Information
FOR USE IN OPIOID-TOLERANT PATIENTS ONLY
Fentanyl transdermal system contains a high concentration of a potent Schedule II opioid agonist, fentanyl. Schedule II opioid substances which include fentanyl, hydromorphone, methadone, morphine, oxycodone, and oxymorphone have the highest potential for abuse and associated risk of fatal overdose due to respiratory depression. Fentanyl can be abused and is subject to criminal diversion. The high content of fentanyl in the patches (fentanyl transdermal system) may be a particular target for abuse and diversion.
Fentanyl transdermal system is indicated for management of persistent, moderate to severe chronic pain that:
- requires continuous, around-the-clock opioid administration for an extended period of time, and
- cannot be managed by other means such as non-steroidal analgesics, opioid combination products, or immediate-release opioids
Fentanyl transdermal system should ONLY be used in patients who are already receiving opioid therapy, who have demonstrated opioid tolerance, and who require a total daily dose at least equivalent to fentanyl transdermal system 25 mcg/hr. Patients who are considered opioid-tolerant are those who have been taking, for a week or longer, at least 60 mg of morphine daily, or at least 30 mg of oral oxycodone daily, or at least 8 mg of oral hydromorphone daily or an equianalgesic dose of another opioid.
Because serious or life-threatening hypoventilation could occur, fentanyl transdermal system is contraindicated:
- in patients who are not opioid-tolerant
- in the management of acute pain or in patients who require opioid analgesia for a short period of time
- in the management of post-operative pain, including use after out-patient or day surgeries (e.g., tonsillectomies)
- in the management of mild pain
- in the management of intermittent pain (e.g., use on an as needed basis [prn])
(See CONTRAINDICATIONS for further information.)
Since the peak fentanyl levels occur between 24 and 72 hours of treatment, prescribers should be aware that serious or life threatening hypoventilation may occur, even in opioid-tolerant patients, during the initial application period.
The concomitant use of fentanyl transdermal system with all cytochrome P450 3A4 inhibitors (such as ritonavir, ketoconazole, itraconazole, troleandomycin, clarithromycin, nelfinavir, nefazodone, amiodarone, amprenavir, aprepitant, diltiazem, erythromycin, fluconazole, fosamprenavir, grapefruit juice, and verapamil) may result in an increase in fentanyl plasma concentrations, which could increase or prolong adverse drug effects and may cause potentially fatal respiratory depression. Patients receiving fentanyl transdermal system and any CYP3A4 inhibitor should be carefully monitored for an extended period of time and dosage adjustments should be made if warranted (see CLINICAL PHARMACOLOGY, Drug Interactions; WARNINGS; PRECAUTIONS; and DOSAGE AND ADMINISTRATION for further information).
The safety of fentanyl transdermal system has not been established in children under 2 years of age. Fentanyl transdermal system should be administered to children only if they are opioid-tolerant and 2 years of age or older (see PRECAUTIONS, Pediatric Use).
Fentanyl transdermal system is ONLY for use in patients who are already tolerant to opioid therapy of comparable potency. Use in non-opioid tolerant patients may lead to fatal respiratory depression. Overestimating the fentanyl transdermal system dose when converting patients from another opioid medication can result in fatal overdose with the first dose. Due to the mean elimination half-life of 17 hours of fentanyl transdermal system, patients who are thought to have had a serious adverse event, including overdose, will require monitoring and treatment for at least 24 hours.
Fentanyl transdermal system can be abused in a manner similar to other opioid agonists, legal or illicit. This risk should be considered when administering, prescribing, or dispensing fentanyl transdermal system in situations where the healthcare professional is concerned about increased risk of misuse, abuse or diversion.
Persons at increased risk for opioid abuse include those with a personal or family history of substance abuse (including drug or alcohol abuse or addiction) or mental illness (e.g., major depression). Patients should be assessed for their clinical risks for opioid abuse or addiction prior to being prescribed opioids. All patients receiving opioids should be routinely monitored for signs of misuse, abuse and addiction. Patients at increased risk of opioid abuse may still be appropriately treated with modified-release opioid formulations; however, these patients will require intensive monitoring for signs of misuse, abuse, or addiction.
Fentanyl transdermal systems are intended for transdermal use (on intact skin) only. Do not use a fentanyl transdermal system if the seal is broken or the patch is cut, damaged, or changed in any way.
Avoid exposing the fentanyl transdermal system application site and surrounding area to direct external heat sources, such as heating pads or electric blankets, heat or tanning lamps, saunas, hot tubs, and heated water beds, while wearing the system. Avoid taking hot baths or sunbathing. There is a potential for temperature-dependent increases in fentanyl released from the system resulting in possible overdose and death. Patients wearing fentanyl transdermal systems who develop fever or increased core body temperature due to strenuous exertion should be monitored for opioid side effects and the fentanyl transdermal system dose should be adjusted if necessary.
-
DESCRIPTION
Fentanyl transdermal system is a transdermal system providing continuous systemic delivery of fentanyl, a potent opioid analgesic, for 72 hours. The chemical name is N-phenyl-N-[1-(2-phenylethyl)-4-piperidyl] propanamide. The structural formula is:
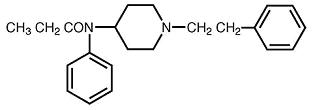
C22H28N2O M.W. 336.5
The n-octanol:water partition coefficient is 860:1. The pKa is 8.4.
System Components and Structure
The amount of fentanyl released from each system per hour is proportional to the surface area (25 mcg/hr per 10.7 cm2). The composition per unit area of all system sizes is identical.
Dose* (mcg/hr) Size (cm2) Fentanyl Content (mg) - * Nominal delivery rate per hour
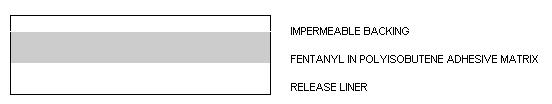
25 10.7 2.76 50 21.4 5.52 75 32.1 8.28 100 42.8 11.04 Fentanyl transdermal system is a rectangular unit comprising a protective liner and two functional layers. Proceeding from the outer surface toward the surface adhering to skin, these layers are:
- A BACKING LAYER OF POLYESTER FILM;
- FENTANYL IN A POLYISOBUTENE ADHESIVE MATRIX THAT CONTROLS THE RATE OF FENTANYL DELIVERY TO THE SKIN SURFACE; AND
- A PROTECTIVE POLYESTER RELEASE LINER.
Before use, a protective liner covering the adhesive layer is removed and discarded.
The active component of the system is fentanyl. The remaining components are pharmacologically inactive.
-
CLINICAL PHARMACOLOGY
Pharmacology
Fentanyl is an opioid analgesic. Fentanyl interacts predominantly with the opioid mu-receptor. These mu-binding sites are discretely distributed in the human brain, spinal cord, and other tissues. In clinical settings, fentanyl exerts its principal pharmacologic effects on the central nervous system.
In addition to analgesia, alterations in mood, euphoria, dysphoria, and drowsiness commonly occur. Fentanyl depresses the respiratory centers, depresses the cough reflex, and constricts the pupils. Analgesic blood levels of fentanyl may cause nausea and vomiting directly by stimulating the chemoreceptor trigger zone, but nausea and vomiting are significantly more common in ambulatory than in recumbent patients, as is postural syncope.
Opioids increase the tone and decrease the propulsive contractions of the smooth muscle of the gastrointestinal tract. The resultant prolongation in gastrointestinal transit time may be responsible for the constipating effect of fentanyl. Because opioids may increase biliary tract pressure, some patients with biliary colic may experience worsening rather than relief of pain.
While opioids generally increase the tone of urinary tract smooth muscle, the net effect tends to be variable, in some cases producing urinary urgency, in others, difficulty in urination. At therapeutic dosages, fentanyl usually does not exert major effects on the cardiovascular system. However, some patients may exhibit orthostatic hypotension and fainting.
Histamine assays and skin wheal testing in clinical studies indicate that clinically significant histamine release rarely occurs with fentanyl administration. Clinical assays show no clinically significant histamine release in dosages up to 50 mcg/kg.
Pharmacokinetics (see graph and tables)
Fentanyl transdermal system releases fentanyl from the drug matrix at a nearly constant amount per unit time. The concentration gradient existing between the adhesive matrix and the lower concentration in the skin drives drug release. Fentanyl moves in the direction of the lower concentration at a rate determined by the diffusion of fentanyl through the skin layers. While the actual rate of fentanyl delivery to the skin varies over the 72 hour application period, each system is labeled with a nominal flux which represents the average amount of drug delivered to the systemic circulation per hour across average skin.
While there is variation in dose delivered among patients, the nominal flux of the systems (25, 50, 75, and 100 mcg of fentanyl per hour) is sufficiently accurate as to allow individual titration of dosage for a given patient.
Following fentanyl transdermal system application, the skin under the system absorbs fentanyl, and a depot of fentanyl concentrates in the upper skin layers. Fentanyl then becomes available to the systemic circulation. Serum fentanyl concentrations increase gradually following initial fentanyl transdermal system application, generally leveling off between 12 and 24 hours and remaining relatively constant, with some fluctuation, for the remainder of the 72 hour application period. Peak serum concentrations of fentanyl generally occurred between 24 and 72 hours after initial application (see TABLE A). Serum fentanyl concentrations achieved are proportional to the fentanyl transdermal system delivery rate. With continuous use, serum fentanyl concentrations continue to rise for the first few system applications. After several sequential 72 hour applications, patients reach and maintain a steady state serum concentration that is determined by individual variation in skin permeability and body clearance of fentanyl (see graph and TABLE B).
The kinetics of fentanyl in normal subjects following application of a 25 mcg/hr fentanyl transdermal system were bioequivalent with or without a Bioclusive™ overlay (polyurethane film dressing).
After system removal, serum fentanyl concentrations decline gradually, falling about 50% in approximately 17 (range 13 to 22) hours. Continued absorption of fentanyl from the skin accounts for a slower disappearance of the drug from the serum than is seen after an IV infusion, where the apparent half-life is approximately 7 (range 3 to 12) hours.
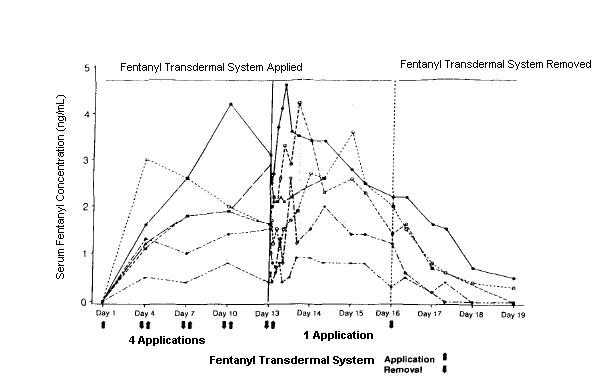
Serum Fentanyl Concentrations Following Multiple Applications of A Fentanyl Transdermal System 100 mcg/hr (n = 10)
TABLE A FENTANYL PHARMACOKINETIC PARAMETERS FOLLOWING FIRST 72 HOUR APPLICATION OF A FENTANYL TRANSDERMAL SYSTEM Mean (SD) Time to Maximal Concentration Tmax (hr) Mean (SD) Maximal Concentration Cmax (ng/mL) Fentanyl Transdermal System 25 mcg/hr 38.1 (18.0) 0.6 (0.3) Fentanyl Transdermal System 50 mcg/hr 34.8 (15.4) 1.4 (0.5) Fentanyl Transdermal System 75 mcg/hr 33.5 (14.5) 1.7 (0.7) Fentanyl Transdermal System 100 mcg/hr 36.8 (15.7) 2.5 (1.2) NOTE:After system removal there is continued systemic absorption from residual fentanyl in the skin so that serum concentrations fall 50%, on average, in 17 hours.
