XARELTO- rivaroxaban tablet, film coated XARELTO- rivaroxaban tablet, film coated XARELTO- rivaroxaban kit
XARELTO by
Drug Labeling and Warnings
XARELTO by is a Prescription medication manufactured, distributed, or labeled by Janssen Pharmaceuticals, Inc., Bayer AG, Bayer Aktiengesellschaft, Janssen Ortho LLC, AndersonBrecon Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use XARELTO® (rivaroxaban) safely and effectively. See full prescribing information for XARELTO.
XARELTO (rivaroxaban) tablets, for oral use
Initial U.S. Approval: 2011WARNING: (A) PREMATURE DISCONTINUATION OF XARELTO INCREASES THE RISK OF THROMBOTIC EVENTS, (B) SPINAL/EPIDURAL HEMATOMA
See full prescribing information for complete boxed warning.
(A) Premature discontinuation of XARELTO increases the risk of thrombotic events
Premature discontinuation of any oral anticoagulant, including XARELTO, increases the risk of thrombotic events. To reduce this risk, consider coverage with another anticoagulant if XARELTO is discontinued for a reason other than pathological bleeding or completion of a course of therapy. (2.2, 2.3, 5.1, 14.1)
(B) Spinal/epidural hematoma
Epidural or spinal hematomas have occurred in patients treated with XARELTO who are receiving neuraxial anesthesia or undergoing spinal puncture. These hematomas may result in long-term or permanent paralysis. (5.2, 5.3, 6.2)
Monitor patients frequently for signs and symptoms of neurological impairment and if observed, treat urgently. Consider the benefits and risks before neuraxial intervention in patients who are or who need to be anticoagulated. (5.3)
RECENT MAJOR CHANGES
INDICATIONS AND USAGE
XARELTO is a factor Xa inhibitor indicated:
- to reduce risk of stroke and systemic embolism in nonvalvular atrial fibrillation (1.1)
- for treatment of deep vein thrombosis (DVT) (1.2)
- for treatment of pulmonary embolism (PE) (1.3)
- for reduction in the risk of recurrence of DVT or PE (1.4)
- for the prophylaxis of DVT, which may lead to PE in patients undergoing knee or hip replacement surgery (1.5)
- for prophylaxis of venous thromboembolism (VTE) in acutely ill medical patients (1.6)
- to reduce the risk of major cardiovascular events in patients with chronic coronary artery disease (CAD) or peripheral artery disease (PAD) (1.7)
DOSAGE AND ADMINISTRATION
- Nonvalvular Atrial Fibrillation: 15 or 20 mg, once daily with food (2.1)
- Treatment of DVT and/or PE: 15 mg orally twice daily with food for the first 21 days followed by 20 mg orally once daily with food for the remaining treatment (2.1)
- Reduction in the Risk of Recurrence of DVT and/or PE in patients at continued risk for DVT and/or PE: 10 mg once daily with or without food, after at least 6 months of standard anticoagulant treatment (2.1)
- Prophylaxis of DVT Following Hip or Knee Replacement Surgery: 10 mg orally once daily with or without food (2.1)
- Prophylaxis of VTE in Acutely Ill Medical Patients at Risk for Thromboembolic Complications Not at High Risk of Bleeding: 10 mg once daily, with or without food, in hospital and after hospital discharge for a total recommended duration of 31 to 39 days (2.1)
- CAD or PAD: 2.5 mg orally twice daily with or without food, in combination with aspirin (75–100 mg) once daily (2.1)
DOSAGE FORMS AND STRENGTHS
Tablets: 2.5 mg, 10 mg, 15 mg, and 20 mg (3)
WARNINGS AND PRECAUTIONS
- Risk of bleeding: XARELTO can cause serious and fatal bleeding. An agent to reverse the activity of rivaroxaban is available. (5.2)
- Pregnancy-related hemorrhage: Use XARELTO with caution in pregnant women due to the potential for obstetric hemorrhage and/or emergent delivery. (5.7, 8.1)
- Prosthetic heart valves: XARELTO use not recommended (5.8)
- Increased Risk of Thrombosis in Patients with Triple Positive Antiphospholipid Syndrome: XARELTO use not recommended. (5.10)
ADVERSE REACTIONS
The most common adverse reaction (>5%) was bleeding. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Janssen Pharmaceuticals, Inc. at 1-800-526-7736 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
USE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 4/2020
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: (A) PREMATURE DISCONTINUATION OF XARELTO INCREASES THE RISK OF THROMBOTIC EVENTS, (B) SPINAL/EPIDURAL HEMATOMA
1 INDICATIONS AND USAGE
1.1 Reduction of Risk of Stroke and Systemic Embolism in Nonvalvular Atrial Fibrillation
1.2 Treatment of Deep Vein Thrombosis
1.3 Treatment of Pulmonary Embolism
1.4 Reduction in the Risk of Recurrence of Deep Vein Thrombosis and/or Pulmonary Embolism
1.5 Prophylaxis of Deep Vein Thrombosis Following Hip or Knee Replacement Surgery
1.6 Prophylaxis of Venous Thromboembolism in Acutely Ill Medical Patients at Risk for Thromboembolic Complications Not at High Risk of Bleeding
1.7 Reduction of Risk of Major Cardiovascular Events in Patients with Chronic Coronary Artery Disease (CAD) or Peripheral Artery Disease (PAD)
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage
2.2 Switching to and from XARELTO
2.3 Discontinuation for Surgery and other Interventions
2.4 Missed Dose
2.5 Administration Options
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Increased Risk of Thrombotic Events after Premature Discontinuation
5.2 Risk of Bleeding
5.3 Spinal/Epidural Anesthesia or Puncture
5.4 Use in Patients with Renal Impairment
5.5 Use in Patients with Hepatic Impairment
5.6 Use with P-gp and Strong CYP3A Inhibitors or Inducers
5.7 Risk of Pregnancy-Related Hemorrhage
5.8 Patients with Prosthetic Heart Valves
5.9 Acute PE in Hemodynamically Unstable Patients or Patients Who Require Thrombolysis or Pulmonary Embolectomy
5.10 Increased Risk of Thrombosis in Patients with Triple Positive Antiphospholipid Syndrome
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 General Inhibition and Induction Properties
7.2 Drugs that Inhibit Cytochrome P450 3A Enzymes and Drug Transport Systems
7.3 Drugs that Induce Cytochrome P450 3A Enzymes and Drug Transport Systems
7.4 Anticoagulants and NSAIDs/Aspirin
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
8.7 Hepatic Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
12.6 QT/QTc Prolongation
13 NON-CLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Stroke Prevention in Nonvalvular Atrial Fibrillation
14.2 Treatment of Deep Vein Thrombosis (DVT) and/or Pulmonary Embolism (PE)
14.3 Reduction in the Risk of Recurrence of DVT and/or PE
14.4 Prophylaxis of Deep Vein Thrombosis Following Hip or Knee Replacement Surgery
14.5 Prophylaxis of Venous Thromboembolism in Acutely Ill Medical Patients at Risk for Thromboembolic Complications Not at High Risk of Bleeding
14.6 Reduction of Risk of Major Cardiovascular Events in Patients with Chronic CAD or PAD
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: (A) PREMATURE DISCONTINUATION OF XARELTO INCREASES THE RISK OF THROMBOTIC EVENTS, (B) SPINAL/EPIDURAL HEMATOMA
A. Premature discontinuation of XARELTO increases the risk of thrombotic events
Premature discontinuation of any oral anticoagulant, including XARELTO, increases the risk of thrombotic events. If anticoagulation with XARELTO is discontinued for a reason other than pathological bleeding or completion of a course of therapy, consider coverage with another anticoagulant [see Dosage and Administration (2.2, 2.3), Warnings and Precautions (5.1), and Clinical Studies (14.1)].
B. Spinal/epidural hematoma
Epidural or spinal hematomas have occurred in patients treated with XARELTO who are receiving neuraxial anesthesia or undergoing spinal puncture. These hematomas may result in long-term or permanent paralysis. Consider these risks when scheduling patients for spinal procedures. Factors that can increase the risk of developing epidural or spinal hematomas in these patients include:
- use of indwelling epidural catheters
- concomitant use of other drugs that affect hemostasis, such as non-steroidal anti-inflammatory drugs (NSAIDs), platelet inhibitors, other anticoagulants
- a history of traumatic or repeated epidural or spinal punctures
- a history of spinal deformity or spinal surgery
- optimal timing between the administration of XARELTO and neuraxial procedures is not known
[see Warnings and Precautions (5.2, 5.3) and Adverse Reactions (6.2)].
Monitor patients frequently for signs and symptoms of neurological impairment. If neurological compromise is noted, urgent treatment is necessary [see Warnings and Precautions (5.3)].
Consider the benefits and risks before neuraxial intervention in patients anticoagulated or to be anticoagulated for thromboprophylaxis [see Warnings and Precautions (5.3)].
-
1 INDICATIONS AND USAGE
1.1 Reduction of Risk of Stroke and Systemic Embolism in Nonvalvular Atrial Fibrillation
XARELTO is indicated to reduce the risk of stroke and systemic embolism in patients with nonvalvular atrial fibrillation.
There are limited data on the relative effectiveness of XARELTO and warfarin in reducing the risk of stroke and systemic embolism when warfarin therapy is well-controlled [see Clinical Studies (14.1)].
1.2 Treatment of Deep Vein Thrombosis
XARELTO is indicated for the treatment of deep vein thrombosis (DVT).
1.3 Treatment of Pulmonary Embolism
XARELTO is indicated for the treatment of pulmonary embolism (PE).
1.4 Reduction in the Risk of Recurrence of Deep Vein Thrombosis and/or Pulmonary Embolism
XARELTO is indicated for the reduction in the risk of recurrence of DVT and/or PE in patients at continued risk for recurrent DVT and/or PE after completion of initial treatment lasting at least 6 months.
1.5 Prophylaxis of Deep Vein Thrombosis Following Hip or Knee Replacement Surgery
XARELTO is indicated for the prophylaxis of DVT, which may lead to PE in patients undergoing knee or hip replacement surgery.
1.6 Prophylaxis of Venous Thromboembolism in Acutely Ill Medical Patients at Risk for Thromboembolic Complications Not at High Risk of Bleeding
XARELTO is indicated for the prophylaxis of venous thromboembolism (VTE) and VTE related death during hospitalization and post hospital discharge in adult patients admitted for an acute medical illness who are at risk for thromboembolic complications due to moderate or severe restricted mobility and other risk factors for VTE and not at high risk of bleeding [see Warnings and Precautions (5.2) and Clinical Studies (14.1)].
1.7 Reduction of Risk of Major Cardiovascular Events in Patients with Chronic Coronary Artery Disease (CAD) or Peripheral Artery Disease (PAD)
XARELTO, in combination with aspirin, is indicated to reduce the risk of major cardiovascular events (cardiovascular (CV) death, myocardial infarction (MI) and stroke) in patients with chronic coronary artery disease (CAD) or peripheral artery disease (PAD).
-
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage
Table 1: Recommended Dosage Indication Renal Considerations* Dosage Food/Timing† - * Calculate CrCl based on actual weight. See Warnings and Precautions (5.4) and Use in Specific Populations (8.6)
- † See Clinical Pharmacology (12.3)
- ‡ Patients with CrCl <30 mL/min were not studied, but administration of XARELTO is expected to result in serum concentrations of rivaroxaban similar to those in patients with moderate renal impairment (CrCl 30 to <50 mL/min) [see Use in Specific Populations (8.6)]
- § See Dosage and Administration (2.3)
Reduction in Risk of Stroke in Nonvalvular Atrial Fibrillation CrCl >50 mL/min 20 mg once daily Take with evening meal CrCl ≤50 mL/min‡ 15 mg once daily Take with evening meal Treatment of DVT and/or PE CrCl ≥15 mL/min‡ 15 mg twice daily
▼ after 21 days, transition to ▼
20 mg once dailyTake with food, at the same time each day CrCl <15 mL/min Avoid Use Reduction in the Risk of Recurrence of DVT and/or PE in patients at continued risk for DVT and/or PE CrCl ≥15 mL/min‡ 10 mg once daily, after at least 6 months of standard anticoagulant treatment Take with or without food CrCl <15 mL/min Avoid Use Prophylaxis of DVT Following: - - Hip Replacement Surgery§
CrCl ≥15 mL/min‡ 10 mg once daily for 35 days, 6–10 hours after surgery once hemostasis has been established Take with or without food CrCl <15 mL/min Avoid Use - - Knee Replacement Surgery§
CrCl ≥15 mL/min‡ 10 mg once daily for 12 days, 6–10 hours after surgery once hemostasis has been established Take with or without food CrCl <15 mL/min Avoid Use Prophylaxis of VTE in Acutely Ill Medical Patients at Risk for Thromboembolic Complications Not at High Risk of Bleeding CrCl ≥15 mL/min‡ 10 mg once daily, in hospital and after hospital discharge, for a total recommended duration of 31 to 39 days Take with or without food CrCl <15 mL/min Avoid Use Reduction of Risk of Major Cardiovascular Events (CV Death, MI, and Stroke) in Chronic CAD or PAD No dose adjustment needed based on CrCl 2.5 mg twice daily, plus aspirin (75–100 mg) once daily Take with or without food 2.2 Switching to and from XARELTO
Switching from Warfarin to XARELTO - When switching patients from warfarin to XARELTO, discontinue warfarin and start XARELTO as soon as the International Normalized Ratio (INR) is below 3.0 to avoid periods of inadequate anticoagulation.
