CVS Health Pain Relief by NATURAL ESSENTIALS, INC. Drug Facts
CVS Health Pain Relief by
Drug Labeling and Warnings
CVS Health Pain Relief by is a Otc medication manufactured, distributed, or labeled by NATURAL ESSENTIALS, INC.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
CVS HEALTH PAIN RELIEF- menthol, unspecified form gel
NATURAL ESSENTIALS, INC.
----------
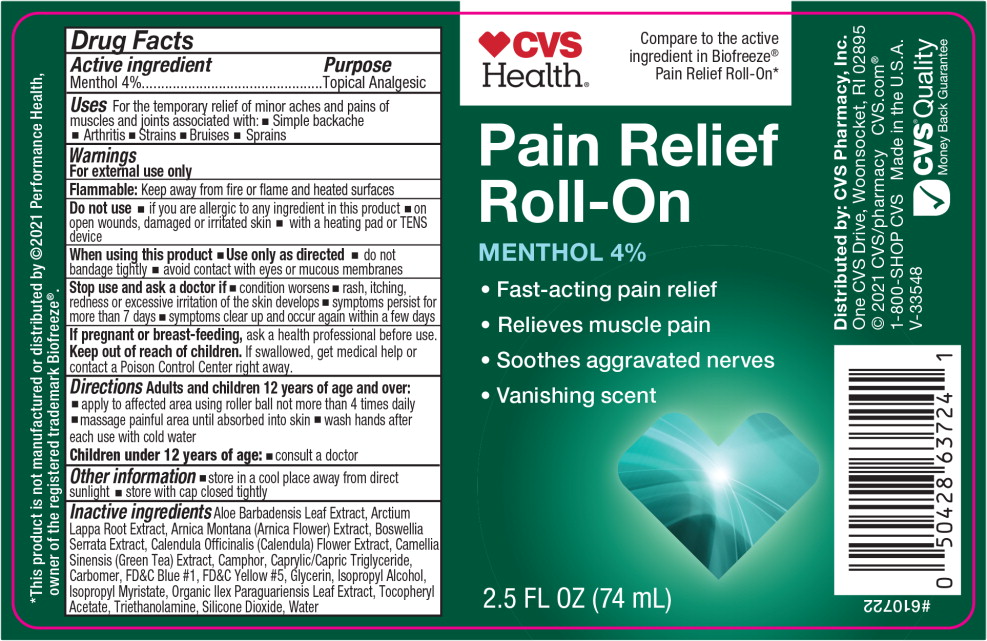
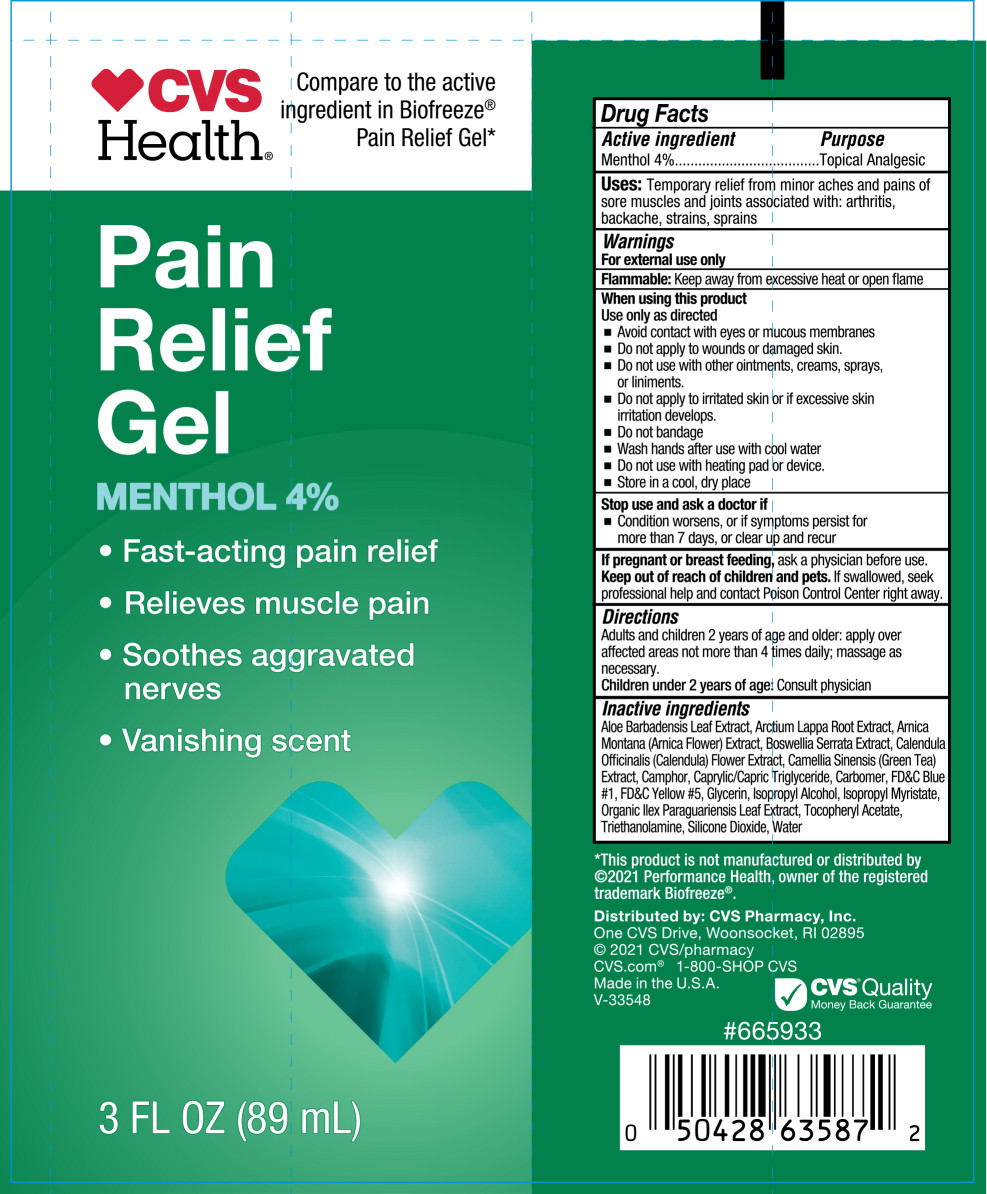
Drug Facts
Uses
For the temporary relief of minor aches and pains of muscles and joints associated with:
- Simple backache
- Arthritis
- Strains
- Bruises
- Sprains
Warnings
For external use only
Flammable:Keep away from fire or flame and heated surfaces
Do not use
- if you are allergic to any ingredient in this product
- on open wounds, damaged or irritated skin
- with a heating pad or TENS device
When using this product
- Use only as directed
- do not bandage tightly
- avoid contact with eyes or mucous membranes
Directions
Inactive ingredients
Aloe Barbadensis Leaf Extract, Arctium Lappa Root Extract, Arnica Montana (Arnica Flower) Extract, Boswellia Serrata Extract, Calendula Officinalis (Calendula) Flower Extract, Camellia Sinensis (Green Tea) Extract, Camphor, Caprylic/Capric Triglyceride, Carbomer, FD&C Blue #1, FD&C Yellow #5, Glycerin, Isopropyl Alcohol, Isopropyl Myristate, Organic Ilex Paraguariensis Leaf Extract, Tocopheryl Acetate, Triethanolamine, Silicone Dioxide, Water
| CVS HEALTH PAIN RELIEF
menthol, unspecified form gel |
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
| Labeler - NATURAL ESSENTIALS, INC. (947484713) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| NATURAL ESSENTIALS, INC. | 947484713 | manufacture(66902-920) | |