TABLE B RANGE OF PHARMACOKINETIC PARAMETERS OF INTRAVENOUS FENTANYL IN PATIENTS Clearance (L/hr) Range (70 kg) Volume of Distribution Vss (L/kg) Range Half-Life t½ (hr) Range - * Estimated
Surgical Patients 27 to 75 3 to 8 3 to 12 Hepatically Impaired Patients 3 to 80* 0.8 to 8* 4 to 12* Renally Impaired Patients 30 to 78 - - NOTE:Information on volume of distribution and half-life not available for renally impaired patients.
Fentanyl plasma protein binding capacity decreases with increasing ionization of the drug. Alterations in pH may affect its distribution between plasma and the central nervous system. Fentanyl accumulates in the skeletal muscle and fat and is released slowly into the blood. The average volume of distribution for fentanyl is 6 L/kg (range 3 to 8; N = 8).
Fentanyl is metabolized primarily via human cytochrome P450 3A4 isoenzyme system. In humans, the drug appears to be metabolized primarily by oxidative N-dealkylation to norfentanyl and other inactive metabolites that do not contribute materially to the observed activity of the drug. Within 72 hours of IV fentanyl administration, approximately 75% of the dose is excreted in urine, mostly as metabolites with less than 10% representing unchanged drug. Approximately 9% of the dose is recovered in the feces, primarily as metabolites. Mean values for unbound fractions of fentanyl in plasma are estimated to be between 13 and 21%.
Skin does not appear to metabolize fentanyl delivered transdermally. This was determined in a human keratinocyte cell assay and in clinical studies in which 92% of the dose delivered from the system was accounted for as unchanged fentanyl that appeared in the systemic circulation.
Hepatic or Renal Disease
Insufficient information exists to make recommendations regarding the use of fentanyl transdermal system in patients with impaired renal or hepatic function. Fentanyl is metabolized primarily via human cytochrome P450 3A4 isoenzyme system and mostly eliminated in urine. If the drug is used in these patients, it should be used with caution because of the hepatic metabolism and renal excretion of fentanyl.
Pediatric Use
In 1.5 to 5 year old, non-opioid-tolerant pediatric patients, the fentanyl plasma concentrations were approximately twice as high as that of adult patients. In older pediatric patients, the pharmacokinetic parameters were similar to that of adults. However, these findings have been taken into consideration in determining the dosing recommendations for opioid-tolerant pediatric patients (2 years of age and older). For pediatric dosing information, refer to DOSAGE AND ADMINISTRATION section.
Geriatric Use
Information from a pilot study of the pharmacokinetics of IV fentanyl in geriatric patients (N = 4) indicates that the clearance of fentanyl may be greatly decreased in the population above the age of 60. The relevance of these fndings to fentanyl transdermal system is unknown at this time.
Respiratory depression is the chief hazard in elderly or debilitated patients, usually following large initial doses in non-tolerant patients or when opioids are given in conjunction with other agents that depress respiration.
Fentanyl transdermal system should be used with caution in elderly, cachetic or debilitated patients as they may have altered pharmacokinetics due to poor fat stores, muscle wasting, or altered clearance (see DOSAGE AND ADMINISTRATION).
Drug Interactions
The interaction between ritonavir, a CYP3A4 inhibitor, and fentanyl was investigated in eleven healthy volunteers in a randomized crossover study. Subjects received oral ritonavir or placebo for 3 days. The ritonavir dose was 200 mg tid on Day 1 and 300 mg tid on Day 2 followed by one morning dose of 300 mg on Day 3. On Day 2, fentanyl was given as a single IV dose at 5 mcg/kg two hours after the afternoon dose of oral ritonavir or placebo. Naloxone was administered to counteract the side effects of fentanyl. The results suggested that ritonavir might decrease the clearance of fentanyl by 67%, resulting in a 174% (range 52% to 420%) increase in fentanyl AUC0-∞. Coadministration of ritonavir in patients receiving fentanyl transdermal system has not been studied; however, an increase in fentanyl AUC is expected (see BOX WARNING, WARNINGS, PRECAUTIONS and DOSAGE AND ADMINISTRATION).
Fentanyl is metabolized mainly via the human cytochrome P450 3A4 isoenzyme system (CYP3A4), therefore, potential interactions may occur when fentanyl transdermal system is given concurrently with agents that affect CYP3A4 activity. Coadministration with agents that induce CYP3A4 activity may reduce the efficacy of fentanyl transdermal system. The concomitant use of transdermal fentanyl with all CYP3A4 inhibitors (such as ritonavir, ketoconazole, itraconazole, troleandomycin, clarithromycin, nelfinavir, nefazadone, amiodarone, amprenavir, aprepitant, diltiazem, erythromycin, fluconazole, fosamprenavir, grapefruit juice, and verapamil) may result in an increase in fentanyl plasma concentrations, which could increase or prolong adverse drug effects and may cause potentially fatal respiratory depression. Patients receiving fentanyl transdermal system and any CYP3A4 inhibitor should be carefully monitored for an extended period of time and dosage adjustments should be made if warranted (see BOXWARNING, WARNINGS, PRECAUTIONS, and DOSAGE AND ADMINISTRATION for further information).
Ventilatory Effects
Because of the risk for serious or life-threatening hypoventilation, fentanyl transdermal system is CONTRAINDICATED in the treatment of post-operative and acute pain and in patients who are not opioid-tolerant. In clinical trials of 357 patients with acute pain treated with fentanyl transdermal system, 13 patients experienced hypoventilation. Hypoventilation was manifested by respiratory rates of less than 8 breaths/minute or a pCO2 greater than 55 mm Hg. In these studies, the incidence of hypoventilation was higher in nontolerant women (10) than in men (3) and in patients weighing less than 63 kg (9 of 13). Although patients with impaired respiration were not common in the trials, they had higher rates of hypoventilation. In addition, postmarketing reports have been received that describe opioid-naïve post-operative patients who have experienced clinically significant hypoventilation and death with fentanyl transdermal system.
While most adult and pediatric patients using fentanyl transdermal system chronically develop tolerance to fentanyl induced hypoventilation, episodes of slowed respirations may occur at any time during therapy.
Hypoventilation can occur throughout the therapeutic range of fentanyl serum concentrations, especially for patients who have an underlying pulmonary condition or who receive usual doses of opioids or other CNS drugs associated with hypoventilation in addition to fentanyl transdermal system. The use of fentanyl transdermal system is contraindicated in patients who are not tolerant to opioid therapy.
The use of fentanyl transdermal system should be monitored by clinical evaluation, especially within the initial 24 to 72 hours when serum concentrations from the initial patch will peak, and following increases in dosage. Fentanyl transdermal system should be administered to children only if they are opioid-tolerant and 2 years of age or older.
See BOX WARNING, CONTRAINDICATIONS, WARNINGS, PRECAUTIONS, ADVERSE REACTIONS and OVERDOSAGE for additional information on hypoventilation.
-
INDICATIONS AND USAGE
Fentanyl transdermal system is indicated for management of persistent, moderate to severe chronic pain that:
- requires continuous, around-the-clock opioid administration for an extended period of time, and
- cannot be managed by other means such as non-steroidal analgesics, opioid combination products, or immediate-release opioids
Fentanyl transdermal system should ONLY be used in patients who are already receiving opioid therapy, who have demonstrated opioid tolerance, and who require a total daily dose at least equivalent to fentanyl transdermal system 25 mcg/hr (see DOSAGE ANDADMINISTRATION). Patients who are considered opioid-tolerant are those who have been taking, for a week or longer, at least 60 mg of morphine daily, or at least 30 mg of oral oxycodone daily, or at least 8 mg of oral hydromorphone daily, or an equianalgesic dose of another opioid.
Because serious or life-threatening hypoventilation could result, fentanyl transdermal system is contraindicated for use on an as needed basis (i.e., prn), for the management of post-operative or acute pain, or in patients who are not opioid-tolerant or who require opioid analgesia for a short period of time (see BOX WARNING and CONTRAINDICATIONS).
An evaluation of the appropriateness and adequacy of treating with immediate-release opioids is advisable prior to initiating therapy with any modified-release opioid. Prescribers should individualize treatment in every case, initiating therapy at the appropriate point along a progression from non-opioid analgesics, such as non-steroidal anti-inflammatory drugs and acetaminophen, to opioids, in a plan of pain management such as outlined by the World Health Organization, the Agency for Health Research and Quality, the Federation of State Medical Boards Model Policy, or the American Pain Society.
Patients should be assessed for their clinical risks for opioid abuse or addiction prior to being prescribed opioids. Patients receiving opioids should be routinely monitored for signs of misuse, abuse, and addiction. Persons at increased risk for opioid abuse include those with a personal or family history of substance abuse (including drug or alcohol abuse or addiction) or mental illness (e.g., major depression). Patients at increased risk may still be appropriately treated with modified-release opioid formulations; however these patients will require intensive monitoring for signs of misuse, abuse, or addiction.
-
CONTRAINDICATIONS
Because serious or life-threatening hypoventilation could occur, fentanyl transdermal system is contraindicated:
- in patients who are not opioid-tolerant
- in the management of acute pain or in patients who require opioid analgesia for a short period of time
- in the management of post-operative pain, including use after out-patient or day surgeries, (e.g., tonsillectomies)
- in the management of mild pain
- in the management of intermittent pain (e.g., use on an as needed basis [prn])
- in situations of significant respiratory depression, especially in unmonitored settings where there is a lack of resuscitative equipment
- in patients who have acute or severe bronchial asthma
Fentanyl transdermal system is contraindicated in patients who have or are suspected of having paralytic ileus.
Fentanyl transdermal system is contraindicated in patients with known hypersensitivity to fentanyl or any components of this product.
-
WARNINGS
Fentanyl transdermal system is intended for transdermal use (on intact skin) only. Do not use a fentanyl transdermal system if the seal is broken or the patch is cut, damaged, or changed in any way.
The safety of fentanyl transdermal system has not been established in children under 2 years of age. Fentanyl transdermal system should be administered to children only if they are opioid-tolerant and 2 years of age or older (see PRECAUTIONS, Pediatric Use).
Fentanyl transdermal system is ONLY for use in patients who are already tolerant to opioid therapy of comparable potency. Use in non-opioid tolerant patients may lead to fatal respiratory depression. Overestimating the fentanyl transdermal system dose when converting patients from another opioid medication can result in fatal overdose with the first dose. The mean elimination half-life of fentanyl transdermal system is 17 hours. Therefore, patients who have experienced serious adverse events, including overdose, will require monitoring for at least 24 hours after fentanyl transdermal system removal since serum fentanyl concentrations decline gradually and reach an approximate 50% reduction in serum concentrations 17 hours after system removal.