Switching from XARELTO to Warfarin - No clinical trial data are available to guide converting patients from XARELTO to warfarin. XARELTO affects INR, so INR measurements made during coadministration with warfarin may not be useful for determining the appropriate dose of warfarin. One approach is to discontinue XARELTO and begin both a parenteral anticoagulant and warfarin at the time the next dose of XARELTO would have been taken.
Switching from XARELTO to Anticoagulants other than Warfarin - For patients currently taking XARELTO and transitioning to an anticoagulant with rapid onset, discontinue XARELTO and give the first dose of the other anticoagulant (oral or parenteral) at the time that the next XARELTO dose would have been taken [see Drug Interactions (7.4)].
Switching from Anticoagulants other than Warfarin to XARELTO - For patients currently receiving an anticoagulant other than warfarin, start XARELTO 0 to 2 hours prior to the next scheduled evening administration of the drug (e.g., low molecular weight heparin or non-warfarin oral anticoagulant) and omit administration of the other anticoagulant. For unfractionated heparin being administered by continuous infusion, stop the infusion and start XARELTO at the same time.
2.3 Discontinuation for Surgery and other Interventions
If anticoagulation must be discontinued to reduce the risk of bleeding with surgical or other procedures, XARELTO should be stopped at least 24 hours before the procedure to reduce the risk of bleeding [see Warnings and Precautions (5.2)]. In deciding whether a procedure should be delayed until 24 hours after the last dose of XARELTO, the increased risk of bleeding should be weighed against the urgency of intervention. XARELTO should be restarted after the surgical or other procedures as soon as adequate hemostasis has been established, noting that the time to onset of therapeutic effect is short [see Warnings and Precautions (5.1)]. If oral medication cannot be taken during or after surgical intervention, consider administering a parenteral anticoagulant.
2.4 Missed Dose
- For patients receiving 2.5 mg twice daily: if a dose is missed, the patient should take a single 2.5 mg XARELTO dose as recommended at the next scheduled time.
- For patients receiving 15 mg twice daily: The patient should take XARELTO immediately to ensure intake of 30 mg XARELTO per day. Two 15 mg tablets may be taken at once.
- For patients receiving 20 mg, 15 mg or 10 mg once daily: The patient should take the missed XARELTO dose immediately. The dose should not be doubled within the same day to make up for a missed dose.
2.5 Administration Options
For patients who are unable to swallow whole tablets, XARELTO tablets (all strengths) may be crushed and mixed with applesauce immediately prior to use and administered orally. After the administration of a crushed XARELTO 15 mg or 20 mg tablet, the dose should be immediately followed by food. Administration with food is not required for the 2.5 mg or 10 mg tablets [see Clinical Pharmacology (12.3)].
Administration via nasogastric (NG) tube or gastric feeding tube: After confirming gastric placement of the tube, XARELTO tablets (all strengths) may be crushed and suspended in 50 mL of water and administered via an NG tube or gastric feeding tube. Since rivaroxaban absorption is dependent on the site of drug release, avoid administration of XARELTO distal to the stomach which can result in reduced absorption and thereby, reduced drug exposure. After the administration of a crushed XARELTO 15 mg or 20 mg tablet, the dose should then be immediately followed by enteral feeding. Enteral feeding is not required following administration of the 2.5 mg or 10 mg tablets [see Clinical Pharmacology (12.3)].
Crushed XARELTO tablets (all strengths) are stable in water and in applesauce for up to 4 hours. An in vitro compatibility study indicated that there is no adsorption of rivaroxaban from a water suspension of a crushed XARELTO tablet to PVC or silicone nasogastric (NG) tubing.
-
3 DOSAGE FORMS AND STRENGTHS
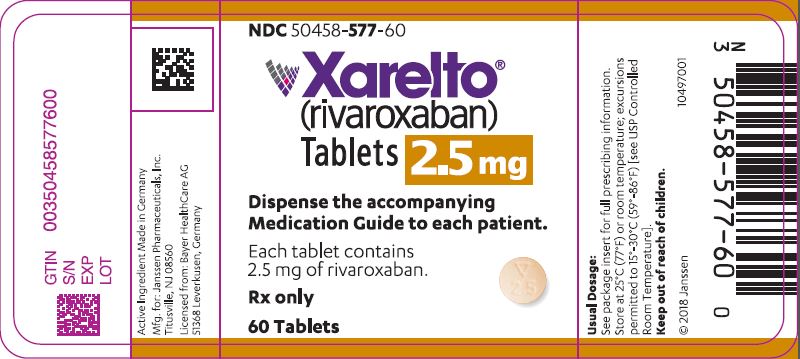
- 2.5 mg tablets: Round, light yellow, and film-coated with a triangle pointing down above a "2.5" marked on one side and "Xa" on the other side
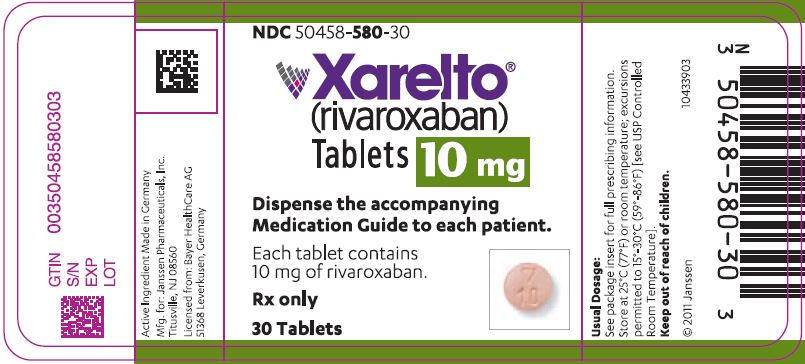
- 10 mg tablets: Round, light red, biconvex and film-coated with a triangle pointing down above a "10" marked on one side and "Xa" on the other side
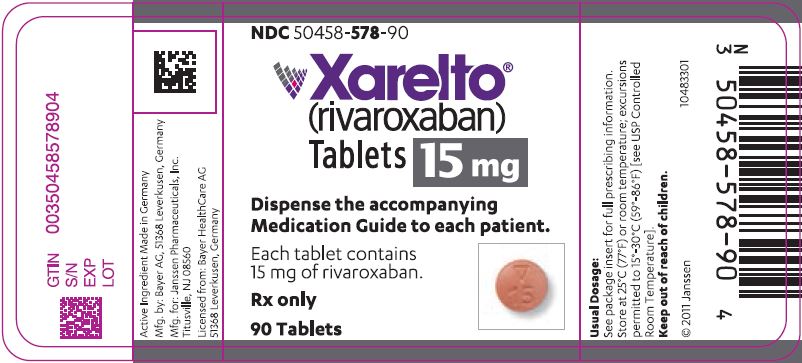
- 15 mg tablets: Round, red, biconvex, and film-coated with a triangle pointing down above a "15" marked on one side and "Xa" on the other side
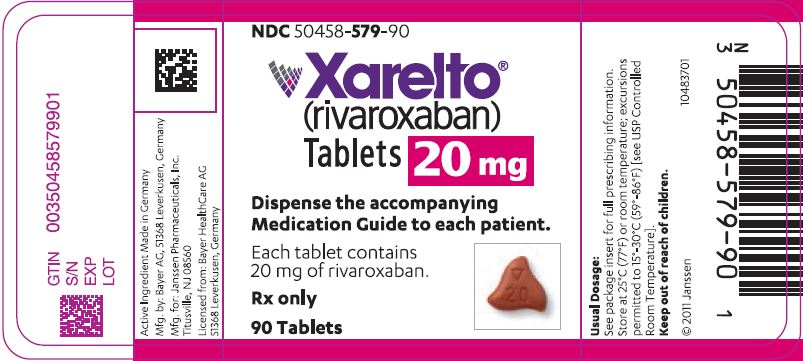
- 20 mg tablets: Triangle-shaped, dark red, and film-coated with a triangle pointing down above a "20" marked on one side and "Xa" on the other side
-
4 CONTRAINDICATIONS
XARELTO is contraindicated in patients with:
- active pathological bleeding [see Warnings and Precautions (5.2)]
- severe hypersensitivity reaction to XARELTO (e.g., anaphylactic reactions) [see Adverse Reactions (6.2)]
-
5 WARNINGS AND PRECAUTIONS
5.1 Increased Risk of Thrombotic Events after Premature Discontinuation
Premature discontinuation of any oral anticoagulant, including XARELTO, in the absence of adequate alternative anticoagulation increases the risk of thrombotic events. An increased rate of stroke was observed during the transition from XARELTO to warfarin in clinical trials in atrial fibrillation patients. If XARELTO is discontinued for a reason other than pathological bleeding or completion of a course of therapy, consider coverage with another anticoagulant [see Dosage and Administration (2.2, 2.3) and Clinical Studies (14.1)].
5.2 Risk of Bleeding
XARELTO increases the risk of bleeding and can cause serious or fatal bleeding. In deciding whether to prescribe XARELTO to patients at increased risk of bleeding, the risk of thrombotic events should be weighed against the risk of bleeding.
Promptly evaluate any signs or symptoms of blood loss and consider the need for blood replacement. Discontinue XARELTO in patients with active pathological hemorrhage. The terminal elimination half-life of rivaroxaban is 5 to 9 hours in healthy subjects aged 20 to 45 years.
Concomitant use of other drugs that impair hemostasis increases the risk of bleeding. These include aspirin, P2Y12 platelet inhibitors, dual antiplatelet therapy, other antithrombotic agents, fibrinolytic therapy, non-steroidal anti-inflammatory drugs (NSAIDs) [see Drug Interactions (7.4)], selective serotonin reuptake inhibitors, and serotonin norepinephrine reuptake inhibitors.
Concomitant use of drugs that are known combined P-gp and strong CYP3A inhibitors increases rivaroxaban exposure and may increase bleeding risk [see Drug Interactions (7.2)].
Risk of Hemorrhage in Acutely Ill Medical Patients at High Risk of Bleeding
Acutely ill medical patients with the following conditions are at increased risk of bleeding with the use of XARELTO for primary VTE prophylaxis: history of bronchiectasis, pulmonary cavitation, or pulmonary hemorrhage, active cancer (i.e. undergoing acute, in-hospital cancer treatment), active gastroduodenal ulcer in the three months prior to treatment, history of bleeding in the three months prior to treatment, or dual antiplatelet therapy. XARELTO is not for use for primary VTE prophylaxis in these hospitalized, acutely ill medical patients at high risk of bleeding.
Reversal of Anticoagulant Effect
An agent to reverse the anti-factor Xa activity of rivaroxaban is available. Because of high plasma protein binding, rivaroxaban is not dialyzable [see Clinical Pharmacology (12.3)]. Protamine sulfate and vitamin K are not expected to affect the anticoagulant activity of rivaroxaban. Use of procoagulant reversal agents, such as prothrombin complex concentrate (PCC), activated prothrombin complex concentrate or recombinant factor VIIa, may be considered but has not been evaluated in clinical efficacy and safety studies. Monitoring for the anticoagulation effect of rivaroxaban using a clotting test (PT, INR or aPTT) or anti-factor Xa (FXa) activity is not recommended.
