quinine sulfate capsules USP 324mg
Qualaquin by
Drug Labeling and Warnings
Qualaquin by is a Prescription medication manufactured, distributed, or labeled by Frontida BioPharm, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
QUALAQUIN- quinine sulfate capsule
Frontida BioPharm, Inc.
----------
quinine sulfate capsules USP 324mg
Principal Display Panel
NDC: 71557-050-30
Quinine Sulfate Capsules USP
324 mg
DISPENSE THE ACCOMPANYING MEDICATION GUIDE TO EACH PATIENT
Rx Only
30 Tablets
SUN PHARMA

Principal Display Panel
NDC: 71557-047-30
30 CAPSULES
Qualaquin ®
quinine sulfate capsules USP
324 mg
DISPENSE THE ACCOMPANYING MEDICATION GUIDE TO EACH PATIENT
Rx Only
SUN PHARMA

| QUALAQUIN
quinine sulfate capsule |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Labeler - Frontida BioPharm, Inc. (080243260) |
Revised: 1/2021
Document Id: 5fdcdaf4-0760-4db6-b4b5-549e2154479f
Set id: 0cc93792-8c06-4a6e-a5cb-d11ef44272e9
Version: 4
Effective Time: 20210125
Trademark Results [Qualaquin]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
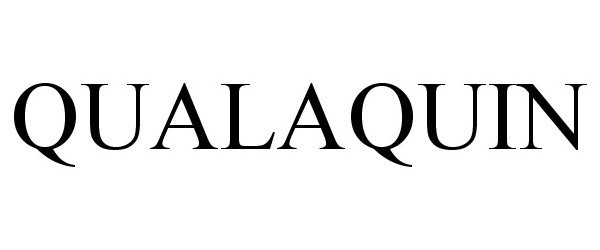 QUALAQUIN 78835655 3263197 Live/Registered |
MUTUAL PHARMACEUTICAL COMPANY, INC. 2006-03-13 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.