Fentanyl transdermal system should be prescribed only by persons knowledgeable in the continuous administration of potent opioids, in the management of patients receiving potent opioids for treatment of pain, and in the detection and management of hypoventilation including the use of opioid antagonists.
All patients and their caregivers should be advised to avoid exposing the fentanyl transdermal system application site and surrounding area to direct external heat sources, such as heating pads or electric blankets, heat or tanning lamps, saunas, hot tubs, and heated water beds, etc., while wearing the system. Patients should be advised against taking hot baths or sunbathing. There is a potential for temperature-dependent increases in fentanyl released from the system resulting in possible overdose and death.
Based on a pharmacokinetic model, serum fentanyl concentrations could theoretically increase by approximately one-third for patients with a body temperature of 40ºC (104ºF) due to temperature-dependent increases in fentanyl released from the system and increased skin permeability. Patients wearing fentanyl transdermal systems who develop fever or increased core body temperature due to strenuous exertion should be monitored for opioid side effects and the fentanyl transdermal system dose should be adjusted if necessary.
Death and other serious medical problems have occurred when people were accidentally exposed to fentanyl transdermal system. Examples of accidental exposure include transfer of a fentanyl transdermal system from an adult’s body to a child while hugging, accidental sitting on a patch and possible accidental exposure of a caregiver’s skin to the medication in the patch while the caregiver was applying or removing the patch.
Placing fentanyl transdermal system in the mouth, chewing it, swallowing it, or using it in ways other than indicated may cause choking or overdose that could result in death.
Misuse, Abuse and Diversion of Opioids
Fentanyl is an opioid agonist of the morphine-type. Such drugs are sought by drug abusers and people with addiction disorders and are subject to criminal diversion.
Fentanyl can be abused in a manner similar to other opioids, legal or illicit. This should be considered when prescribing or dispensing fentanyl transdermal system in situations where the physician or pharmacist is concerned about an increased risk of misuse, abuse, or diversion.
Fentanyl transdermal system has been reported as being abused by other methods and routes of administration. These practices will result in uncontrolled delivery of the opioid and pose a significant risk to the abuser that could result in overdose and death (see WARNINGS and DRUG ABUSE AND ADDICTION).
Concerns about abuse, addiction, and diversion should not prevent the proper management of pain. However, all patients treated with opioids require careful monitoring for signs of abuse and addiction, since use of opioid analgesic products carries the risk of addiction even under appropriate medical use.
Healthcare professionals should contact their state professional licensing board or state controlled substances authority for information on how to prevent and detect abuse or diversion of this product.
Hypoventilation (Respiratory Depression)
Serious or life-threatening hypoventilation may occur at any time during the use of fentanyl transdermal system especially during the initial 24 to 72 hours following initiation of therapy and following increases in dose.
Because significant amounts of fentanyl are absorbed from the skin for 17 hours or more after the patch is removed, hypoventilation may persist beyond the removal of fentanyl transdermal system. Consequently, patients with hypoventilation should be carefully observed for degree of sedation and their respiratory rate monitored until respiration has stabilized.
The use of concomitant CNS active drugs requires special patient care and observation.
Respiratory depression is the chief hazard of opioid agonists, including fentanyl the active ingredient in fentanyl transdermal system. Respiratory depression is more likely to occur in elderly or debilitated patients, usually following large initial doses in non-tolerant patients, or when opioids are given in conjunction with other drugs that depress respiration.
Respiratory depression from opioids is manifested by a reduced urge to breathe and a decreased rate of respiration, often associated with the “sighing” pattern of breathing (deep breaths separated by abnormally long pauses). Carbon dioxide retention from opioid-induced respiratory depression can exacerbate the sedating effects of opioids. This makes overdoses involving drugs with sedative properties and opioids especially dangerous.
Fentanyl transdermal system should be used with extreme caution in patients with significant chronic obstructive pulmonary disease or cor pulmonale, and in patients having a substantially decreased respiratory reserve, hypoxia, hypercapnia, or preexisting respiratory depression. In such patients, even usual therapeutic doses of fentanyl transdermal system may decrease respiratory drive to the point of apnea. In these patients, alternative non-opioid analgesics should be considered, and opioids should be employed only under careful medical supervision at the lowest effective dose.
Chronic Pulmonary Disease
Because potent opioids can cause serious or life-threatening hypoventilation, fentanyl transdermal system should be administered with caution to patients with preexisting medical conditions predisposing them to hypoventilation. In such patients, normal analgesic doses of opioids may further decrease respiratory drive to the point of respiratory failure.
Head Injuries and Increased Intracranial Pressure
Fentanyl transdermal system should not be used in patients who may be particularly susceptible to the intracranial effects of CO2 retention such as those with evidence of increased intracranial pressure, impaired consciousness, or coma. Opioids may obscure the clinical course of patients with head injury. Fentanyl transdermal system should be used with caution in patients with brain tumors.
Interactions with other CNS Depressants
The concomitant use of fentanyl transdermal system with other central nervous system depressants, including but not limited to other opioids, sedatives, hypnotics, tranquilizers (e.g., benzodiazepines), general anesthetics, phenothiazines, skeletal muscle relaxants, and alcohol, may cause respiratory depression, hypotension, and profound sedation or potentially result in coma. When such combined therapy is contemplated, the dose of one or both agents should be significantly reduced.
Interactions with Alcohol and Drugs of Abuse
Fentanyl may be expected to have additive CNS depressant effects when used in conjunction with alcohol, other opioids, or illicit drugs that cause central nervous system depression.
Interactions with CYP3A4 Inhibitors
The concomitant use of fentanyl transdermal with all CYP3A4 inhibitors (such as ritonavir, ketoconazole, itraconazole, troleandomycin, clarithromycin, nelfinavir, nefazodone, amiodarone, amprenavir, aprepitant, diltiazem, erythromycin, fluconazole, fosamprenavir, grapefruit juice, and verapamil) may result in an increase in fentanyl plasma concentrations, which could increase or prolong adverse drug effects and may cause potentially fatal respiratory depression. Patients receiving fentanyl transdermal system and any CYP3A4 inhibitors should be carefully monitored for an extended period of time, and dosage adjustments should be made if warranted (see BOXWARNING; CLINICAL PHARMACOLOGY, Drug Interactions; PRECAUTIONS; and DOSAGE AND ADMINISTRATION for further information).
-
PRECAUTIONS
General
Fentanyl transdermal system should not be used to initiate opioid therapy in patients who are not opioid-tolerant. Children converting to fentanyl transdermal system should be opioid-tolerant and 2 years of age or older (see BOX WARNING).
Patients, family members, and caregivers should be instructed to keep patches (new and used) out of the reach of children and others for whom fentanyl transdermal system was not prescribed. A considerable amount of active fentanyl remains in fentanyl transdermal system even after use as directed. Accidental or deliberate application or ingestion by a child or adolescent will cause respiratory depression that could result in death.
Cardiac Disease
Fentanyl may produce bradycardia. Fentanyl should be administered with caution to patients with bradyarrhythmias.
Hepatic or Renal Disease
Insufficient information exists to make recommendations regarding the use of fentanyl transdermal system in patients with impaired renal or hepatic function. If the drug is used in these patients, it should be used with caution because of the hepatic metabolism and renal excretion of fentanyl.
Use in Pancreatic/Biliary Tract Disease
Fentanyl transdermal system may cause spasm of the sphincter of Oddi and should be used with caution in patients with biliary tract disease, including acute pancreatitis. Opioids like fentanyl transdermal system may cause increases in the serum amylase concentration.
Tolerance
Tolerance is a state of adaptation in which exposure to a drug induces changes that result in a diminution of one or more of the drug’s effects over time. Tolerance may occur to both the desired and undesired effects of drugs, and may develop at different rates for different effects.
Physical Dependence
Physical dependence is a state of adaptation that is manifested by an opioid specific withdrawal syndrome that can be produced by abrupt cessation, rapid dose reduction, decreasing blood level of the drug, and/or administration of an antagonist. The opioid abstinence or withdrawal syndrome is characterized by some or all of the following: restlessness, lacrimation, rhinorrhea, yawning, perspiration, chills, piloerection, myalgia, mydriasis, irritability, anxiety, backache, joint pain, weakness, abdominal cramps, insomnia, nausea, anorexia, vomiting, diarrhea, or increased blood pressure, respiratory rate, or heart rate. In general, opioids should not be abruptly discontinued (see DOSAGE AND ADMINISTRATION, Discontinuation of Fentanyl Transdermal System).
Ambulatory Patients
Strong opioid analgesics impair the mental or physical abilities required for the performance of potentially dangerous tasks, such as driving a car or operating machinery. Patients who have been given fentanyl transdermal system should not drive or operate dangerous machinery unless they are tolerant to the effects of the drug.
Information for Patients
Patients and their caregivers should be provided with a Medication Guide each time fentanyl transdermal system is dispensed because new information may be available.
Patients receiving fentanyl transdermal systems should be given the following instructions by the physician:
- Patients should be advised that fentanyl transdermal systems contain fentanyl, an opioid pain medicine similar to morphine, hydromorphone, methadone, oxycodone, and oxymorphone.
- Patients should be advised that each fentanyl transdermal system may be worn continuously for 72 hours, and that each patch should be applied to a different skin site after removal of the previous transdermal patch.
- Patients should be advised that fentanyl transdermal systems should be applied to intact, non-irritated, and non-irradiated skin on a flat surface such as the chest, back, flank, or upper arm. Additionally, patients should be advised of the following:
- In young children or persons with cognitive impairment, the patch should be put on the upper back to lower the chances that the patch will be removed and placed in the mouth.
- Hair at the application site should be clipped (not shaved) prior to patch application.
- If the site of fentanyl transdermal system application must be cleansed prior to application of the patch, do so with clear water.
- Do not use soaps, oils, lotions, alcohol, or any other agents that might irritate the skin or alter its characteristics.
- Allow the skin to dry completely prior to patch application.
4. Patients should be advised that fentanyl transdermal system should be applied immediately upon removal from the sealed blister package and after removal of the protective liner. Additionally the patient should be advised of the following:
- The fentanyl transdermal system should not be used if the seal is broken, or if the patch is cut, damaged, or changed in any way.
- The transdermal patch should be pressed firmly in place with the palm of the hand for 30 seconds, making sure the contact is complete, especially around the edges.
- The patch should not be folded so that only part of the patch is exposed.
5. Patients should be advised that the dose of fentanyl transdermal system or the number of patches applied to the skin should NEVER be adjusted without the prescribing healthcare professional’s instruction.
6. Patients should be advised that while wearing the patch, they should avoid exposing the fentanyl transdermal system application site and surrounding area to direct external heat sources, such as:
- heating pads,
- electric blankets,
- sunbathing,
- heat or tanning lamps,
- saunas,
- hot tubs or hot baths, and
- heated water beds, etc.
7. Patients should also be advised of a potential for temperature dependent increases in fentanyl release from the patch that could result in an overdose of fentanyl; therefore, patients who develop a high fever or increased body temperature due to strenuous exertion while wearing the patch should contact their physician.