5.3 Spinal/Epidural Anesthesia or Puncture
When neuraxial anesthesia (spinal/epidural anesthesia) or spinal puncture is employed, patients treated with anticoagulant agents for prevention of thromboembolic complications are at risk of developing an epidural or spinal hematoma which can result in long-term or permanent paralysis [see Boxed Warning].
To reduce the potential risk of bleeding associated with the concurrent use of XARELTO and epidural or spinal anesthesia/analgesia or spinal puncture, consider the pharmacokinetic profile of XARELTO [see Clinical Pharmacology (12.3)]. Placement or removal of an epidural catheter or lumbar puncture is best performed when the anticoagulant effect of XARELTO is low; however, the exact timing to reach a sufficiently low anticoagulant effect in each patient is not known.
An indwelling epidural or intrathecal catheter should not be removed before at least 2 half-lives have elapsed (i.e., 18 hours in young patients aged 20 to 45 years and 26 hours in elderly patients aged 60 to 76 years), after the last administration of XARELTO [see Clinical Pharmacology (12.3)]. The next XARELTO dose should not be administered earlier than 6 hours after the removal of the catheter. If traumatic puncture occurs, delay the administration of XARELTO for 24 hours.
Should the physician decide to administer anticoagulation in the context of epidural or spinal anesthesia/analgesia or lumbar puncture, monitor frequently to detect any signs or symptoms of neurological impairment, such as midline back pain, sensory and motor deficits (numbness, tingling, or weakness in lower limbs), bowel and/or bladder dysfunction. Instruct patients to immediately report if they experience any of the above signs or symptoms. If signs or symptoms of spinal hematoma are suspected, initiate urgent diagnosis and treatment including consideration for spinal cord decompression even though such treatment may not prevent or reverse neurological sequelae.
5.4 Use in Patients with Renal Impairment
Nonvalvular Atrial Fibrillation
Periodically assess renal function as clinically indicated (i.e., more frequently in situations in which renal function may decline) and adjust therapy accordingly [see Dosage and Administration (2.1)]. Consider dose adjustment or discontinuation of XARELTO in patients who develop acute renal failure while on XARELTO [see Use in Specific Populations (8.6)].
Treatment of Deep Vein Thrombosis (DVT), Pulmonary Embolism (PE), and Reduction in the Risk of Recurrence of DVT and of PE
In patients with CrCl <30 mL/min, rivaroxaban exposure and pharmacodynamic effects are increased compared to patients with normal renal function. There are limited clinical data in patients with CrCl 15 to <30 mL/min; therefore, observe closely and promptly evaluate any signs or symptoms of blood loss in these patients. There are no clinical data in patients with CrCl <15 mL/min (including patients on dialysis); therefore, avoid the use of XARELTO in these patients.
Discontinue XARELTO in patients who develop acute renal failure while on treatment [see Use in Specific Populations (8.6)].
Prophylaxis of Deep Vein Thrombosis Following Hip or Knee Replacement Surgery
In patients with CrCl <30 mL/min, rivaroxaban exposure and pharmacodynamic effects are increased compared to patients with normal renal function. There are limited clinical data in patients with CrCl 15 to <30 mL/min; therefore, observe closely and promptly evaluate any signs or symptoms of blood loss in these patients. There are no clinical data in patients with CrCl <15 mL/min (including patients on dialysis); therefore, avoid the use of XARELTO in these patients.
Discontinue XARELTO in patients who develop acute renal failure while on treatment [see Use in Specific Populations (8.6)].
Prophylaxis of Venous Thromboembolism in Acutely Ill Medical Patients at Risk for Thromboembolic Complications Not at High Risk of Bleeding
In patients with CrCl <30 mL/min, rivaroxaban exposure and pharmacodynamic effects are increased compared to patients with normal renal function. There are limited clinical data in patients with CrCl 15 to <30 mL/min; therefore, observe closely and promptly evaluate any signs or symptoms of blood loss in these patients. There are no clinical data in patients with CrCl <15 mL/min (including patients on dialysis); therefore, avoid the use of XARELTO in these patients.
Discontinue XARELTO in patients who develop acute renal failure while on treatment [see Use in Specific Populations (8.6)].
5.5 Use in Patients with Hepatic Impairment
No clinical data are available for patients with severe hepatic impairment.
Avoid use of XARELTO in patients with moderate (Child-Pugh B) and severe (Child-Pugh C) hepatic impairment or with any hepatic disease associated with coagulopathy since drug exposure and bleeding risk may be increased [see Use in Specific Populations (8.7)].
5.6 Use with P-gp and Strong CYP3A Inhibitors or Inducers
Avoid concomitant use of XARELTO with known combined P-gp and strong CYP3A inhibitors [see Drug Interactions (7.2)].
Avoid concomitant use of XARELTO with drugs that are known combined P-gp and strong CYP3A inducers [see Drug Interactions (7.3)].
5.7 Risk of Pregnancy-Related Hemorrhage
In pregnant women, XARELTO should be used only if the potential benefit justifies the potential risk to the mother and fetus. XARELTO dosing in pregnancy has not been studied. The anticoagulant effect of XARELTO cannot be monitored with standard laboratory testing. Promptly evaluate any signs or symptoms suggesting blood loss (e.g., a drop in hemoglobin and/or hematocrit, hypotension, or fetal distress) [see Warnings and Precautions (5.2) and Use in Specific Populations (8.1)].
5.8 Patients with Prosthetic Heart Valves
On the basis of the GALILEO study, use of XARELTO is not recommended in patients who have had transcatheter aortic valve replacement (TAVR) because patients randomized to XARELTO experienced higher rates of death and bleeding compared to those randomized to an anti-platelet regimen. The safety and efficacy of XARELTO have not been studied in patients with other prosthetic heart valves or other valve procedures. Use of XARELTO is not recommended in patients with prosthetic heart valves.
5.9 Acute PE in Hemodynamically Unstable Patients or Patients Who Require Thrombolysis or Pulmonary Embolectomy
Initiation of XARELTO is not recommended acutely as an alternative to unfractionated heparin in patients with pulmonary embolism who present with hemodynamic instability or who may receive thrombolysis or pulmonary embolectomy.
5.10 Increased Risk of Thrombosis in Patients with Triple Positive Antiphospholipid Syndrome
Direct-acting oral anticoagulants (DOACs), including XARELTO, are not recommended for use in patients with triple-positive antiphospholipid syndrome (APS). For patients with APS (especially those who are triple positive [positive for lupus anticoagulant, anticardiolipin, and anti-beta 2-glycoprotein I antibodies]), treatment with DOACs has been associated with increased rates of recurrent thrombotic events compared with vitamin K antagonist therapy.
-
6 ADVERSE REACTIONS
The following clinically significant adverse reactions are also discussed in other sections of the labeling:
- Increased Risk of Stroke After Discontinuation in Nonvalvular Atrial Fibrillation [see Boxed Warning and Warnings and Precautions (5.1)]
- Bleeding Risk [see Warnings and Precautions (5.2, 5.4, 5.5, 5.6, 5.7)]
- Spinal/Epidural Hematoma [see Boxed Warning and Warnings and Precautions (5.3)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
During clinical development for the approved indications, 31,691 patients were exposed to XARELTO. These included 7111 patients who received XARELTO 15 mg or 20 mg orally once daily for a mean of 19 months (5558 for 12 months and 2512 for 24 months) to reduce the risk of stroke and systemic embolism in nonvalvular atrial fibrillation (ROCKET AF); 6962 patients who received XARELTO 15 mg orally twice daily for three weeks followed by 20 mg orally once daily to treat DVT or PE (EINSTEIN DVT, EINSTEIN PE), 10 mg or 20 mg orally once daily (EINSTEIN Extension, EINSTEIN CHOICE) to reduce the risk of recurrence of DVT and/or PE; 4487 patients who received XARELTO 10 mg orally once daily for prophylaxis of DVT following hip or knee replacement surgery (RECORD 1–3); 3997 patients who received 10 mg orally once daily for prophylaxis of VTE and VTE-related death in acutely ill medical patients (MAGELLAN) and 9134 patients who received XARELTO 2.5 mg orally twice daily, in combination with aspirin 100 mg once daily, for the reduction in risk of major cardiovascular events in patients with chronic CAD or PAD (COMPASS).
Hemorrhage
The most common adverse reactions with XARELTO were bleeding complications [see Warnings and Precautions (5.2)].
Nonvalvular Atrial Fibrillation
In the ROCKET AF trial, the most frequent adverse reactions associated with permanent drug discontinuation were bleeding events, with incidence rates of 4.3% for XARELTO vs. 3.1% for warfarin. The incidence of discontinuations for non-bleeding adverse events was similar in both treatment groups.
Table 2 shows the number of patients experiencing various types of bleeding events in the ROCKET AF trial.
Table 2: Bleeding Events in ROCKET AF*- On Treatment Plus 2 Days Parameter XARELTO
N=7111
n (%/year)Warfarin
N=7125
n (%/year)XARELTO vs. Warfarin HR
(95% CI)Abbreviations: HR = Hazard Ratio, CI = Confidence interval, CRNM = Clinically Relevant Non-Major. - * Major bleeding events within each subcategory were counted once per patient, but patients may have contributed events to multiple subcategories. These events occurred during treatment or within 2 days of stopping treatment.
- † Defined as clinically overt bleeding associated with a decrease in hemoglobin of ≥2 g/dL, a transfusion of ≥2 units of packed red blood cells or whole blood, bleeding at a critical site, or with a fatal outcome.
- ‡ Intracranial bleeding events included intraparenchymal, intraventricular, subdural, subarachnoid and/or epidural hematoma.
- § Hemorrhagic stroke in this table specifically refers to non-traumatic intraparenchymal and/or intraventricular hematoma in patients on treatment plus 2 days.
- ¶ Gastrointestinal bleeding events included upper GI, lower GI, and rectal bleeding.
- # Fatal bleeding is adjudicated death with the primary cause of death from bleeding.
Major Bleeding† 395 (3.6) 386 (3.5) 1.04 (0.90, 1.20) Intracranial Hemorrhage (ICH) ‡ 55 (0.5) 84 (0.7) 0.67 (0.47, 0.93) Hemorrhagic Stroke§ 36 (0.3) 58 (0.5) 0.63 (0.42, 0.96) Other ICH 19 (0.2) 26 (0.2) 0.74 (0.41, 1.34) Gastrointestinal (GI)¶ 221 (2.0) 140 (1.2) 1.61 (1.30, 1.99) Fatal Bleeding# 27 (0.2) 55 (0.5) 0.50 (0.31, 0.79) ICH 24 (0.2) 42 (0.4) 0.58 (0.35, 0.96) Non-intracranial 3 (0.0) 13 (0.1) 0.23 (0.07, 0.82) Figure 1 shows the risk of major bleeding events across major subgroups.
Figure 1: Risk of Major Bleeding Events by Baseline Characteristics in ROCKET AF – On Treatment Plus 2 Days
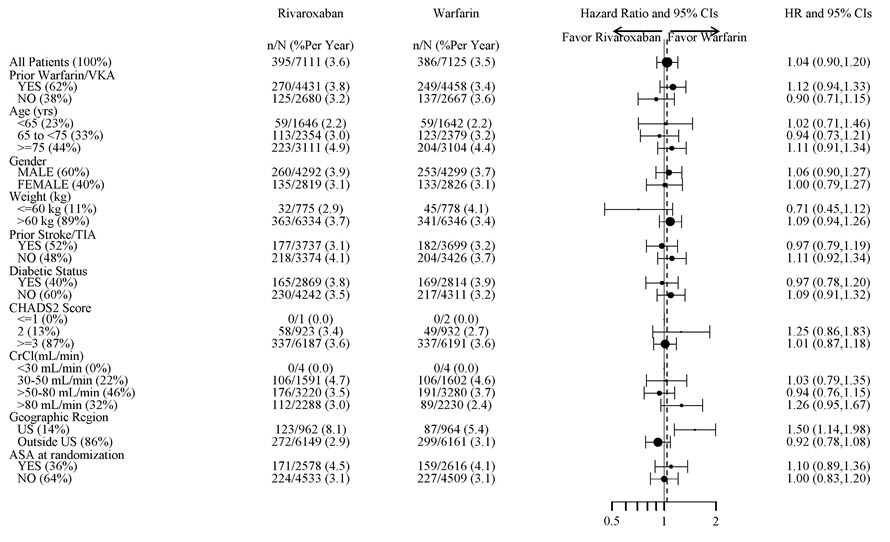
Note: The figure above presents effects in various subgroups all of which are baseline characteristics and all of which were pre-specified (diabetic status was not pre-specified in the subgroup but was a criterion for the CHADS2 score). The 95% confidence limits that are shown do not take into account how many comparisons were made, nor do they reflect the effect of a particular factor after adjustment for all other factors. Apparent homogeneity or heterogeneity among groups should not be over-interpreted.
