Zynalox Pain Relief Tape
Zynalox Pain Relief Tape by
Drug Labeling and Warnings
Zynalox Pain Relief Tape by is a Otc medication manufactured, distributed, or labeled by V2 Pharma LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
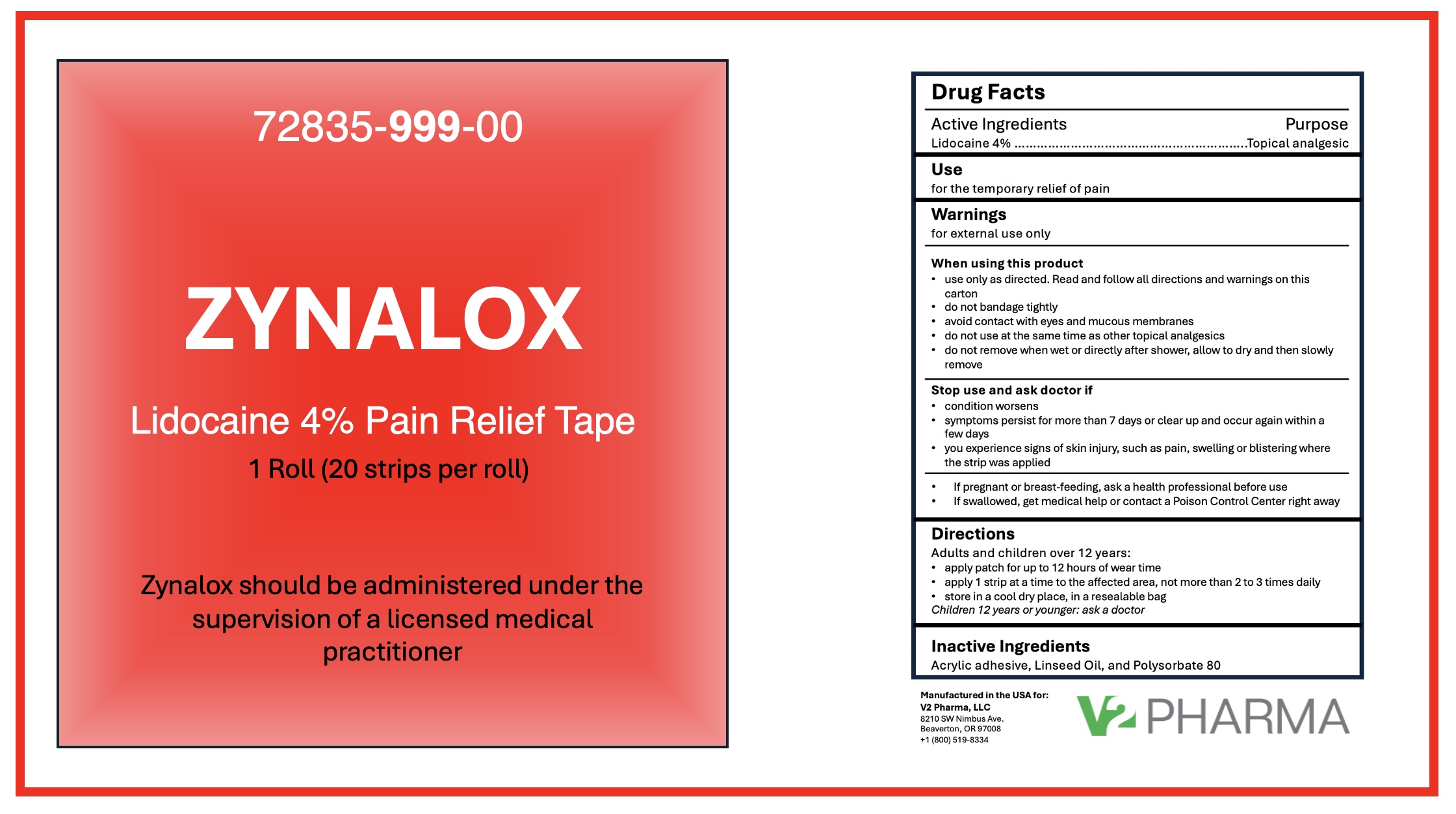
ZYNALOX PAIN RELIEF TAPE- lidocaine 4% tape
V2 Pharma LLC
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
Zynalox Pain Relief Tape
Warnings
for external use only
When using this product
use only as directed. Read and follow all directions and warnings on this package
do not bandage tightly
avoid contact with eyes and mucous membranes
do not use at the same time as other topical analgesics
do not remove when wet or directly after shower, allow to dry and then slowly remove
Stop use and ask a doctor if
condition worsens
symptoms persist for more than 7 days or clear up and occur again within a few days
you experience signs of skin injury, such as pain, swelling or blistering where the tape was applied
If pregnant or breast-feeding, ask a health professional before use
Directions
adults and children over 12 years:
apply strip for up to 12 hours of wear time
apply 1 strip at a time to the affected area, not more than 2 to 3 times daily
store in a cool dry place
children 12 years or younger: ask a doctor
| ZYNALOX PAIN RELIEF TAPE
lidocaine 4% tape |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - V2 Pharma LLC (102457346) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Active Intelligence, LLC | 080416593 | manufacture(72835-999) | |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.