8. Patients should be advised that if they experience problems with adhesion of the fentanyl transdermal system, they may tape the edges of the patch with first aid tape. If problems with adhesion persist, patients may overlay the patch with a transparent adhesive film dressing (e.g., Bioclusive™).
9. Patients should be advised that if the patch falls off before 72 hours a new patch may be applied to a different skin site.
10. Patients should be advised to fold (so that the adhesive side adheres to itself) and immediately flush down the toilet used fentanyl transdermal systems after removal from the skin.
11. Patients should be instructed that, if the drug matrix accidentally contacts the skin, the area should be washed clean with clear water and not soap, alcohol, or other chemicals, because these products may increase the ability of fentanyl to go through the skin.
12. Patients should be advised that fentanyl transdermal system may impair mental and/or physical ability required for the performance of potentially hazardous tasks (e.g., driving, operating machinery).
13. Patients should be advised to refrain from any potentially dangerous activity when starting on fentanyl transdermal system or when their dose is being adjusted, until it is established that they have not been adversely affected.
14. Patients should be advised that fentanyl transdermal system should not be combined with alcohol or other CNS depressants (e.g., sleep medications, tranquilizers) because dangerous additive effects may occur, resulting in serious injury or death.
15. Patients should be advised to consult their physician or pharmacist if other medications are being or will be used with fentanyl transdermal system.
16. Patients should be advised of the potential for severe constipation.
17. Patients should be advised that if they have been receiving treatment with fentanyl transdermal system and cessation of therapy is indicated, it may be appropriate to taper fentanyl transdermal system dose, rather than abruptly discontinue it, due to the risk of precipitating withdrawal symptoms.
18. Patients should be advised that fentanyl transdermal system contains fentanyl, a drug with high potential for abuse.
19. Patients, family members and caregivers should be advised to protect fentanyl transdermal system from theft or misuse in the work or home environment.
20. Patients should be instructed to keep fentanyl transdermal system in a secure place out of the reach of children due to the high risk of fatal respiratory depression.
21. Patients should be advised that fentanyl transdermal system should never be given to anyone other than the individual for whom it was prescribed because of the risk of death or other serious medical problems to that person for whom it was not intended.
22. Patients should be informed that, if the patch dislodges and accidentally sticks to the skin of another person, they should immediately take the patch off, wash the exposed area with water and seek medical attention for the accidentally exposed individual.
23. When fentanyl transdermal system is no longer needed, the unused patches should be removed from their blisters, folded so that the adhesive side of the patch adheres to itself, and flushed down the toilet.
24. Women of childbearing potential who become, or are planning to become pregnant, should be advised to consult a physician prior to initiating or continuing therapy with fentanyl transdermal system.
25. Patients should be informed that accidental exposure or misuse may lead to death or other serious medical problems.
26. MRI: Skin burns have been reported at the patch site in several patients wearing an aluminized transdermal system during a magnetic resonance imaging scan (MRI). Because this fentanyl transdermal system contains aluminum, it is recommended to remove the patch before undergoing any MRI procedures.
Agents Affecting Cytochrome P450 3A4 Isoenzyme System
Fentanyl is metabolized mainly via the human cytochrome P450 3A4 isoenzyme system (CYP3A4), therefore potential interactions may occur when fentanyl transdermal system is given concurrently with agents that affect CYP3A4 activity. Coadministration with agents that induce CYP3A4 activity may reduce the efficacy of fentanyl transdermal system. The concomitant use of transdermal fentanyl with all CYP3A4 inhibitors (such as ritonavir, ketoconazole, itraconazole, troleandomycin, clarithromycin, nelfinavir, nefazadone, amiodarone, amprenavir, aprepitant, diltiazem, erythromycin, fluconazole, fasamprenavir, grapefruit juice, and verapamil) may result in an increase in fentanyl plasma concentrations, which could increase or prolong adverse drug effects and may cause fatal respiratory depression. Patients receiving fentanyl transdermal system and any CYP3A4 inhibitor should be carefully monitored for an extended period of time, and dosage adjustments should be made if warranted (see BOX WARNING; CLINICAL PHARMACOLOGY, Drug Interactions; WARNINGS; and DOSAGE AND ADMINISTRATION for further information).
Central Nervous System Depressants
The concomitant use of fentanyl transdermal system with other central nervous system depressants, including but not limited to other opioids, sedatives, hypnotics, tranquilizers (e.g., benzodiazepines), general anesthetics, phenothiazines, skeletal muscle relaxants, and alcohol, may cause respiratory depression, hypotension, and profound sedation, or potentially result in coma or death. When such combined therapy is contemplated, the dose of one or both agents should be significantly reduced.
MAO Inhibitors
Fentanyl transdermal system is not recommended for use in patients who have received MAOI within 14 days because severe and unpredictable potentiation by MAO inhibitors has been reported with opioid analgesics.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies in animals to evaluate the carcinogenic potential of fentanyl HCl have not been conducted. There was no evidence of mutagenicity in the Ames Salmonella mutagenicity assay, the primary rat hepatocyte unscheduled DNA synthesis assay, the BALB/c 3T3 transformation test, and the human lymphocyte and CHO chromosomal aberration in-vitro assays.
The potential effects of fentanyl on male and female fertility were examined in the rat model via two separate experiments. In the male fertility study, male rats were treated with fentanyl (0, 0.025, 0.1, or 0.4 mg/kg/day) via continuous intravenous infusion for 28 days prior to mating; female rats were not treated. In the female fertility study, female rats were treated with fentanyl (0, 0.025, 0.1, or 0.4 mg/kg/day) via continuous intravenous infusion for 14 days prior to mating until day 16 of pregnancy; male rats were not treated. Analysis of fertility parameters in both studies indicated that an intravenous dose of fentanyl up to 0.4 mg/kg/day to either the male or the female alone produced no effects on fertility (this dose is approximately 1.6 times the daily human dose administered by a 100 mcg/hr patch on a mg/m2 basis). In a separate study, a single daily bolus dose of fentanyl was shown to impair fertility in rats when given in intravenous doses of 0.3 times the human dose for a period of 12 days.
Pregnancy category C
No epidemiological studies of congenital anomalies in infants born to women treated with fentanyl during pregnancy have been reported.
The potential effects of fentanyl on embryo-fetal development were studied in the rat, mouse, and rabbit models. Published literature reports that administration of fentanyl (0, 10, 100, or 500 mcg/kg/day) to pregnant female Sprague-Dawley rats from day 7 to 21 via implanted microosmotic minipumps did not produce any evidence of teratogenicity (the high dose is approximately 2 times the daily human dose administered by a 100 mcg/hr patch on a mg/m2 basis). In contrast, the intravenous administration of fentanyl (0, 0.01, or 0.03 mg/kg) to bred female rats from gestation day 6 to 18 suggested evidence of embryotoxicity and a slight increase in mean delivery time in the 0.03 mg/kg/day group. There was no clear evidence of teratogenicity noted.
Pregnant female New Zealand White rabbits were treated with fentanyl (0, 0.025, 0.1, 0.4 mg/kg) via intravenous infusion from day 6 to day 18 of pregnancy. Fentanyl produced a slight decrease in the body weight of the live fetuses at the high dose, which may be attributed to maternal toxicity. Under the conditions of the assay, there was no evidence for fentanyl induced adverse effects on embryo-fetal development at doses up to 0.4 mg/kg (approximately 3 times the daily human dose administered by a 100 mcg/hr patch on a mg/m2 basis).
There are no adequate and well-controlled studies in pregnant women. Fentanyl transdermal system should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nonteratogenic Effects
Chronic maternal treatment with fentanyl during pregnancy has been associated with transient respiratory depression, behavioral changes, or seizures characteristic of neonatal abstinence syndrome in newborn infants. Symptoms of neonatal respiratory or neurological depression were no more frequent than expected in most studies of infants born to women treated acutely during labor with intravenous or epidural fentanyl. Transient neonatal muscular rigidity has been observed in infants whose mothers were treated with intravenous fentanyl.
The potential effects of fentanyl on prenatal and postnatal development were examined in the rat model. Female Wistar rats were treated with 0, 0.025, 0.1, or 0.4 mg/kg/day fentanyl via intravenous infusion from day 6 of pregnancy through 3 weeks of lactation. Fentanyl treatment (0.4 mg/kg/day) significantly decreased body weight in male and female pups and also decreased survival in pups at day 4. Both the mid-dose and high-dose fentanyl animals demonstrated alterations in some physical landmarks of development (delayed incisor eruption and eye opening) and transient behavioral development (decreased locomotor activity at day 28 which recovered by day 50). The mid-dose and the high-dose are 0.4 and 1.6 times the daily human dose administered by a 100 mcg/hr patch on a mg/m2 basis.
Labor and Delivery
Fentanyl readily passes across the placenta to the fetus; therefore, fentanyl transdermal system is not recommended for analgesia during labor and delivery.
Nursing Mothers
Fentanyl is excreted in human milk; therefore, fentanyl transdermal system is not recommended for use in nursing women because of the possibility of effects in their infants.
Pediatric Use
The safety of fentanyl transdermal system was evaluated in three open-label trials in 291 pediatric patients with chronic pain, 2 years of age through 18 years of age. Starting doses of 25 mcg/hr and higher were used by 181 patients who had been on prior daily opioid doses of at least 45 mg/day of oral morphine or an equianalgesic dose of another opioid. Initiation of fentanyl transdermal system therapy in pediatric patients taking less than 60 mg/day of oral morphine or an equianalgesic dose of another opioid has not been evaluated in controlled clinical trials. Approximately 90% of the total daily opioid requirement (fentanyl transdermal system plus rescue medication) was provided by fentanyl transdermal system.
Fentanyl transdermal system was not studied in children under 2 years of age.
Fentanyl transdermal system should be administered to children only if they are opioid-tolerant and 2 years of age or older (see DOSAGE AND ADMINISTRATION and BOX WARNING).
To guard against accidental ingestion by children, use caution when choosing the application site for fentanyl transdermal system (see DOSAGE AND ADMINISTRATION) and monitor adhesion of the system closely.
Geriatric Use
Information from a pilot study of the pharmacokinetics of IV fentanyl in geriatric patients (N = 4) indicates that the clearance of fentanyl may be greatly decreased in the population above the age of 60. The relevance of these findings to transdermal fentanyl system is unknown at this time.
Respiratory depression is the chief hazard in elderly or debilitated patients, usually following large initial doses in non-tolerant patients or when opioids are given in conjunction with other agents that depress respiration.
Fentanyl transdermal system should be used with caution in elderly, cachectic, or debilitated patients as they may have altered pharmacokinetics due to poor fat stores, muscle wasting or altered clearance (see DOSAGE AND ADMINISTRATION).
-
ADVERSE REACTIONS
In postmarketing experience, deaths from hypoventilation due to inappropriate use of fentanyl transdermal system have been reported (see BOX WARNING and CONTRAINDICATIONS).
Premarketing Clinical Trial Experience
Although fentanyl transdermal system use in post-operative or acute pain and in patients who are not opioid-tolerant is CONTRAINDICATED, the safety of fentanyl transdermal system was originally evaluated in 357 post-operative adult patients for 1 to 3 days and 153 cancer patients for a total of 510 patients. The duration of fentanyl transdermal system use varied in cancer patients; 56% of patients used fentanyl transdermal system for over 30 days, 28% continued treatment for more than 4 months, and 10% used fentanyl transdermal system for more than 1 year.