Treatment of Deep Vein Thrombosis (DVT) and/or Pulmonary Embolism (PE)
EINSTEIN DVT and EINSTEIN PE Studies
In the pooled analysis of the EINSTEIN DVT and EINSTEIN PE clinical studies, the most frequent adverse reactions leading to permanent drug discontinuation were bleeding events, with XARELTO vs. enoxaparin/Vitamin K antagonist (VKA) incidence rates of 1.7% vs. 1.5%, respectively. The mean duration of treatment was 208 days for XARELTO-treated patients and 204 days for enoxaparin/VKA-treated patients.
Table 3 shows the number of patients experiencing major bleeding events in the pooled analysis of the EINSTEIN DVT and EINSTEIN PE studies.
Table 3: Bleeding Events* in the Pooled Analysis of EINSTEIN DVT and EINSTEIN PE Studies Parameter XARELTO†
N=4130
n (%)Enoxaparin/VKA†
N=4116
n (%)- * Bleeding event occurred after randomization and up to 2 days after the last dose of study drug. Although a patient may have had 2 or more events, the patient is counted only once in a category.
- † Treatment schedule in EINSTEIN DVT and EINSTEIN PE studies: XARELTO 15 mg twice daily for 3 weeks followed by 20 mg once daily; enoxaparin/VKA [enoxaparin: 1 mg/kg twice daily, VKA: individually titrated doses to achieve a target INR of 2.5 (range: 2.0–3.0)]
- ‡ Treatment-emergent major bleeding events with at least >2 subjects in any pooled treatment group
- § Major bleeding which is not fatal or in a critical organ, but resulting in a decrease in Hb ≥ 2 g/dL and/or transfusion of ≥2 units of whole blood or packed red blood cells
Major bleeding event 40 (1.0) 72 (1.7) Fatal bleeding 3 (<0.1) 8 (0.2) Intracranial 2 (<0.1) 4 (<0.1) Non-fatal critical organ bleeding 10 (0.2) 29 (0.7) Intracranial‡ 3 (<0.1) 10 (0.2) Retroperitoneal‡ 1 (<0.1) 8 (0.2) Intraocular‡ 3 (<0.1) 2 (<0.1) Intra-articular‡ 0 4 (<0.1) Non-fatal non-critical organ bleeding§ 27 (0.7) 37 (0.9) Decrease in Hb ≥ 2 g/dL 28 (0.7) 42 (1.0) Transfusion of ≥2 units of whole blood or packed red blood cells 18 (0.4) 25 (0.6) Clinically relevant non-major bleeding 357 (8.6) 357 (8.7) Any bleeding 1169 (28.3) 1153 (28.0) Reduction in the Risk of Recurrence of DVT and/or PE
EINSTEIN CHOICE Study
In the EINSTEIN CHOICE clinical study, the most frequent adverse reactions associated with permanent drug discontinuation were bleeding events, with incidence rates of 1% for XARELTO 10 mg, 2% for XARELTO 20 mg, and 1% for acetylsalicylic acid (aspirin) 100 mg. The mean duration of treatment was 293 days for XARELTO 10 mg-treated patients and 286 days for aspirin 100 mg-treated patients.
Table 4 shows the number of patients experiencing bleeding events in the EINSTEIN CHOICE study.
Table 4: Bleeding Events* in EINSTEIN CHOICE Parameter XARELTO†
10 mg
N=1127
n (%)Acetylsalicylic Acid (aspirin)† 100 mg
N=1131
n (%)- * Bleeding event occurred after the first dose and up to 2 days after the last dose of study drug. Although a patient may have had 2 or more events, the patient is counted only once in a category.
- † Treatment schedule: XARELTO 10 mg once daily or aspirin 100 mg once daily.
- ‡ Major bleeding which is not fatal or in a critical organ, but resulting in a decrease in Hb ≥ 2 g/dL and/or transfusion of ≥ 2 units of whole blood or packed red blood cells.
- § Bleeding which was clinically overt, did not meet the criteria for major bleeding, but was associated with medical intervention, unscheduled contact with a physician, temporary cessation of treatment, discomfort for the patient, or impairment of activities of daily life.
Major bleeding event 5 (0.4) 3 (0.3) Fatal bleeding 0 1 (<0.1) Non-fatal critical organ bleeding 2 (0.2) 1 (<0.1) Non-fatal non-critical organ bleeding‡ 3 (0.3) 1 (<0.1) Clinically relevant non-major (CRNM) bleeding§ 22 (2.0) 20 (1.8) Any bleeding 151 (13.4) 138 (12.2) In the EINSTEIN CHOICE study, there was an increased incidence of bleeding, including major and CRNM bleeding in the XARELTO 20 mg group compared to the XARELTO 10 mg or aspirin 100 mg groups.
Prophylaxis of Deep Vein Thrombosis Following Hip or Knee Replacement Surgery
In the RECORD clinical trials, the overall incidence rate of adverse reactions leading to permanent treatment discontinuation was 3.7% with XARELTO.
The rates of major bleeding events and any bleeding events observed in patients in the RECORD clinical trials are shown in Table 5.
Table 5: Bleeding Events* in Patients Undergoing Hip or Knee Replacement Surgeries (RECORD 1–3) XARELTO 10 mg Enoxaparin† - * Bleeding events occurring any time following the first dose of double-blind study medication (which may have been prior to administration of active drug) until two days after the last dose of double-blind study medication. Patients may have more than one event.
- † Includes the placebo-controlled period for RECORD 2, enoxaparin dosing was 40 mg once daily (RECORD 1–3)
- ‡ Includes major bleeding events
Total treated patients N=4487
n (%)N=4524
n (%)Major bleeding event 14 (0.3) 9 (0.2) Fatal bleeding 1 (<0.1) 0 Bleeding into a critical organ 2 (<0.1) 3 (0.1) Bleeding that required re-operation 7 (0.2) 5 (0.1) Extra-surgical site bleeding requiring transfusion of >2 units of whole blood or packed cells 4 (0.1) 1 (<0.1) Any bleeding event‡ 261 (5.8) 251 (5.6) Hip Surgery Studies N=3281
n (%)N=3298
n (%)Major bleeding event 7 (0.2) 3 (0.1) Fatal bleeding 1 (<0.1) 0 Bleeding into a critical organ 1 (<0.1) 1 (<0.1) Bleeding that required re-operation 2 (0.1) 1 (<0.1) Extra-surgical site bleeding requiring transfusion of >2 units of whole blood or packed cells 3 (0.1) 1 (<0.1) Any bleeding event‡ 201 (6.1) 191 (5.8) Knee Surgery Study N=1206
n (%)N=1226
n (%)Major bleeding event 7 (0.6) 6 (0.5) Fatal bleeding 0 0 Bleeding into a critical organ 1 (0.1) 2 (0.2) Bleeding that required re-operation 5 (0.4) 4 (0.3) Extra-surgical site bleeding requiring transfusion of >2 units of whole blood or packed cells 1 (0.1) 0 Any bleeding event‡ 60 (5.0) 60 (4.9) Following XARELTO treatment, the majority of major bleeding complications (≥60%) occurred during the first week after surgery.
Prophylaxis of Venous Thromboembolism in Acutely Ill Medical Patients at Risk for Thromboembolic Complications Not at High Risk of Bleeding
In the MAGELLAN study, the most frequent adverse reactions associated with permanent drug discontinuation were bleeding events. Cases of pulmonary hemorrhage and pulmonary hemorrhage with bronchiectasis were observed. Patients with bronchiectasis/pulmonary cavitation, active cancer (i.e., undergoing acute, in-hospital cancer treatment), dual antiplatelet therapy or active gastroduodenal ulcer or any bleeding in the previous three months all had an excess of bleeding with XARELTO compared with enoxaparin/placebo and are excluded from all MAGELLAN data presented in Table 6. The incidence of bleeding leading to drug discontinuation was 2.5% for XARELTO vs. 1.4% for enoxaparin/placebo.
Table 6 shows the number of patients experiencing various types of bleeding events in the MAGELLAN study.
Table 6: Bleeding Events in MAGELLAN* Study–Safety Analysis Set - On Treatment Plus 2 Days MAGELLAN Study† XARELTO 10 mg
N=3218
n (%)Enoxaparin 40 mg /placebo
N=3229
n (%)- * Patients at high risk of bleeding (i.e. bronchiectasis/pulmonary cavitation, active cancer, dual antiplatelet therapy or active gastroduodenal ulcer or any bleeding in the previous three months) were excluded.
- † Patients received either XARELTO or placebo once daily for 35 ±4 days starting in hospital and continuing post hospital discharge or received enoxaparin or placebo once daily for 10 ±4 days in the hospital.
- ‡ Defined as clinically overt bleeding associated with a drop in hemoglobin of ≥2 g/dL, a transfusion of ≥2 units of packed red blood cells or whole blood, bleeding at a critical site, or with a fatal outcome.
- § Major bleeding events within each subcategory were counted once per patient, but patients may have contributed events to multiple subcategories. These events occurred during treatment or within 2 days of stopping treatment.
- ¶ Fatal bleeding is adjudicated death with the primary cause of death from bleeding.
Major bleeding‡§ 22 (0.7) 15 (0.5) Critical site bleeding 7 (0.2) 4 (0.1) Fatal bleeding¶ 3 (<0.1) 1 (<0.1) Clinically relevant non-major bleeding events (CRNM) 93 (2.9) 34 (1.1) Reduction of Risk of Major Cardiovascular Events in Patients with Chronic CAD or PAD
In the COMPASS trial, the most frequent adverse reactions associated with permanent drug discontinuation were bleeding events, with incidence rates of 2.7% for XARELTO 2.5 mg twice daily in combination with aspirin 100 mg once daily vs. 1.2% for aspirin 100 mg once daily.
Table 7 shows the number of patients experiencing various types of major bleeding events in the COMPASS trial.
Table 7: Major Bleeding Events* in COMPASS - On Treatment Plus 2 days XARELTO plus aspirin†
N=9134Aspirin alone†
N=9107XARELTO plus aspirin vs. Aspirin alone Parameter n (%/year) n (%/year) HR (95 % CI) CI: confidence interval; HR: hazard ratio; ISTH: International Society on Thrombosis and Hemostasis - * Major bleeding events within each subcategory were counted once per patient, but patients may have contributed events to multiple subcategories. These events occurred during treatment or within 2 days of stopping treatment.
- † Treatment schedule: XARELTO 2.5 mg twice daily plus aspirin 100 mg once daily, or aspirin 100 mg once daily
- ‡ Defined as i) fatal bleeding, or ii) symptomatic bleeding in a critical area or organ, such as intraarticular, intramuscular with compartment syndrome, intraspinal, intracranial, intraocular, respiratory, pericardial, liver, pancreas, retroperitoneal, adrenal gland or kidney; or iii) bleeding into the surgical site requiring reoperation, or iv) bleeding leading to hospitalization.
Modified ISTH Major Bleeding‡ 263 (1.6) 144 (0.9) 1.84 (1.50, 2.26) - Fatal bleeding event 12 (<0.1) 8 (<0.1) 1.51 (0.62, 3.69) Intracranial hemorrhage (ICH) 6 (<0.1) 3 (<0.1) 2.01 (0.50, 8.03) Non-intracranial 6 (<0.1) 5 (<0.1) 1.21 (0.37, 3.96) - Symptomatic bleeding in critical organ (non-fatal) 58 (0.3) 43 (0.3) 1.36 (0.91, 2.01) ICH 23 (0.1) 21 (0.1) 1.09 (0.61, 1.98) Hemorrhagic Stroke 18 (0.1) 13 (<0.1) 1.38 (0.68, 2.82) Other ICH 6 (<0.1) 9 (<0.1) 0.67 (0.24, 1.88) - Bleeding into the surgical site requiring reoperation (non-fatal, not in critical organ) 7 (<0.1) 6 (<0.1) 1.17 (0.39, 3.48) - Bleeding leading to hospitalization (non-fatal, not in critical organ, not requiring reoperation) 188 (1.1) 91 (0.5) 2.08 (1.62, 2.67) Major GI bleeding 117 (0.7) 49 (0.3) 2.40 (1.72, 3.35) Figure 2 shows the risk of modified ISTH major bleeding events across major subgroups.