Hypoventilation was the most serious adverse reaction observed in 13 (4%) post-operative patients and in 3 (2%) of the cancer patients. Hypotension and hypertension were observed in 11 (3%) and 4 (1%) of the opioid-naive patients.
Various adverse events were reported; a causal relationship to fentanyl transdermal system was not always determined. The frequencies presented here reflect the actual frequency of each adverse effect in patients who received fentanyl transdermal system. There has been no attempt to correct for a placebo effect, concomitant use of other opioids, or to subtract the frequencies reported by placebo-treated patients in controlled trials.
Adverse reactions reported in 153 cancer patients at a frequency of 1% or greater are presented in TABLE 1; similar reactions were seen in the 357 post-operative patients.
In the pediatric population, the safety of fentanyl transdermal system has been evaluated in 291 patients with chronic pain 2 to 18 years of age. The duration of fentanyl transdermal system use varied; 20% of pediatric patients were treated for ≤ 15 days; 46% for 16 to 30 days; 16% for 31 to 60 days; and 17% for at least 61 days. Twenty-five patients were treated with fentanyl transdermal system for at least 4 months and 9 patients for more than 9 months.
There was no apparent pediatric-specific risk associated with fentanyl transdermal system use in children as young as 2 years old when used as directed. The most common adverse events were fever (35%), vomiting (33%), and nausea (24%).
Adverse events reported in pediatric patients at a rate of ≥ 1% are presented in TABLE 1.
TABLE 1: ADVERSE EVENTS (at rate of ≥ 1%) Adult (N = 380) and Pediatric (N = 291) Clinical Trial Experience Body System Adults Pediatrics - * Reactions occurring in 3% to 10% of fentanyl transdermal system patients
- † Reactions occurring in 10% or more of fentanyl transdermal system patients
Body as a Whole Abdominal pain*, headache*, fatigue*, back pain, fever, influenza-like symptoms*, accidental injury, rigors Pain*, headache*, fever, syncope, abdominal pain, allergic reaction, flushing Cardiovascular Arrhythmia, chest pain Hypertension, tachycardia Digestive Nausea†, vomiting†, constipation†, dry mouth†, anorexia*, diarrhea*, dyspepsia*, flatulence Nausea†, vomiting†, constipation*, dry mouth, diarrhea Nervous Somnolence†, insomnia, confusion†, asthenia†, dizziness*, nervousness*, hallucinations*, anxiety*, depression*, euphoria*, tremor,abnormal coordination, speech disorder, abnormal thinking, abnormal gait, abnormal dreams, agitation, paresthesia, amnesia, syncope, paranoid reaction Somnolence*, nervousness*, insomnia*, asthenia*, hallucinations, anxiety, depression, convulsions, dizziness, tremor, speech disorder, agitation, stupor, confusion, paranoid reaction Respiratory Dyspnea*, hypoventilation*, apnea*, hemoptysis, pharyngitis*, hiccups, bronchitis, rhinitis, sinusitis, upper respiratory tract infection* Dyspnea, respiratory depression, rhinitis, coughing Skin and Appendages Sweating†, pruritus*, rash, application site reaction – erythema, papules, itching, edema Pruritus*, application site reaction*, sweating increased, rash, rash erythematous, skin reaction localized Urogenital Urinary retention* Micturition disorder Urinary retention The following adverse effects have been reported in less than 1% of the 510 adult post-operative and cancer patients studied:
Cardiovascular: bradycardia
Digestive: abdominal distention
Nervous: aphasia, hypertonia, vertigo, stupor, hypotonia, depersonalization, hostility
Respiratory: stertorous breathing, asthma, respiratory disorder
Skin and Appendages, General: exfoliative dermatitis, pustules
Special Senses: amblyopia
Urogenital: bladder pain, oliguria, urinary frequency
Postmarketing Experience – Adults
The following adverse reactions have been reported in association with the use of fentanyl transdermal system and not reported in the premarketing adverse reactions section above:
Body as a Whole: edema
Cardiovascular: tachycardia
Metabolic and Nutritional: weight loss
Special Senses: blurred vision
Urogenital: decreased libido, anorgasmia, ejaculatory difficulty
-
DRUG ABUSE AND ADDICTION
Fentanyl transdermal system contains a high concentration of fentanyl, a potent Schedule II opioid agonist. Schedule II opioid substances, which include hydromorphone, methadone, morphine, oxycodone, and oxymorphone, have the highest potential for abuse and risk of fatal overdose due to respiratory depression. Fentanyl, like morphine and other opioids used in analgesia, can be abused and is subject to criminal diversion.
The high content of fentanyl in the patches (fentanyl transdermal system) may be a particular target for abuse and diversion.
Addiction is a primary, chronic, neurobiologic disease, with genetic, psychosocial, and environmental factors influencing its development and manifestations. It is characterized by behaviors that include one or more of the following: impaired control over drug use, compulsive use, continued use despite harm, and craving. Drug addiction is a treatable disease, utilizing a multidisciplinary approach, but relapse is common.
“Drug seeking” behavior is very common in addicts and drug abusers. Drug-seeking tactics include emergency calls or visits near the end of office hours, refusal to undergo appropriate examination, testing or referral, repeated “loss” of prescriptions, tampering with prescriptions and reluctance to provide prior medical records or contact information for other treating physician(s). “Doctor shopping” to obtain additional prescriptions is common among drug abusers and people suffering from untreated addiction.
Abuse and addiction are separate and distinct from physical dependence and tolerance. Physicians should be aware that addiction may be accompanied by concurrent tolerance and symptoms of physical dependence. In addition, abuse of opioids can occur in the absence of true addiction and is characterized by misuse for non-medical purposes, often in combination with other psychoactive substances. Since fentanyl transdermal system may be diverted for non-medical use, careful record keeping of prescribing information, including quantity, frequency, and renewal requests is strongly advised.
Proper assessment of the patient, proper prescribing practices, periodic re-evaluation of therapy, and proper dispensing and storage are appropriate measures that help to limit abuse of opioid drugs.
Fentanyl transdermal systems are intended for transdermal use (to be applied on the skin) only. Do not use a fentanyl transdermal system if the seal is broken or the patch is cut, damaged, or changed in any way.
-
OVERDOSAGE
Clinical Presentation
The manifestations of fentanyl overdosage are an extension of its pharmacologic actions with the most serious significant effect being hypoventilation.
Treatment
For the management of hypoventilation, immediate countermeasures include removing the fentanyl transdermal system and physically or verbally stimulating the patient. These actions can be followed by administration of a specific narcotic antagonist such as naloxone. The duration of hypoventilation following an overdose may be longer than the effects of the narcotic antagonist’s action (the half-life of naloxone ranges from 30 to 81 minutes). The interval between IV antagonist doses should be carefully chosen because of the possibility of re-narcotization after system removal; repeated administration of naloxone may be necessary. Reversal of the narcotic effect may result in acute onset of pain and the release of catecholamines.
Always ensure a patent airway is established and maintained, administer oxygen and assist or control respiration as indicated and use an oropharyngeal airway or endotracheal tube if necessary. Adequate body temperature and fluid intake should be maintained.
If severe or persistent hypotension occurs, the possibility of hypovolemia should be considered and managed with appropriate parenteral fluid therapy.
-
DOSAGE AND ADMINISTRATION
Special Precautions
Fentanyl transdermal system contains a high concentration of a potent Schedule II opioid agonist, fentanyl. Schedule II opioid substances which include fentanyl, hydromorphone, methadone, morphine, oxycodone, and oxymorphone have the highest potential for abuse and associated risk of fatal overdose due to respiratory depression. Fentanyl can be abused and is subject to criminal diversion. The high content of fentanyl in fentanyl transdermal system may be a particular target for abuse and diversion.
Fentanyl transdermal systems are intended for transdermal use (on intact skin) only. The fentanyl transdermal system should not be used if the seal is broken, or the patch is cut, damaged, or changed in any way.
Each fentanyl transdermal system may be worn continuously for 72 hours. The next patch should be applied to a different skin site after removal of the previous transdermal system.
If problems with adhesion of the fentanyl transdermal system occur, the edges of the patch may be taped with first aid tape. If problems with adhesion persist, the patch may be overlayed with a transparent adhesive film dressing (e.g., Bioclusive™).
If the patch falls off before 72 hours, dispose of it by folding in half and flushing down the toilet. A new patch may be applied to a different skin site.
Fentanyl transdermal system is ONLY for use in patients who are already tolerant to opioid therapy of comparable potency. Use in non-opioid tolerant patients may lead to fatal respiratory depression. Overestimating the fentanyl transdermal system dose when converting patients from another opioid medication can result in fatal overdose with the first dose. Due to the mean elimination half-life of 17 hours of fentanyl transdermal system, patients who are thought to have had a serious adverse event, including overdose, will require monitoring and treatment for at least 24 hours.
The concomitant use of fentanyl transdermal system with all cytochrome P450 3A4 inhibitors (such as ritonavir, ketoconazole, itraconazole, troleandomycin, clarithromycin, nelfinavir, nefazodone, amiodarone, amprenavir, aprepitant, diltiazem, erythromycin, fluconazole, fasamprenavir, grapefruit juice, and verapamil) may result in an increase in fentanyl plasma concentrations, which could increase or prolong adverse drug effects and may cause potentially fatal respiratory depression. Patients receiving fentanyl transdermal system and any CYP3A4 inhibitor should be carefully monitored for an extended period of time and dosage adjustments should be made if warranted (see BOX WARNING; WARNINGS; CLINICAL PHARMACOLOGY, Drug Interactions; WARNINGS; and PRECAUTIONS for further information).
Pediatric patients converting to fentanyl transdermal system with a 25 mcg/hr patch should be opiod-tolerant and receiving at least 60 mg of oral morphine or the equivalent per day. The dose conversion schedule described in Table C, and method of titration described below are recommended in opioid-tolerant pediatric patients over 2 years of age with chronic pain (see PRCAUTIONS, Pediatric Use).
Respiratory depression is the chief hazard in elderly or debilitated patients, usually following large initial doses in non-tolerant patients, or when opioids are given in conjunction with other agents that depress respiration.
Fentanyl transdermal system should be used with caution in elderly, cachectic, or debilitated patients as they may have altered pharmacokinetics due to poor fat stores, muscle wasting, or altered clearance (see CLINICAL PHARMACOLOGY, Special Populations, Geriatric Use).
General Principles
Fentanyl transdermal system is indicated for management of persistent, moderate to severe chronic pain that:
- requires continuous, around-the-clock opioid administration for an extended period of time
- cannot be managed by other means such as non-steroidal analgesics, opioid combination products, or immediate-release opioids.
Fentanyl transdermal system should ONLY be used in patients who are already receiving opioid therapy, who have demonstrated opioid tolerance, and who require a total daily dose at least equivalent to fentanyl transdermal system 25 mcg/hr. Patients who are considered opioid-tolerant are those who have been taking, for a week or longer, at least 60 mg of morphone daily, or at least 30 mg of oral oxycodone daily, or at least 8 mg oral hydromorphone daily, or an equianalgesic dose of another opioid.
Because serious or life-threatening hypoventilation could occur, fentanyl transdermal system is contraindicated:
- in patients who are not opioid-tolerant
- in the management of acute pain or in patients who require opioid analgesia for a short period of time.
- in the management of post-operative pain, including use after out-patient or day surgeries (e.g., tonsillectomies)
- in the management of mild pain
- in the management of intermittent pain (e.g., use on an as needed basis [prn])
(See CONTRAINDICATIONS for further information.)
Safety of fentanyl transdermal system has not been established in children under 2 years of age. Fentanyl transdermal system should be administered to children only if they are opioid-tolerant and 2 years of age or older (see PRECAUTIONS, Pediatric Use).
Prescribers should individualize treatment using a progressive plan of pain management such as outlined by the World Health Organization, the Agency for Health Research and Quality, the Federation of State Medical Boards Model Policy, or the American Pain Society.
With all opioids, the safety of patients using the products is dependent on health care practitioners prescribing them in strict conformity with their approved labeling with respect to patient selection, dosing, and proper conditions for use.
As with all opioids, dosage should be individualized. The most important factor to be considered in determining the appropriate dose is the extent of preexisting opioid-tolerance (see BOXWARNING and CONTRAINDICATIONS). Initial doses should be reduced in elderly or debilitated patients (see PRECAUTIONS).
Fentanyl transdermal system should be applied to intact, non-irritated, and non-irradiated skin on a flat surface such as the chest, back, flank, or upper arm. In young children and persons with cognitive impairment, adhesion should be monitored and the upper back is the preferred location to minimize the potential of inappropriate patch removal. Hair at the application site should be clipped (not shaved) prior to system application. If the site of fentanyl transdermal system application must be cleansed prior to application of the patch, do so with clear water. Do not use soaps, oils, lotions, alcohol, or any other agents that might irritate the skin or alter its characteristics. Allow the skin to dry completely prior to patch application.
Fentanyl transdermal system should be applied immediately upon removal from the sealed blister package. Do not use if the seal is broken. Do not alter the patch (e.g., cut) in any way prior to application and do not use cut or damaged patches.
The transdermal system should be pressed firmly in place with the palm of the hand for 30 seconds, making sure the contact is complete, especially around the edges. If the drug matrix accidentally contacts the skin of the patient or caregiver, the skin should be washed with copious amounts of water. Do not use soap, alcohol, or other solvents because they may enhance the drug’s ability to penetrate the skin.
Fentanyl transdermal system should be kept out of the reach of children. Used patches should be folded so that the adhesive side of the patch adheres to itself, then the patch should be flushed down the toilet immediately upon removal. Patients should dispose of any patches remaining from a prescription as soon as they are no longer needed. Unused patches should be removed from their blisters, folded so that the adhesive side of the patch adheres to itself, and flushed down the toilet.
Dose Selection
Doses must be individualized based upon the status of each patient and should be assessed at regular intervals after fentanyl transdermal system application. Reduced doses of fentanyl transdermal system are suggested for the elderly and other groups discussed in PRECAUTIONS.
Fentanyl transdermal system is ONLY for use in patients who are already tolerant to opioid therapy of comparable potency. Use in non-opioid tolerant patients may lead to fatal respiratory depression.
In selecting an initial fentanyl transdermal system dose, attention should be given to 1) the daily dose, potency, and characteristics of the opioid the patient has been taking previously (e.g., whether it is a pure agonist or mixed agonist-antagonist), 2) the reliability of the relative potency estimates used to calculate the fentanyl transdermal system dose needed (potency estimates may vary with the route of administration), 3) the degree of opioid tolerance, and 4) the general condition and medical status of the patient. Each patient should be maintained at the lowest dose providing acceptable pain control.
Initial Fentanyl Transdermal System Dose Selection
Overestimating the fentanyl transdermal system dose when converting patients from another opioid medication can result in fatal overdose with the first dose. Due to the mean elimination half-life of 17 hours of fentanyl transdermal system, patients who are thought to have had a serious adverse event, including overdose, will require monitoring and treatment for at least 24 hours.
There has been no systemic evaluation of fentanyl transdermal system as an initial opioid analgesic in the management of chronic pain, since most patients in the clinical trials were converted to fentanyl transdermal system from other narcotics. The efficacy of fentanyl transdermal system 12 mcg/hr as an initiating dose has not been determined. In addition, patients who are not opioid-tolerant have experienced hypoventilation and death during use of fentanyl transdermal system. Therefore, fentanyl transdermal system should be used only in patients who are opioid-tolerant.
To convert adult and pediatric patients from oral or parenteral opioids to fentanyl transdermal system use TABLE C:
Alternatively, for adult and pediatric patients taking opioids or doses not listed in TABLE C, use the following methodology:
- Calculate the previous 24 hour analgesic requirement.
- Convert this amount to the equianalgesic oral morphine dose using TABLE D.
- TABLE E displays the range of 24 hour oral morphine doses that are recommended for conversion to each fentanyl transdermal system dose. Use this table to find the calculated 24 hour morphine dose and the corresponding fentanyl transdermal system dose. Initiate fentanyl transdermal system treatment using the recommended dose and titrate patients upwards (no more frequently than every 3 days after the initial dose or than every 6 days thereafter) until analgesic efficacy is attained. The recommended starting dose when converting from other opioids to fentanyl transdermal system is likely too low for 50% of patients. This starting dose is recommended to minimize the potential for overdosing patients with the first dose. For delivery rates in excess of 100 mcg/hr, multiple systems may be used.
TABLE C1 DOSE CONVERSION GUIDELINES Current Analgesic Daily Dosage (mg/d) Oral morphine 60 to 134 135 to 224 225 to 314 315 to 404 IM/IV morphine 10 to 22 23 to 37 38 to 52 53 to 67 Oral oxycodone 30 to 67 67.5 to 112 112.5 to 157 157.5 to 202 IM/IV oxycodone 15 to 33 33.1 to 56 56.1 to 78 78.1 to 101 Oral codeine 150 to 447 448 to 747 748 to 1047 1048 to 1347 Oral hydromorphone 8 to 17 17.1 to 28 28.1 to 39 39.1 to 51 IV hydromorphone 1.5 to 3.4 3.5 to 5.6 5.7 to 7.9 8 to 10 IM meperidine 75 to 165 166 to 278 279 to 390 391 to 503 Oral methadone 20 to 44 45 to 74 75 to 104 105 to 134 IM methadone 10 to 22 23 to 37 38 to 52 53 to 67 ↓ ↓ ↓ ↓ Recommended fentanyl transdermal system dose 25 mcg/hr 50 mcg/hr 75 mcg/hr 100 mcg/hr Alternatively, for adult and pediatric patients taking opioids or doses not listed in TABLE C, use the conversion methodology outlined above with TABLE D.
1 TABLE C should not be used to convert from fentanyl transdermal system to other therapies because this conversion to fentanyl transdermal system is conservative. Use of TABLE C for conversion to other analgesic therapies can overestimate the dose of the new agent. Overdosage of the new analgesic agent is possible (see DOSAGE AND ADMINISTRATION, Discontinuation of Fentanyl Transdermal System).
TABLE D*† EQUIANALGESIC POTENCY CONVERSION - * TABLE D should not be used to convert from fentanyl transdermal system to other therapies because this conversion to fentanyl transdermal system is conservative. Use of TABLE D for conversion to other analgesic therapies can overestimate the dose of the new agent. Overdosage of the new analgesic agent is possible (see DOSAGE AND ADMINISTRATION, Discontinuation of Fentanyl Transdermal System).
- † All IM and PO doses in this chart are considered equivalent to 10 mg of IM morphine in analgesic effect. IM denotes intramuscular, PO oral, and PR rectal.
- ‡ Based on single-dose studies in which an intramuscular dose of each drug listed was compared with morphine to establish the relative potency. Oral doses are those recommended when changing from parenteral to an oral route. Reference: Foley, K.M. (1985) The treatment of cancer pain. NEJM 313(2):84-95.
- § Although controlled studies are not available, in clinical practice it is customary to consider the doses of opioid given IM, IV, or subcutaneously to be equivalent. There may be some differences in pharmacokinetic parameters such as Cmax and Tmax.
- ¶ The conversion ratio of 10 mg parenteral morphine = 30 mg oral morphine is based on clinical experience in patients with chronic pain. The conversion ratio of 10 mg parenteral morphine = 60 mg oral morphine is based on a potency study in acute pain. Reference: Ashburn and Lipman (1993) Management of pain in the cancer patient. Anesth Analg 76:402-416.
Name Equianalgesic Dose (mg) IM‡,§ PO Morphine 10 60 (30)¶ Hydromorphone (Dilaudid®) 1.5 7.5 Methadone (Dolophine®) 10 20 Oxycodone 15 30 Levorphanol (Levo-Dromoran®) 2 4 Oxymorphone (Numorphan®) 1 10 (PR) Meperidine (Demerol®) 75 - Codeine 130 200 TABLE E* RECOMMENDED INITIAL FENTANYL TRANSDERMAL SYSTEM DOSE BASED UPON DAILY ORAL MORPHINE DOSE - * TABLE E should not be used to convert from fentanyl transdermal system to other therapies because this conversion to fentanyl transdermal system is conservative. Use of TABLE E for conversion to other analgesic therapies can overestimate the dose of the new agent. Overdosage of the new analgesic agent is possible (see DOSAGE AND ADMINISTRATION, Discontinuation of Fentanyl Transdermal System).
Oral 24 hour Morphine (mg/day) Fentanyl Transdermal System Dose (mcg/hr) 60 to 134 25 135 to 224 50 225 to 314 75 315 to 404 100 405 to 494 125 495 to 584 150 585 to 674 175 675 to 764 200 765 to 854 225 855 to 944 250 945 to 1034 275 1035 to 1124 300 NOTE: In clinical trials, these ranges of daily oral morphine doses were used as a basis for conversion to fentanyl transdermal system.
The majority of patients are adequately maintained with fentanyl transdermal system administered every 72 hours. Some patients may not achieve adequate analgesia using this dosing interval and may require systems to be applied every 48 hours rather than every 72 hours. An increase in the fentanyl transdermal system dose should be evaluated before changing dosing intervals in order to maintain patients on a 72 hour regimen. Dosing intervals less than every 72 hours were not studied in children and adolescents and are not recommended.
Because of the increase in serum fentanyl concentration over the first 24 hours following initial system application, the initial evaluation of the maximum analgesic effect of fentanyl transdermal system cannot be made before 24 hours of wearing. The initial fentanyl transdermal system dose may be increased after 3 days (see DOSAGE AND ADMINISTRATION, Dose Titration).
During the initial application of fentanyl transdermal system, patients should use short-acting analgesics as needed until analgesic efficacy with fentanyl transdermal system is attained. Thereafter, some patients still may require periodic supplemental doses of other short-acting analgesics for “breakthrough” pain.
Dose Titration
The recommended initial fentanyl transdermal system dose based upon the daily oral morphine dose is conservative, and 50% of patients are likely to require a dose increase after initial application of fentanyl transdermal system. The initial fentanyl transdermal system dosage may be increased after 3 days based on the daily dose of supplemental opioid analgesics required by the patient in the second or third day of the initial application.