Figure 2: Risk of Modified ISTH Major Bleeding Events by Baseline Characteristics in COMPASS – On Treatment Plus 2 Days
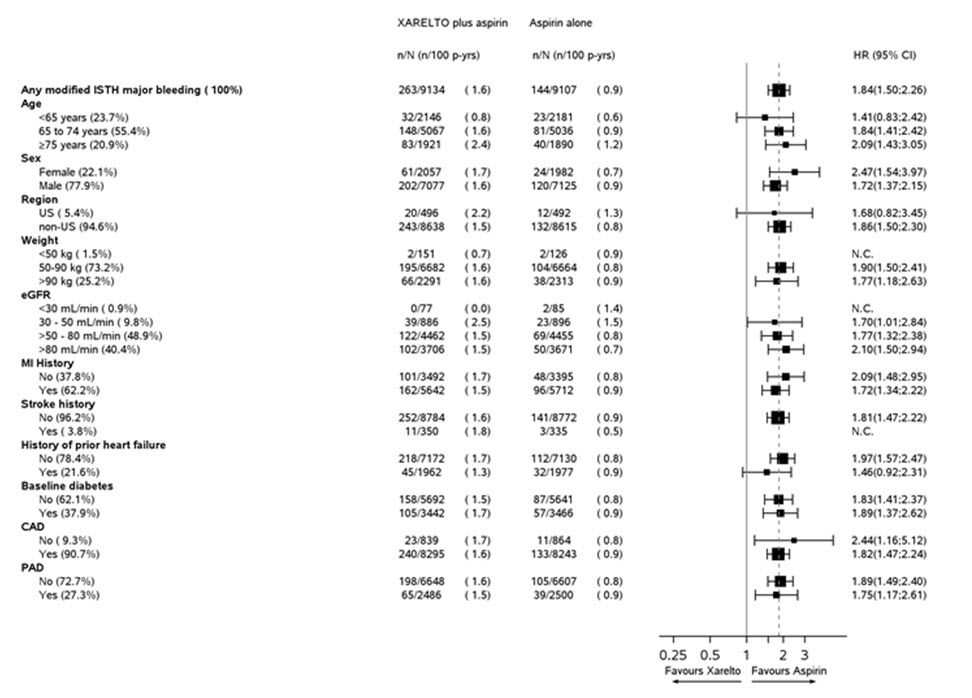
Other Adverse Reactions
Non-hemorrhagic adverse reactions reported in ≥1% of XARELTO-treated patients in the EINSTEIN DVT and EINSTEIN PE studies are shown in Table 8.
Table 8: Other Adverse Reactions* Reported by ≥1% of XARELTO-Treated Patients in EINSTEIN DVT and EINSTEIN PE Studies Body System
Adverse Reaction- * Adverse reaction with Relative Risk >1.5 for XARELTO versus comparator
EINSTEIN DVT Study XARELTO 20 mg
N=1718
n (%)Enoxaparin/VKA
N=1711
n (%)Gastrointestinal disorders Abdominal pain 46 (2.7) 25 (1.5) General disorders and administration site conditions Fatigue 24 (1.4) 15 (0.9) Musculoskeletal and connective tissue disorders Back pain 50 (2.9) 31 (1.8) Muscle spasm 23 (1.3) 13 (0.8) Nervous system disorders Dizziness 38 (2.2) 22 (1.3) Psychiatric disorders Anxiety 24 (1.4) 11 (0.6) Depression 20 (1.2) 10 (0.6) Insomnia 28 (1.6) 18 (1.1) EINSTEIN PE Study XARELTO 20 mg
N=2412
n (%)Enoxaparin/VKA
N=2405
n (%)Skin and subcutaneous tissue disorders Pruritus 53 (2.2) 27 (1.1) Non-hemorrhagic adverse reactions reported in ≥1% of XARELTO-treated patients in RECORD 1–3 studies are shown in Table 9.
Table 9: Other Adverse Drug Reactions* Reported by ≥1% of XARELTO-Treated Patients in RECORD 1–3 Studies Body System
Adverse ReactionXARELTO
10 mg
N=4487
n (%)Enoxaparin†
N=4524
n (%)- * Adverse reaction occurring any time following the first dose of double-blind medication, which may have been prior to administration of active drug, until two days after the last dose of double-blind study medication
- † Includes the placebo-controlled period of RECORD 2, enoxaparin dosing was 40 mg once daily (RECORD 1–3)
Injury, poisoning and procedural complications Wound secretion 125 (2.8) 89 (2.0) Musculoskeletal and connective tissue disorders Pain in extremity 74 (1.7) 55 (1.2) Muscle spasm 52 (1.2) 32 (0.7) Nervous system disorders Syncope 55 (1.2) 32 (0.7) Skin and subcutaneous tissue disorders Pruritus 96 (2.1) 79 (1.8) Blister 63 (1.4) 40 (0.9) 6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of XARELTO. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and lymphatic system disorders: agranulocytosis, thrombocytopenia
Gastrointestinal disorders: retroperitoneal hemorrhage
Hepatobiliary disorders: jaundice, cholestasis, hepatitis (including hepatocellular injury)
Immune system disorders: hypersensitivity, anaphylactic reaction, anaphylactic shock, angioedema
Nervous system disorders: cerebral hemorrhage, subdural hematoma, epidural hematoma, hemiparesis
Skin and subcutaneous tissue disorders: Stevens-Johnson syndrome, drug reaction with eosinophilia and systemic symptoms (DRESS)
-
7 DRUG INTERACTIONS
7.1 General Inhibition and Induction Properties
Rivaroxaban is a substrate of CYP3A4/5, CYP2J2, and the P-gp and ATP-binding cassette G2 (ABCG2) transporters. Combined P-gp and strong CYP3A inhibitors increase exposure to rivaroxaban and may increase the risk of bleeding. Combined P-gp and strong CYP3A inducers decrease exposure to rivaroxaban and may increase the risk of thromboembolic events.
7.2 Drugs that Inhibit Cytochrome P450 3A Enzymes and Drug Transport Systems
Interaction with Combined P-gp and Strong CYP3A Inhibitors
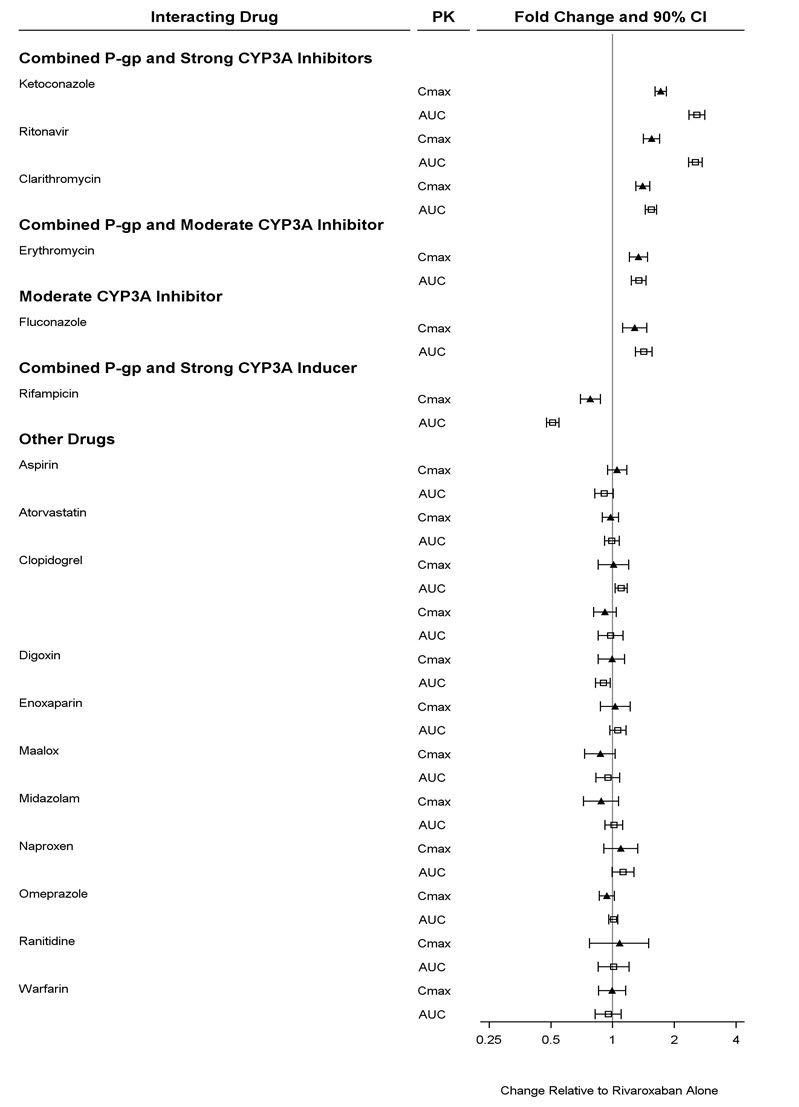
Avoid concomitant administration of XARELTO with known combined P-gp and strong CYP3A inhibitors (e.g., ketoconazole and ritonavir) [see Warnings and Precautions (5.6) and Clinical Pharmacology (12.3)].
Although clarithromycin is a combined P-gp and strong CYP3A inhibitor, pharmacokinetic data suggests that no precautions are necessary with concomitant administration with XARELTO as the change in exposure is unlikely to affect the bleeding risk [see Clinical Pharmacology (12.3)].
Interaction with Combined P-gp and Moderate CYP3A Inhibitors in Patients with Renal Impairment
XARELTO should not be used in patients with CrCl 15 to <80 mL/min who are receiving concomitant combined P-gp and moderate CYP3A inhibitors (e.g., erythromycin) unless the potential benefit justifies the potential risk [see Warnings and Precautions (5.4) and Clinical Pharmacology (12.3)].
7.3 Drugs that Induce Cytochrome P450 3A Enzymes and Drug Transport Systems
Avoid concomitant use of XARELTO with drugs that are combined P-gp and strong CYP3A inducers (e.g., carbamazepine, phenytoin, rifampin, St. John's wort) [see Warnings and Precautions (5.6) and Clinical Pharmacology (12.3)].
7.4 Anticoagulants and NSAIDs/Aspirin
Coadministration of enoxaparin, warfarin, aspirin, clopidogrel and chronic NSAID use may increase the risk of bleeding [see Clinical Pharmacology (12.3)].
Avoid concurrent use of XARELTO with other anticoagulants due to increased bleeding risk unless benefit outweighs risk. Promptly evaluate any signs or symptoms of blood loss if patients are treated concomitantly with aspirin, other platelet aggregation inhibitors, or NSAIDs [see Warnings and Precautions (5.2)].
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
The limited available data on XARELTO in pregnant women are insufficient to inform a drug-associated risk of adverse developmental outcomes. Use XARELTO with caution in pregnant patients because of the potential for pregnancy related hemorrhage and/or emergent delivery. The anticoagulant effect of XARELTO cannot be reliably monitored with standard laboratory testing. Consider the benefits and risks of XARELTO for the mother and possible risks to the fetus when prescribing XARELTO to a pregnant woman [see Warnings and Precautions (5.2, 5.7)].
Adverse outcomes in pregnancy occur regardless of the health of the mother or the use of medications. The estimated background risk of major birth defects and miscarriage for the indicated populations is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2–4% and 15–20%, respectively.
Clinical Considerations
Disease-Associated Maternal and/or Embryo/Fetal Risk
Pregnancy is a risk factor for venous thromboembolism and that risk is increased in women with inherited or acquired thrombophilias. Pregnant women with thromboembolic disease have an increased risk of maternal complications including pre-eclampsia. Maternal thromboembolic disease increases the risk for intrauterine growth restriction, placental abruption and early and late pregnancy loss.