Physicians are advised that it may take up to 6 days after increasing the dose of fentanyl transdermal system for the patient to reach equilibrium on the new dose (see graph in CLINICAL PHARMACOLOGY). Therefore, patients should wear a higher dose through two applications before any further increase in dosage is made on the basis of the average daily use of a supplemental analgesic.
Appropriate dosage increments should be based on the daily dose of supplementary opioids, using the ratio of 45 mg/24 hours of oral morphine to a 12.5 mcg/hr increase in fentanyl transdermal system dose.
Discontinuation of Fentanyl Transdermal System
To convert patients to another opioid, remove fentanyl transdermal system and titrate the dose of the new analgesic based upon the patient’s report of pain until adequate analgesia has been attained. Upon system removal, 17 hours or more are required for a 50% decrease in serum fentanyl concentrations. Opioid withdrawal symptoms (such as nausea, vomiting, diarrhea, anxiety, and shivering) are possible in some patients after conversion or dose adjustment. For patients requiring discontinuation of opioids, a gradual downward titration is recommended since it is not known at what dose level the opioid may be discontinued without producing the signs and symptoms of abrupt withdrawal.
TABLES C, D, and E should not be used to convert from fentanyl transdermal system to other therapies. Because the conversion of fentanyl transdermal system is conservative, use of TABLES C, D, and E for conversion to other analgesic therapies can overestimate the dose of the new agent. Overdosage of the new analgesic agent is possible.
-
HOW SUPPLIED
Fentanyl transdermal system is supplied in cartons containing 5 individually packaged systems. See chart for information regarding individual systems:
Label Strength (mcg/hr) Patch Size (cm2) Fentanyl Content (mg) 25 10.7 2.76 50 21.4 5.52 75 32.1 8.28 100 42.8 11.04 They are supplied by Dispensing Solutions Inc. as follows:
NDC Strength Quantity/Form Source NDC 68258-3023-1 25 ug/1 h 5 BLISTER PACK 0093-6900-45 68258-3024-1 75 ug/1 h 5 BLISTER PACK 0093-6902-45 68258-3025-1 100 ug/1 h 5 BLISTER PACK 0093-6903-45 Safety and Handling
Fentanyl transdermal systems are supplied in sealed blister packages which pose little risk of exposure to health care workers. If the drug matrix accidentally contacts the skin, the area should be washed with copious amounts of water. Do not use soap, alcohol, or other solvents because they may enhance the drug’s ability to penetrate the skin. Do not use a fentanyl transdermal system if the seal is broken or the patch is cut, damaged, or changed in any way.
KEEP FENTANYL TRANSDERMAL SYSTEM OUT OF THE REACH OF CHILDREN AND PETS.
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature]. Apply immediately after removal from individually sealed blister package. Do not use if the seal is broken. For transdermal use only.
A SCHEDULE CII NARCOTIC. DEA ORDER FORM REQUIRED.
Bioclusive™ is a trademark of Ethicon, Inc.
Dilaudid® is a registered trademark of Abbott Laboratories.
Dolophine® is a registered trademark of Roxane Laboratories, Inc.
Levo-Dromoran® is a registered trademark of Valeant Pharmaceuticals International.
Numorphan® is a registered trademark of Endo Pharmaceuticals.
Demerol® is a registered trademark of Sanofi-Aventis U.S.
Manufactured By:
Aveva Drug Delivery Systems
A Nitto Denko Company
Miramar, FL 33025
Distributed By:
TEVA PHARMACEUTICALS USA
Sellersville, PA 18960
And Relabeled By:
Dispensing Solutions Inc.
3000 West Warner Ave
Santa Ana, CA 92704
United States
-
MEDICATION GUIDE
Fentanyl Transdermal System CII
Rx only
IMPORTANT:
- Keep fentanyl transdermal system in a safe place away from children and pets. Accidental use by a child or pet is a medical emergency and may result in death. If a child or pet accidentally uses fentanyl transdermal system, get emergency help right away.
- Make sure you read the separate “Instructions for Applying a Fentanyl Transdermal System.” Always use a fentanyl transdermal system the right way. Fentanyl transdermal system can cause serious breathing problems and death, especially if it is used the wrong way.
- Fentanyl transdermal system is a federally controlled substance (C-II) because it can be abused. Keep fentanyl transdermal system in a safe place to prevent theft. Selling or giving away fentanyl transdermal system may harm others, and is against the law.
- Tell your doctor if you (or a family member) have ever abused or been dependent on alcohol, prescription medicines or street drugs.
Read the Medication Guide that comes with fentanyl transdermal system before you start using it and each time you get a new prescription. There may be new information. This Medication Guide does not take the place of talking to your healthcare provider about your medical condition or your treatment. Make sure you read and understand all the instructions for using fentanyl transdermal system. Do not use fentanyl transdermal system unless you understand everything. Talk to your healthcare provider if you have questions.
What is the most important information I should know about fentanyl transdermal system?
Fentanyl transdermal system is a skin patch that contains fentanyl. Fentanyl is a very strong opioid narcotic pain medicine that can cause serious and life-threatening breathing problems. Serious and life-threatening breathing problems can happen because of an overdose or if the dose you are using is too high for you. Call your doctor right away or get emergency medical help if you:
- have trouble breathing, or have slow or shallow breathing
- have a slow heartbeat
- have severe sleepiness
- have cold, clammy skin
- feel faint, dizzy, confused, or cannot think, walk, or talk normally
- have a seizure
- have hallucinations
Fentanyl transdermal system is only for adults and children over the age of two with persistent, moderate to severe chronic pain and who:
- are already using another strong opioid narcotic pain medicine around-the-clock, and have been using the medicine regularly for a week or longer. This is called being opioid-tolerant
- have pain that cannot be controlled with other medicines
Do not use fentanyl transdermal system:
- if you are not already using another opioid narcotic medicine and are not opioid tolerant
- if you need opioid pain medicines for only a short time
- for pain from surgery, medical or dental procedures
- if your pain can be taken care of by occasional use of other pain medicines
- in children who are less than 2 years of age
- if you have asthma symptoms or have severe asthma
A fentanyl transdermal system must be used only on the skin of the person for whom it was prescribed. If the patch comes off and accidentally sticks to the skin of another person, take the patch off of that person right away, wash the area with water, and get medical care for them right away.
Fentanyl transdermal system is not safe for everyone. Tell your doctor about all of your medical conditions.
Tell your doctor if you are planning to become pregnant, are pregnant, or breastfeeding. Fentanyl transdermal system may cause serious harm to a baby.
Tell your doctor about all the medicines you take. Some medicines may cause serious or life-threatening side effects when used with fentanyl transdermal system. Your doctor will tell you if it is safe to take other medicines while you are using fentanyl transdermal system.
Know the medicines you take. Keep a list of your medicines to show to your doctor and pharmacist.
How should I use fentanyl transdermal system?
Read the separate “Instructions for Applying a Fentanyl Transdermal System.”
-
You must always use fentanyl transdermal systems the right way:
- Do not use a fentanyl transdermal system if the seal is broken, or the patch is cut, damaged, or changed in any way.
- Do not use heat sources such as heating pads, electric blankets, heat lamps, tanning lamps, saunas, hot tubs, or heated waterbeds while wearing a fentanyl transdermal system.
- Do not take hot baths or sunbathe while wearing a fentanyl transdermal system.
- Do not wear this fentanyl transdermal system during any magnetic resonance imaging (MRI) procedures to avoid possible burns.
-
If you have problems with the fentanyl transdermal system not sticking:
- Apply first aid tape only to the edges of the patch.
- If problems with the patch not sticking persist, cover the patch with Bioclusive™. This is a special see-through adhesive dressing. Never cover a fentanyl transdermal system with any other bandage or tape.
- If your fentanyl transdermal system falls off before 3 days or 72 hours, fold the sticky side together and flush down a toilet. Put a new one on at a different skin site.
- Do not change your dose unless your doctor tells you to. Your doctor may change your dose after seeing how the medicine affects you. Do not use fentanyl transdermal system more often than prescribed. Call your doctor if your pain is not well controlled while using fentanyl transdermal system.
- Do not stop using fentanyl transdermal system suddenly. Stopping fentanyl transdermal system suddenly can make you sick with withdrawal symptoms (for example, nausea, vomiting, diarrhea, anxiety, and shivering). Your body can develop a physical dependence on fentanyl transdermal system. If your doctor decides you no longer need fentanyl transdermal system, ask how to slowly reduce this medicine so you don’t have withdrawal symptoms. Do not stop taking fentanyl transdermal system without talking to your doctor.
- Do not wear more than one fentanyl transdermal system at a time, unless your doctor tells you to do so.
-
Call your doctor right away if
- You get a fever higher than 102ºF
- Your body temperature increases from exercise.
A fever or increase in body temperature may cause too much of the medicine in fentanyl transdermal system to pass into your body.
- If you use more fentanyl transdermal systems than your doctor has prescribed, get emergency medical help right away.
- Do not drink any alcohol while using fentanyl transdermal system. Alcohol can increase your chances of having serious side effects.
- Do not drive, operate heavy machinery, or do other possibly dangerous activities until you know how fentanyl transdermal system affects you. Fentanyl transdermal system can make you sleepy. Ask your doctor to tell you when it is okay to do these activities.
- When you remove your fentanyl transdermal system, fold the sticky sides of a used fentanyl transdermal system together and flush it down the toilet. Do not put used fentanyl transdermal systems in a trash can.
What are the possible side effects of fentanyl transdermal system?
Serious side effects include:
- Life-threatening breathing problems. See “What is the most important information I should know about fentanyl transdermal system?”
- Low blood pressure. This can make you feel dizzy if you get up too fast from sitting or lying down.
The common side effects with fentanyl transdermal system are nausea, vomiting, constipation, dry mouth, sleepiness, confusion, weakness, sweating, and pain and redness where the patch was applied.
Constipation is a very common side effect of all opioid medicines. Talk to your doctor about the use of laxatives and stool softeners to prevent or treat constipation while taking fentanyl transdermal system.
Talk to your healthcare provider about any side effect that concerns you.
These are not all the possible side effects of fentanyl transdermal system. For a complete list, ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store fentanyl transdermal system?
- Store fentanyl transdermal system at 20º to 25ºC (68º to 77ºF) [See USP Controlled Room Temperature].
- Keep a fentanyl transdermal system in its protective blister until you are ready to use it.
- Keep fentanyl transdermal system in a safe place out of the reach of children and pets.
- Dispose of fentanyl transdermal systems you no longer need. Open the unused blisters, fold the sticky sides of the patches together, and flush them down the toilet.
General information about the safe and effective use of fentanyl transdermal system
- Do not use fentanyl transdermal system for a condition for which it was not prescribed.
- Do not give fentanyl transdermal system to other people, even if they have the same symptoms you have. Fentanyl transdermal system can harm other people and even cause death. Sharing fentanyl transdermal system is against the law.
- This Medication Guide summarizes the most important information about fentanyl transdermal system. If you would like more information, talk to your doctor. You can ask your doctor or pharmacist for information about fentanyl transdermal system that is written for doctors.
For questions about fentanyl transdermal system, call Teva Pharmaceuticals at 1-888-838-2872, MEDICAL AFFAIRS. If this is a medical emergency, please call 911.