Fetal/Neonatal Adverse Reactions
Based on the pharmacologic activity of Factor Xa inhibitors and the potential to cross the placenta, bleeding may occur at any site in the fetus and/or neonate.
Labor or Delivery
All patients receiving anticoagulants, including pregnant women, are at risk for bleeding and this risk may be increased during labor or delivery [see Warnings and Precautions (5.7)]. The risk of bleeding should be balanced with the risk of thrombotic events when considering the use of XARELTO in this setting.
Data
Human Data
There are no adequate or well-controlled studies of XARELTO in pregnant women, and dosing for pregnant women has not been established. Post-marketing experience is currently insufficient to determine a rivaroxaban-associated risk for major birth defects or miscarriage. In an in vitro placenta perfusion model, unbound rivaroxaban was rapidly transferred across the human placenta.
Animal Data
Rivaroxaban crosses the placenta in animals. Rivaroxaban increased fetal toxicity (increased resorptions, decreased number of live fetuses, and decreased fetal body weight) when pregnant rabbits were given oral doses of ≥10 mg/kg rivaroxaban during the period of organogenesis. This dose corresponds to about 4 times the human exposure of unbound drug, based on AUC comparisons at the highest recommended human dose of 20 mg/day. Fetal body weights decreased when pregnant rats were given oral doses of 120 mg/kg during the period of organogenesis. This dose corresponds to about 14 times the human exposure of unbound drug. In rats, peripartal maternal bleeding and maternal and fetal death occurred at the rivaroxaban dose of 40 mg/kg (about 6 times maximum human exposure of the unbound drug at the human dose of 20 mg/day).
8.2 Lactation
Risk Summary
Rivaroxaban has been detected in human milk. There are insufficient data to determine the effects of rivaroxaban on the breastfed child or on milk production. Rivaroxaban and/or its metabolites were present in the milk of rats. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for XARELTO and any potential adverse effects on the breastfed infant from XARELTO or from the underlying maternal condition (see Data).
Data
Animal Data
Following a single oral administration of 3 mg/kg of radioactive [14C]-rivaroxaban to lactating rats between Day 8 to 10 postpartum, the concentration of total radioactivity was determined in milk samples collected up to 32 hours post-dose. The estimated amount of radioactivity excreted with milk within 32 hours after administration was 2.1% of the maternal dose.
8.3 Females and Males of Reproductive Potential
Females of reproductive potential requiring anticoagulation should discuss pregnancy planning with their physician.
8.5 Geriatric Use
Of the total number of patients in the RECORD 1–3 clinical studies evaluating XARELTO, about 54% were 65 years and over, while about 15% were >75 years. In ROCKET AF, approximately 77% were 65 years and over and about 38% were >75 years. In the EINSTEIN DVT, PE and Extension clinical studies approximately 37% were 65 years and over and about 16% were >75 years. In EINSTEIN CHOICE, approximately 39% were 65 years and over and about 12% were >75 years. In the MAGELLAN study, approximately 67% were 65 years and over and about 37% were >75 years. In the COMPASS study, approximately 76% were 65 years and over and about 17% were >75 years. In clinical trials the efficacy of XARELTO in the elderly (65 years or older) was similar to that seen in patients younger than 65 years. Both thrombotic and bleeding event rates were higher in these older patients [see Clinical Pharmacology (12.3) and Clinical Studies (14)].
8.6 Renal Impairment
In pharmacokinetic studies, compared to healthy subjects with normal creatinine clearance, rivaroxaban exposure increased by approximately 44 to 64% in subjects with renal impairment. Increases in pharmacodynamic effects were also observed [see Clinical Pharmacology (12.3)].
Nonvalvular Atrial Fibrillation
Patients with Chronic Kidney Disease not on Dialysis
In the ROCKET AF trial, patients with CrCl 30 to 50 mL/min were administered XARELTO 15 mg once daily resulting in serum concentrations of rivaroxaban and clinical outcomes similar to those in patients with better renal function administered XARELTO 20 mg once daily. Patients with CrCl <30 mL/min were not studied, but administration of XARELTO 15 mg once daily is expected to result in serum concentrations of rivaroxaban similar to those in patients with moderate renal impairment [see Clinical Pharmacology (12.3)].
Patients with End-Stage Renal Disease on Dialysis
Clinical efficacy and safety studies with XARELTO did not enroll patients with end-stage renal disease (ESRD) on dialysis. In patients with ESRD maintained on intermittent hemodialysis, administration of XARELTO 15 mg once daily will result in concentrations of rivaroxaban and pharmacodynamic activity similar to those observed in the ROCKET AF study [see Clinical Pharmacology (12.2, 12.3)]. It is not known whether these concentrations will lead to similar stroke reduction and bleeding risk in patients with ESRD on dialysis as was seen in ROCKET AF.
Treatment of DVT and/or PE and Reduction in the Risk of Recurrence of DVT and/or PE
In the EINSTEIN trials, patients with CrCl values <30 mL/min at screening were excluded from the studies, but administration of XARELTO is expected to result in serum concentrations of rivaroxaban similar to those in patients with moderate renal impairment (CrCl 30 to <50 mL/min) [see Clinical Pharmacology (12.3)]. Observe closely and promptly evaluate any signs or symptoms of blood loss in patients with CrCl 15 to <30 mL/min. Avoid the use of XARELTO in patients with CrCl <15 mL/min.
Prophylaxis of DVT Following Hip or Knee Replacement Surgery
The combined analysis of the RECORD 1–3 clinical efficacy studies did not show an increase in bleeding risk for patients with CrCl 30 to 50 mL/min and reported a possible increase in total venous thromboemboli in this population. In the RECORD 1–3 trials, patients with CrCl values <30 mL/min at screening were excluded from the studies, but administration of XARELTO 10 mg once daily is expected to result in serum concentrations of rivaroxaban similar to those in patients with moderate renal impairment (CrCl 30 to <50 mL/min) [see Clinical Pharmacology (12.3)]. Observe closely and promptly evaluate any signs or symptoms of blood loss in patients with CrCl 15 to <30 mL/min. Avoid the use of XARELTO in patients with CrCl <15 mL/min.
Prophylaxis of Venous Thromboembolism in Acutely Ill Medical Patients at Risk for Thromboembolic Complications Not at High Risk of Bleeding
Patients with CrCl values <30 mL/min at screening were excluded from the MAGELLAN study. In patients with CrCl <30 mL/min a dose of XARELTO 10 mg once daily is expected to result in serum concentrations of rivaroxaban similar to those in patients with moderate renal impairment (CrCl 30 to <50 mL/min) [see Clinical Pharmacology (12.3)]. Observe closely and promptly evaluate any signs or symptoms of blood loss in patients with CrCl 15 to <30 mL/min. Avoid use of XARELTO in patients with CrCl <15 mL/min.
Reduction of Risk of Major Cardiovascular Events in Patients with Chronic CAD or PAD
Patients with Chronic Kidney Disease not on Dialysis
Patients with a CrCl <15 mL/min at screening were excluded from COMPASS, and limited data are available for patients with a CrCl of 15 to 30 mL/min. In patients with CrCl <30 mL/min, a dose of 2.5 mg XARELTO twice daily is expected to give an exposure similar to that in patients with moderate renal impairment (CrCl 30 to <50 mL/min) [see Clinical Pharmacology (12.3)], whose efficacy and safety outcomes were similar to those with preserved renal function.
Patients with End-Stage Renal Disease on Dialysis
No clinical outcome data is available for the use of XARELTO with aspirin in patients with ESRD on dialysis since these patients were not enrolled in COMPASS. In patients with ESRD maintained on intermittent hemodialysis, administration of XARELTO 2.5 mg twice daily will result in concentrations of rivaroxaban and pharmacodynamic activity similar to those observed in moderate renal impaired patients in the COMPASS study [see Clinical Pharmacology (12.2, 12.3)]. It is not known whether these concentrations will lead to similar CV risk reduction and bleeding risk in patients with ESRD on dialysis as was seen in COMPASS.
8.7 Hepatic Impairment
In a pharmacokinetic study, compared to healthy subjects with normal liver function, AUC increases of 127% were observed in subjects with moderate hepatic impairment (Child-Pugh B).
The safety or PK of XARELTO in patients with severe hepatic impairment (Child-Pugh C) has not been evaluated [see Clinical Pharmacology (12.3)].
Avoid the use of XARELTO in patients with moderate (Child-Pugh B) and severe (Child-Pugh C) hepatic impairment or with any hepatic disease associated with coagulopathy.
-
10 OVERDOSAGE
Overdose of XARELTO may lead to hemorrhage. Discontinue XARELTO and initiate appropriate therapy if bleeding complications associated with overdosage occur. Rivaroxaban systemic exposure is not further increased at single doses >50 mg due to limited absorption. The use of activated charcoal to reduce absorption in case of XARELTO overdose may be considered. Due to the high plasma protein binding, rivaroxaban is not dialyzable [see Warnings and Precautions (5.2) and Clinical Pharmacology (12.3)]. Partial reversal of laboratory anticoagulation parameters may be achieved with use of plasma products. An agent to reverse the anti-factor Xa activity of rivaroxaban is available.
-
11 DESCRIPTION
Rivaroxaban, a factor Xa (FXa) inhibitor, is the active ingredient in XARELTO Tablets with the chemical name 5-Chloro-N-({(5S)-2-oxo-3-[4-(3-oxo-4-morpholinyl)phenyl]-1,3-oxazolidin-5-yl}methyl)-2-thiophenecarboxamide. The molecular formula of rivaroxaban is C19H18ClN3O5S and the molecular weight is 435.89. The structural formula is:
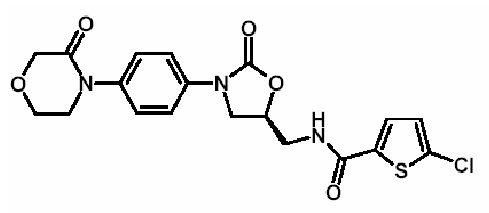
Rivaroxaban is a pure (S)-enantiomer. It is an odorless, non-hygroscopic, white to yellowish powder. Rivaroxaban is only slightly soluble in organic solvents (e.g., acetone, polyethylene glycol 400) and is practically insoluble in water and aqueous media.
Each XARELTO tablet contains 2.5 mg, 10 mg, 15 mg, or 20 mg of rivaroxaban. The inactive ingredients of XARELTO are: croscarmellose sodium, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, and sodium lauryl sulfate. Additionally, the proprietary film coating mixture used for XARELTO 2.5 mg is Opadry® Light Yellow, containing ferric oxide yellow, hypromellose, polyethylene glycol 3350, and titanium dioxide, and for XARELTO 10 mg tablets is Opadry® Pink and for XARELTO 15 mg tablets is Opadry® Red, both containing ferric oxide red, hypromellose, polyethylene glycol 3350, and titanium dioxide, and for XARELTO 20 mg tablets is Opadry® II Dark Red, containing ferric oxide red, polyethylene glycol 3350, polyvinyl alcohol (partially hydrolyzed), talc, and titanium dioxide.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
XARELTO is a selective inhibitor of FXa. It does not require a cofactor (such as Anti-thrombin III) for activity. Rivaroxaban inhibits free FXa and prothrombinase activity. Rivaroxaban has no direct effect on platelet aggregation, but indirectly inhibits platelet aggregation induced by thrombin. By inhibiting FXa, rivaroxaban decreases thrombin generation.
12.2 Pharmacodynamics
Dose-dependent inhibition of FXa activity was observed in humans. Neoplastin® prothrombin time (PT), activated partial thromboplastin time (aPTT) and HepTest® are also prolonged dose-dependently. Anti-factor Xa activity is also influenced by rivaroxaban.
Specific Populations
Renal Impairment
The relationship between systemic exposure and pharmacodynamic activity of rivaroxaban was altered in subjects with renal impairment relative to healthy control subjects [see Use in Specific Populations (8.6)].
Table 10: Percentage Increase in Rivaroxaban PK and PD Measures in Subjects with Renal Impairment Relative to Healthy Subjects from Clinical Pharmacology Studies Creatinine Clearance (mL/min) Measure Parameter 50–79 30–49 15–29 ESRD (on dialysis)* ESRD (post-dialysis)* PT = Prothrombin time; FXa = Coagulation factor Xa; AUC = Area under the plasma concentration-time curve; AUEC = Area under the effect-time curve - * Separate stand-alone study.