What are the ingredients of fentanyl transdermal system?
Active ingredient: fentanyl
Inactive ingredients: isopropyl myristate, octyldodecanol, polybutene, and polyisobutene adhesive.
This Medication Guide has been approved by the United States Food and Drug Administration.
Bioclusive™ is a trademark of Ethicon, Inc.
-
FENTANYL TRANSDERMAL SYSTEM CII
Instructions for Applying a Fentanyl Transdermal system
Rx only
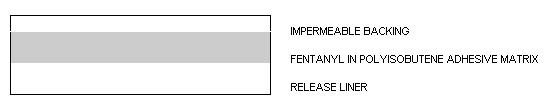
Before Applying Fentanyl Transdermal System
- Fentanyl transdermal system is a patch with medicine inside. The patch is designed to keep the medicine from getting on your hands or body. If the medicine accidentally gets on your skin, wash the area with large amounts of water only. Do not use soap, alcohol, lotions, oils, or other products to remove the medicine because they may increase the medicine’s ability to go through the skin.
- Each fentanyl transdermal system is sealed in its own protective blister. Do not remove a fentanyl transdermal system from the blister until you are ready to use it.
- Do not use a fentanyl transdermal system if the seal is broken or the patch is cut, damaged or changed in any way.
- Fentanyl transdermal systems are available in 4 different doses and patch sizes. Make sure you have the right dose patch or patches that have been prescribed for you.
Applying a Fentanyl Transdermal System
1. Skin Areas Where the Fentanyl Transdermal System May Be Applied:
For adults:
- Put the patch on the chest, back, flank (sides of the waist), or upper arm in a place where there is no hair (see Figures 1 to 4).
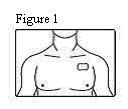
For children (and adults with mental impairment):
- Put the patch on the upper back (see Figure 2). This will lower the chances that the child will remove the patch and put it in their mouth.
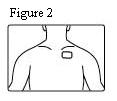
For adults and children
- Do not put a fentanyl transdermal system on skin that is very oily, burned, broken out, cut, irritated, or damaged in any way.
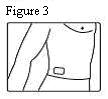
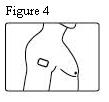
- Avoid sensitive areas or those that move around a lot. If there is hair, do not shave (shaving irritates the skin). Instead, clip hair as close to the skin as possible (see Figure 5).
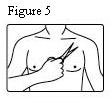
- Talk to your doctor if you have questions about skin application sites.
2. Prepare to Apply a Fentanyl Transdermal System:
- Choose the time of day that is best for you to apply fentanyl transdermal system. Change it at about the same time of day (3 days or 72 hours after you apply the patch) or as directed by your doctor.
- Do not wear more than one fentanyl transdermal system at a time unless your doctor tells you to do so. Before putting on a new fentanyl transdermal system, remove the patch you have been wearing.
- Clean the skin area with clear water only. Pat skin completely dry. Do not use anything on the skin such as soaps, lotions, oils, or alcohol before the patch is applied.
3. Each fentanyl transdermal system is sealed in its own protective blister. Do not remove the fentanyl transdermal system from the blister until you are ready to use it. When you are ready to put on the fentanyl transdermal system, hold the blister so that the fentanyl transdermal system is visible and cut through blister package taking care not to cut through the fentanyl transdermal system. Remove the fentanyl transdermal system from the blister (See Figure 6).
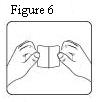
4. Peel: Peel the liner from the back of the patch one half at a time (see Figures 7 and 8) and throw away. Touch the sticky side as little as possible.
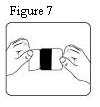
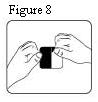
5. Press: Press the patch onto the chosen skin site with the palm of your hand and hold there for at least 30 seconds (see Figure 9). Make sure it sticks well, especially at the edges.
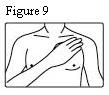
- Fentanyl transdermal system may not stick to all patients. You need to check the patches often to make sure that they are sticking well to the skin.
- If the patch falls off right away after applying, throw it away and put a new one on at a different skin site (see Disposing a Fentanyl Transdermal System).
- If you have a problem with the patch not sticking
- Apply first aid tape only to the edges of the patch.
- If you continue to have problems with the patch sticking, you may cover the patch with Bioclusive™. This is a special see-through adhesive dressing. Never cover a fentanyl transdermal system with any other bandage or tape. Remove the backing from the Bioclusive™ dressing and place it carefully over the fentanyl transdermal system, smoothing it over the patch and your skin.
- If your patch falls off later, but before 3 days (72 hours) of use, discard it properly (see Disposing a Fentanyl Transdermal System) and put a new one on at a different skin site. Be sure to let your doctor know that this has happened, and do not replace the new patch until 3 days (72 hours) after you put it on (or as directed by your doctor).
6. Wash your hands when you have finished applying a fentanyl transdermal system.
7. Remove a fentanyl transdermal system after wearing it for 3 days (72 hours) (see Disposing a Fentanyl Transdermal System). Choose a different place on the skin to apply a new fentanyl transdermal system and repeat Steps 2 through 6.
Do not apply the new patch to the same place as the last one.
Water and Fentanyl Transdermal System
- You can bathe, swim or shower while you are wearing a fentanyl transdermal system. If the patch falls off before 3 days (72 hours) after application, discard it properly (see Disposing a Fentanyl Transdermal System) and put a new one on at a different skin site. Be sure to let your doctor know that this has happened, and do not replace the new patch until 3 days (72 hours) after you put it on (or as directed by your doctor).
Disposing a Fentanyl Transdermal System
- Fold the used fentanyl transdermal system in half so that the sticky side sticks to itself (Figure 10). Flush the used fentanyl transdermal system down the toilet right away (Figure 11). A used fentanyl transdermal system CAN be VERY dangerous for or even lead to death in babies, children, pets, and adults who have not been prescribed fentanyl transdermal system.
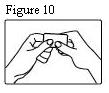
- Throw away any fentanyl transdermal systems that are left over from your prescription as soon as they are no longer needed. Remove the leftover patches from their protective blister and remove the protective liner. Fold the patches in half with the sticky sides together, and flush the patches down the toilet. Do not flush the blister or the protective liner down the toilet. These items can be thrown away in a trashcan.
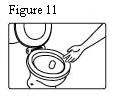
Bioclusive™ is a trademark of Ethicon, Inc.
-
PRINCIPAL DISPLAY PANELS

25 mcg/hr Carton Text
NDC 68258-3023-1 Five (25 mcg/hr) Systems
FENTANYL
TRANSDERMAL SYSTEM
25 mcg/hr CII
5 SYSTEMS
TEVA
In vivo delivery of 25 mcg/hr fentanyl for 72 hours Rx only
Read enclosed Fentanyl Transdermal System Medication Guide for important safety information.
To avoid possible burns, remove the Fentanyl Transdermal System before undergoing
any MRI (magnetic resonance imaging) procedure.
Because it can cause trouble breathing which can be fatal,
DO NOT USE FENTANYL TRANSDERMAL SYSTEM:
For short term or any post-operative pain, or occasional pain
For mild pain or pain that can be treated with non-opioid or
as-needed opioid medication
Unless you have been using other narcotic opioid medicines
(must be opioid tolerant)
ONLY for pain requiring
opioid medicine
around-the-
clock

75 mcg/hr Carton Text
NDC 68258-3024-1 Five (75 mcg/hr) Systems
FENTANYL
TRANSDERMAL SYSTEM
75 mcg/hr CII
5 SYSTEMS
TEVA
In vivo delivery of 75 mcg/hr fentanyl for 72 hours Rx only
Read enclosed Fentanyl Transdermal System Medication Guide for important safety information.
To avoid possible burns, remove the Fentanyl Transdermal System before undergoing
any MRI (magnetic resonance imaging) procedure.
Because it can cause trouble breathing which can be fatal,
DO NOT USE FENTANYL TRANSDERMAL SYSTEM:
For short term or any post-operative pain, or occasional pain
For mild pain or pain that can be treated with non-opioid or
as-needed opioid medication
Unless you have been using other narcotic opioid medicines
(must be opioid tolerant)
ONLY for pain requiring
opioid medicine
around-the-
clock

100 mcg/hr Carton Text
NDC 68258-3025-1 Five (100 mcg/hr) Systems
FENTANYL
TRANSDERMAL SYSTEM
100 mcg/hr CII
5 SYSTEMS
TEVA
In vivo delivery of 100 mcg/hr fentanyl for 72 hours Rx only
Read enclosed Fentanyl Transdermal System Medication Guide for important safety information.
To avoid possible burns, remove the Fentanyl Transdermal System before undergoing
any MRI (magnetic resonance imaging) procedure.
Because it can cause trouble breathing which can be fatal,
DO NOT USE FENTANYL TRANSDERMAL SYSTEM:
For short term or any post-operative pain, or occasional pain
For mild pain or pain that can be treated with non-opioid or
as-needed opioid medication
Unless you have been using other narcotic opioid medicines
(must be opioid tolerant)
ONLY for pain requiring
opioid medicine
around-the-
clock
-
INGREDIENTS AND APPEARANCE
FENTANYL
fentanyl patch, extended releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 68258-3023(NDC: 0093-6900-45) Route of Administration TRANSDERMAL DEA Schedule CII Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength FENTANYL (UNII: UF599785JZ) (FENTANYL - UNII:UF599785JZ) FENTANYL 25 ug in 1 h Inactive Ingredients Ingredient Name Strength ISOPROPYL MYRISTATE (UNII: 0RE8K4LNJS) OCTYLDODECANOL (UNII: 461N1O614Y) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 68258-3023-1 5 in 1 CARTON 1 72 h in 1 PATCH Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA077449 11/06/2006 FENTANYL
fentanyl patch, extended releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 68258-3024(NDC: 0093-6902-45) Route of Administration TRANSDERMAL DEA Schedule CII Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength FENTANYL (UNII: UF599785JZ) (FENTANYL - UNII:UF599785JZ) FENTANYL 75 ug in 1 h Inactive Ingredients Ingredient Name Strength ISOPROPYL MYRISTATE (UNII: 0RE8K4LNJS) OCTYLDODECANOL (UNII: 461N1O614Y) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 68258-3024-1 5 in 1 CARTON 1 72 h in 1 PATCH Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA077449 11/06/2006 FENTANYL
fentanyl patch, extended releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 68258-3025(NDC: 0093-6903-45) Route of Administration TRANSDERMAL DEA Schedule CII Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength FENTANYL (UNII: UF599785JZ) (FENTANYL - UNII:UF599785JZ) FENTANYL 100 ug in 1 h Inactive Ingredients Ingredient Name Strength ISOPROPYL MYRISTATE (UNII: 0RE8K4LNJS) OCTYLDODECANOL (UNII: 461N1O614Y) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 68258-3025-1 5 in 1 CARTON 1 72 h in 1 PATCH Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA077449 11/06/2006 Labeler - Dispensing Solutions Inc. (066070785) Establishment Name Address ID/FEI Business Operations Dispensing Solutions Inc. 066070785 repack, relabel
Trademark Results [Fentanyl]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 FENTANYL 98644202 not registered Live/Pending |
Ghost Global LLC 2024-07-11 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.