Exposure AUC 44 52 64 47 56 FXa Inhibition AUEC 50 86 100 49 33 PT Prolongation AUEC 33 116 144 112 158 Hepatic Impairment
Anti-Factor Xa activity was similar in subjects with normal hepatic function and in mild hepatic impairment (Child-Pugh A class). There is no clear understanding of the impact of hepatic impairment beyond this degree on the coagulation cascade and its relationship to efficacy and safety.
12.3 Pharmacokinetics
Absorption
The absolute bioavailability of rivaroxaban is dose-dependent. For the 2.5 mg and 10 mg dose, it is estimated to be 80% to 100% and is not affected by food. XARELTO 2.5 mg and 10 mg tablets can be taken with or without food. For the 20 mg dose in the fasted state, the absolute bioavailability is approximately 66%. Coadministration of XARELTO with food increases the bioavailability of the 20 mg dose (mean AUC and Cmax increasing by 39% and 76% respectively with food). XARELTO 15 mg and 20 mg tablets should be taken with food [see Dosage and Administration (2.1)].
The maximum concentrations (Cmax) of rivaroxaban appear 2 to 4 hours after tablet intake. The pharmacokinetics of rivaroxaban were not affected by drugs altering gastric pH. Coadministration of XARELTO (30 mg single dose) with the H2-receptor antagonist ranitidine (150 mg twice daily), the antacid aluminum hydroxide/magnesium hydroxide (10 mL) or XARELTO (20 mg single dose) with the PPI omeprazole (40 mg once daily) did not show an effect on the bioavailability and exposure of rivaroxaban (see Figure 4).
Absorption of rivaroxaban is dependent on the site of drug release in the GI tract. A 29% and 56% decrease in AUC and Cmax compared to tablet was reported when rivaroxaban granulate is released in the proximal small intestine. Exposure is further reduced when drug is released in the distal small intestine, or ascending colon. Avoid administration of rivaroxaban distal to the stomach which can result in reduced absorption and related drug exposure.
In a study with 44 healthy subjects, both mean AUC and Cmax values for 20 mg rivaroxaban administered orally as a crushed tablet mixed in applesauce were comparable to that after the whole tablet. However, for the crushed tablet suspended in water and administered via an NG tube followed by a liquid meal, only mean AUC was comparable to that after the whole tablet, and Cmax was 18% lower.
Distribution
Plasma protein binding of rivaroxaban in human plasma is approximately 92% to 95%, with albumin being the main binding component. The steady-state volume of distribution in healthy subjects is approximately 50 L.
Metabolism
Approximately 51% of an orally administered [14C]-rivaroxaban dose was recovered as inactive metabolites in urine (30%) and feces (21%). Oxidative degradation catalyzed by CYP3A4/5 and CYP2J2 and hydrolysis are the major sites of biotransformation. Unchanged rivaroxaban was the predominant moiety in plasma with no major or active circulating metabolites.
Excretion
In a Phase 1 study, following the administration of [14C]-rivaroxaban, approximately one-third (36%) was recovered as unchanged drug in the urine and 7% was recovered as unchanged drug in feces. Unchanged drug is excreted into urine, mainly via active tubular secretion and to a lesser extent via glomerular filtration (approximate 5:1 ratio). Rivaroxaban is a substrate of the efflux transporter proteins P-gp and ABCG2 (also abbreviated Bcrp). Rivaroxaban's affinity for influx transporter proteins is unknown.
Rivaroxaban is a low-clearance drug, with a systemic clearance of approximately 10 L/hr in healthy volunteers following intravenous administration. The terminal elimination half-life of rivaroxaban is 5 to 9 hours in healthy subjects aged 20 to 45 years.
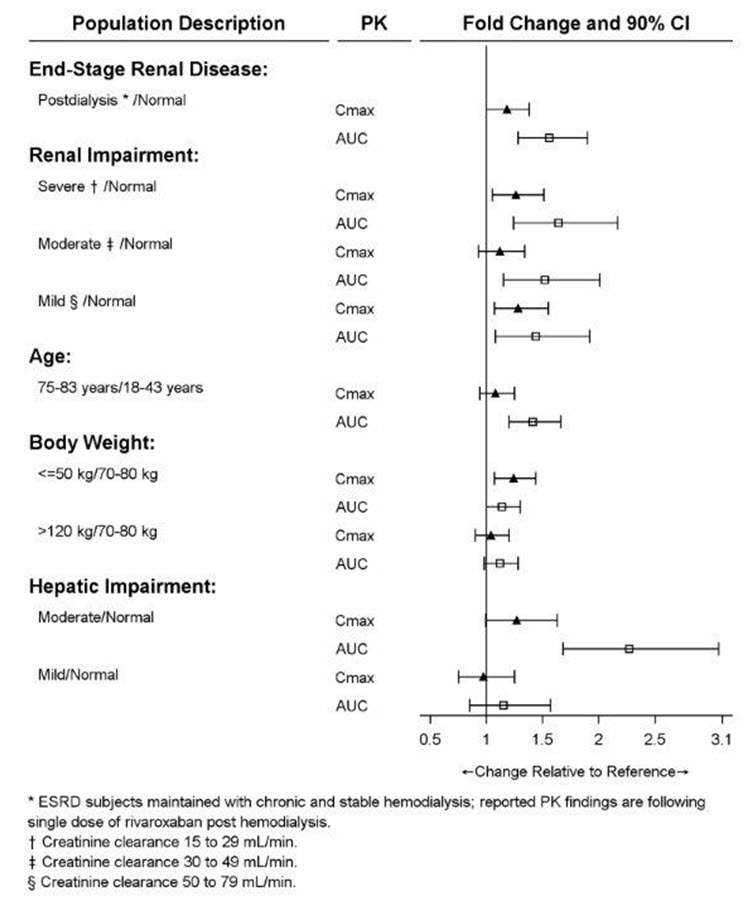
Specific Populations
The effects of level of renal impairment, age, body weight, and level of hepatic impairment on the pharmacokinetics of rivaroxaban are summarized in Figure 3.
Figure 3: Effect of Specific Populations on the Pharmacokinetics of Rivaroxaban
[see Dosage and Administration (2.1)].
Race
Healthy Japanese subjects were found to have 20 to 40% on average higher exposures compared to other ethnicities including Chinese. However, these differences in exposure are reduced when values are corrected for body weight.
Elderly
The terminal elimination half-life is 11 to 13 hours in the elderly subjects aged 60 to 76 years [see Use in Specific Populations (8.5)].
Renal Impairment
The safety and pharmacokinetics of single-dose XARELTO (10 mg) were evaluated in a study in healthy subjects [CrCl ≥80 mL/min (n=8)] and in subjects with varying degrees of renal impairment (see Figure 3). Compared to healthy subjects with normal creatinine clearance, rivaroxaban exposure increased in subjects with renal impairment. Increases in pharmacodynamic effects were also observed [see Use in Specific Populations (8.6)].
Hemodialysis in ESRD subjects: Systemic exposure to rivaroxaban administered as a single 15 mg dose in ESRD subjects dosed 3 hours after the completion of a 4-hour hemodialysis session (post-dialysis) is 56% higher when compared to subjects with normal renal function (see Table 10). The systemic exposure to rivaroxaban administered 2 hours prior to a 4-hour hemodialysis session with a dialysate flow rate of 600 mL/min and a blood flow rate in the range of 320 to 400 mL/min is 47% higher compared to those with normal renal function. The extent of the increase is similar to the increase in patients with CrCl 15 to 50 mL/min taking XARELTO 15 mg. Hemodialysis had no significant impact on rivaroxaban exposure. Protein binding was similar (86% to 89%) in healthy controls and ESRD subjects in this study.
Hepatic Impairment
The safety and pharmacokinetics of single-dose XARELTO (10 mg) were evaluated in a study in healthy subjects (n=16) and subjects with varying degrees of hepatic impairment (see Figure 3). No patients with severe hepatic impairment (Child-Pugh C) were studied. Compared to healthy subjects with normal liver function, significant increases in rivaroxaban exposure were observed in subjects with moderate hepatic impairment (Child-Pugh B) (see Figure 3). Increases in pharmacodynamic effects were also observed [see Use in Specific Populations (8.7)].
Drug Interactions
In vitro studies indicate that rivaroxaban neither inhibits the major cytochrome P450 enzymes CYP1A2, 2C8, 2C9, 2C19, 2D6, 2J2, and 3A nor induces CYP1A2, 2B6, 2C19, or 3A. In vitro data also indicates a low rivaroxaban inhibitory potential for P-gp and ABCG2 transporters.
The effects of coadministered drugs on the pharmacokinetics of rivaroxaban exposure are summarized in Figure 4 [see Drug Interactions (7)].
Figure 4: Effect of Coadministered Drugs on the Pharmacokinetics of Rivaroxaban
Anticoagulants
In a drug interaction study, single doses of enoxaparin (40 mg subcutaneous) and XARELTO (10 mg) given concomitantly resulted in an additive effect on anti-factor Xa activity. In another study, single doses of warfarin (15 mg) and XARELTO (5 mg) resulted in an additive effect on factor Xa inhibition and PT. Neither enoxaparin nor warfarin affected the pharmacokinetics of rivaroxaban (see Figure 4).
NSAIDs/Aspirin
In ROCKET AF, concomitant aspirin use (almost exclusively at a dose of 100 mg or less) during the double-blind phase was identified as an independent risk factor for major bleeding. NSAIDs are known to increase bleeding, and bleeding risk may be increased when NSAIDs are used concomitantly with XARELTO. Neither naproxen nor aspirin affected the pharmacokinetics of rivaroxaban (see Figure 4).
Clopidogrel
In two drug interaction studies where clopidogrel (300 mg loading dose followed by 75 mg daily maintenance dose) and XARELTO (15 mg single dose) were coadministered in healthy subjects, an increase in bleeding time to 45 minutes was observed in approximately 45% and 30% of subjects in these studies, respectively. The change in bleeding time was approximately twice the maximum increase seen with either drug alone. There was no change in the pharmacokinetics of either drug.
Drug-Disease Interactions with Drugs that Inhibit Cytochrome P450 3A Enzymes and Drug Transport Systems
In a pharmacokinetic trial, XARELTO was administered as a single dose in subjects with mild (CrCl = 50 to 79 mL/min) or moderate renal impairment (CrCl = 30 to 49 mL/min) receiving multiple doses of erythromycin (a combined P-gp and moderate CYP3A inhibitor). Compared to XARELTO administered alone in subjects with normal renal function (CrCl >80 mL/min), subjects with mild and moderate renal impairment concomitantly receiving erythromycin reported a 76% and 99% increase in AUCinf and a 56% and 64% increase in Cmax, respectively. Similar trends in pharmacodynamic effects were also observed.
-
13 NON-CLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Rivaroxaban was not carcinogenic when administered by oral gavage to mice or rats for up to 2 years. The systemic exposures (AUCs) of unbound rivaroxaban in male and female mice at the highest dose tested (60 mg/kg/day) were 1- and 2-times, respectively, the human exposure of unbound drug at the human dose of 20 mg/day. Systemic exposures of unbound drug in male and female rats at the highest dose tested (60 mg/kg/day) were 2- and 4-times, respectively, the human exposure.
Rivaroxaban was not mutagenic in bacteria (Ames-Test) or clastogenic in V79 Chinese hamster lung cells in vitro or in the mouse micronucleus test in vivo.
No impairment of fertility was observed in male or female rats when given up to 200 mg/kg/day of rivaroxaban orally. This dose resulted in exposure levels, based on the unbound AUC, at least 13 times the exposure in humans given 20 mg rivaroxaban daily.
-
14 CLINICAL STUDIES
14.1 Stroke Prevention in Nonvalvular Atrial Fibrillation
The evidence for the efficacy and safety of XARELTO was derived from Rivaroxaban Once-daily oral direct factor Xa inhibition Compared with vitamin K antagonist for the prevention of stroke and Embolism Trial in Atrial Fibrillation (ROCKET AF) [NCT00403767], a multi-national, double-blind study comparing XARELTO (at a dose of 20 mg once daily with the evening meal in patients with CrCl >50 mL/min and 15 mg once daily with the evening meal in patients with CrCl 30 to 50 mL/min) to warfarin (titrated to INR 2.0 to 3.0) to reduce the risk of stroke and non-central nervous system (CNS) systemic embolism in patients with nonvalvular atrial fibrillation (AF). Patients had to have one or more of the following additional risk factors for stroke:
- a prior stroke (ischemic or unknown type), transient ischemic attack (TIA) or non-CNS systemic embolism, or
- 2 or more of the following risk factors:
- age ≥75 years,
- hypertension,
- heart failure or left ventricular ejection fraction ≤35%, or
- diabetes mellitus
ROCKET AF was a non-inferiority study designed to demonstrate that XARELTO preserved more than 50% of warfarin's effect on stroke and non-CNS systemic embolism as established by previous placebo-controlled studies of warfarin in atrial fibrillation.
A total of 14264 patients were randomized and followed on study treatment for a median of 590 days. The mean age was 71 years and the mean CHADS2 score was 3.5. The population was 60% male, 83% Caucasian, 13% Asian and 1.3% Black. There was a history of stroke, TIA, or non-CNS systemic embolism in 55% of patients, and 38% of patients had not taken a vitamin K antagonist (VKA) within 6 weeks at time of screening. Concomitant diseases of patients in this study included hypertension 91%, diabetes 40%, congestive heart failure 63%, and prior myocardial infarction 17%. At baseline, 37% of patients were on aspirin (almost exclusively at a dose of 100 mg or less) and few patients were on clopidogrel. Patients were enrolled in Eastern Europe (39%); North America (19%); Asia, Australia, and New Zealand (15%); Western Europe (15%); and Latin America (13%). Patients randomized to warfarin had a mean percentage of time in the INR target range of 2.0 to 3.0 of 55%, lower during the first few months of the study.
In ROCKET AF, XARELTO was demonstrated non-inferior to warfarin for the primary composite endpoint of time to first occurrence of stroke (any type) or non-CNS systemic embolism [HR (95% CI): 0.88 (0.74, 1.03)], but superiority to warfarin was not demonstrated. There is insufficient experience to determine how XARELTO and warfarin compare when warfarin therapy is well-controlled.
Table 11 displays the overall results for the primary composite endpoint and its components.
Table 11: Primary Composite Endpoint Results in ROCKET AF Study (Intent-to-Treat Population) XARELTO Warfarin XARELTO vs. Warfarin Event N=7081
n (%)Event Rate
(per 100 Pt-yrs)N=7090
n (%)Event Rate
(per 100 Pt-yrs)Hazard Ratio
(95% CI)- * The primary endpoint was the time to first occurrence of stroke (any type) or non-CNS systemic embolism. Data are shown for all randomized patients followed to site notification that the study would end.
- † Defined as primary hemorrhagic strokes confirmed by adjudication in all randomized patients followed up to site notification
Primary Composite Endpoint* 269 (3.8) 2.1 306 (4.3) 2.4 0.88 (0.74, 1.03) Stroke 253 (3.6) 2.0 281 (4.0) 2.2 Hemorrhagic Stroke† 33 (0.5) 0.3 57 (0.8) 0.4 Ischemic Stroke 206 (2.9) 1.6 208 (2.9) 1.6 Unknown Stroke Type 19 (0.3) 0.2 18 (0.3) 0.1 Non-CNS Systemic Embolism 20 (0.3) 0.2 27 (0.4) 0.2 Figure 5 is a plot of the time from randomization to the occurrence of the first primary endpoint event in the two treatment arms.
Figure 5: Time to First Occurrence of Stroke (any type) or Non-CNS Systemic Embolism by Treatment Group (Intent-to-Treat Population)
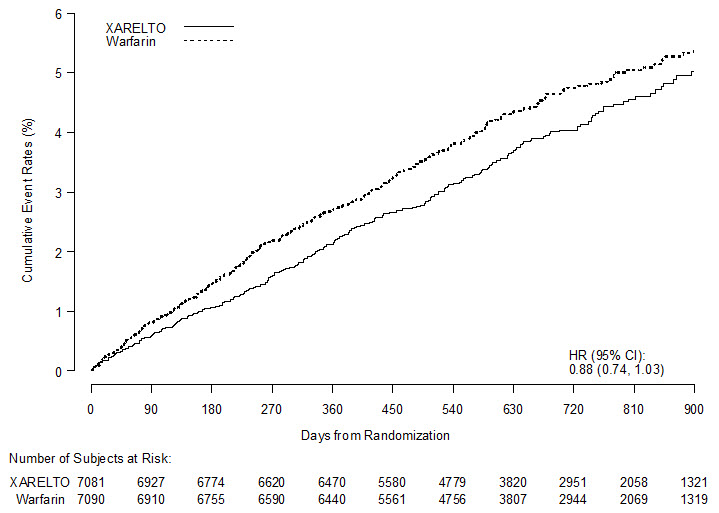
Figure 6 shows the risk of stroke or non-CNS systemic embolism across major subgroups.
- * Data are shown for all randomized patients followed to site notification that the study would end.
Note: The figure above presents effects in various subgroups all of which are baseline characteristics and all of which were pre-specified (diabetic status was not pre-specified in the subgroup, but was a criterion for the CHADS2 score). The 95% confidence limits that are shown do not take into account how many comparisons were made, nor do they reflect the effect of a particular factor after adjustment for all other factors. Apparent homogeneity or heterogeneity among groups should not be over-interpreted.Figure 6: Risk of Stroke or Non-CNS Systemic Embolism by Baseline Characteristics in ROCKET AF* (Intent-to-Treat Population) 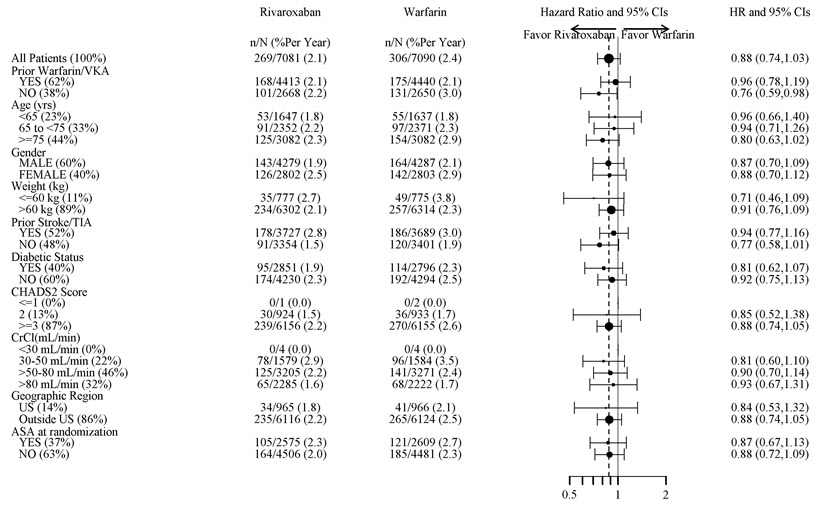
The efficacy of XARELTO was generally consistent across major subgroups.
The protocol for ROCKET AF did not stipulate anticoagulation after study drug discontinuation, but warfarin patients who completed the study were generally maintained on warfarin. XARELTO patients were generally switched to warfarin without a period of coadministration of warfarin and XARELTO, so that they were not adequately anticoagulated after stopping XARELTO until attaining a therapeutic INR. During the 28 days following the end of the study, there were 22 strokes in the 4637 patients taking XARELTO vs. 6 in the 4691 patients taking warfarin.
Few patients in ROCKET AF underwent electrical cardioversion for atrial fibrillation. The utility of XARELTO for preventing post-cardioversion stroke and systemic embolism is unknown.
14.2 Treatment of Deep Vein Thrombosis (DVT) and/or Pulmonary Embolism (PE)
EINSTEIN Deep Vein Thrombosis and EINSTEIN Pulmonary Embolism Studies
XARELTO for the treatment of DVT and/or PE was studied in EINSTEIN DVT [NCT00440193] and EINSTEIN PE [NCT00439777], multi-national, open-label, non-inferiority studies comparing XARELTO (at an initial dose of 15 mg twice daily with food for the first three weeks, followed by XARELTO 20 mg once daily with food) to enoxaparin 1 mg/kg twice daily for at least five days with VKA and then continued with VKA only after the target INR (2.0–3.0) was reached. Patients who required thrombectomy, insertion of a caval filter, or use of a fibrinolytic agent and patients with creatinine clearance <30 mL/min, significant liver disease, or active bleeding were excluded from the studies. The intended treatment duration was 3, 6, or 12 months based on investigator's assessment prior to randomization.
A total of 8281 (3449 in EINSTEIN DVT and 4832 in EINSTEIN PE) patients were randomized and followed on study treatment for a mean of 208 days in the XARELTO group and 204 days in the enoxaparin/VKA group. The mean age was approximately 57 years. The population was 55% male, 70% Caucasian, 9% Asian and about 3% Black. About 73% and 92% of XARELTO-treated patients in the EINSTEIN DVT and EINSTEIN PE studies, respectively, received initial parenteral anticoagulant treatment for a median duration of 2 days. Enoxaparin/VKA-treated patients in the EINSTEIN DVT and EINSTEIN PE studies received initial parenteral anticoagulant treatment for a median duration of 8 days. Aspirin was taken as on treatment concomitant antithrombotic medication by approximately 12% of patients in both treatment groups. Patients randomized to VKA had an unadjusted mean percentage of time in the INR target range of 2.0 to 3.0 of 58% in EINSTEIN DVT study and 60% in EINSTEIN PE study, with the lower values occurring during the first month of the study.
In the EINSTEIN DVT and EINSTEIN PE studies, 49% of patients had an idiopathic DVT/PE at baseline. Other risk factors included previous episode of DVT/PE (19%), recent surgery or trauma (18%), immobilization (16%), use of estrogen-containing drug (8%), known thrombophilic conditions (6%), or active cancer (5%).
In the EINSTEIN DVT and EINSTEIN PE studies, XARELTO was demonstrated to be non-inferior to enoxaparin/VKA for the primary composite endpoint of time to first occurrence of recurrent DVT or non-fatal or fatal PE [EINSTEIN DVT HR (95% CI): 0.68 (0.44, 1.04); EINSTEIN PE HR (95% CI): 1.12 (0.75, 1.68)]. In each study the conclusion of non-inferiority was based on the upper limit of the 95% confidence interval for the hazard ratio being less than 2.0.
Table 12 displays the overall results for the primary composite endpoint and its components for EINSTEIN DVT and EINSTEIN PE studies.
Table 12: Primary Composite Endpoint Results* in EINSTEIN DVT and EINSTEIN PE Studies – Intent-to-Treat Population Event XARELTO 20 mg† Enoxaparin/VKA† XARELTO vs. Enoxaparin/VKA
Hazard Ratio
(95% CI)- * For the primary efficacy analysis, all confirmed events were considered from randomization up to the end of intended treatment duration (3, 6 or 12 months) irrespective of the actual treatment duration. If the same patient had several events, the patient may have been counted for several components.
- † Treatment schedule in EINSTEIN DVT and EINSTEIN PE studies: XARELTO 15 mg twice daily for 3 weeks followed by 20 mg once daily; enoxaparin/VKA [enoxaparin: 1 mg/kg twice daily, VKA: individually titrated doses to achieve a target INR of 2.5 (range: 2.0–3.0)]
EINSTEIN DVT Study N=1731
n (%)N=1718
n (%)Primary Composite Endpoint 36 (2.1) 51 (3.0) 0.68 (0.44, 1.04) Death (PE) 1 (<0.1) 0 Death (PE cannot be excluded) 3 (0.2) 6 (0.3) Symptomatic PE and DVT 1 (<0.1) 0 Symptomatic recurrent PE only 20 (1.2) 18 (1.0) Symptomatic recurrent DVT only 14 (0.8) 28 (1.6) EINSTEIN PE Study N=2419
n (%)N=2413
n (%)Primary Composite Endpoint 50 (2.1) 44 (1.8) 1.12 (0.75, 1.68) Death (PE) 3 (0.1) 1 (<0.1) Death (PE cannot be excluded) 8 (0.3) 6 (0.2) Symptomatic PE and DVT 0 2 (<0.1) Symptomatic recurrent PE only 23 (1.0) 20 (0.8) Symptomatic recurrent DVT only 18 (0.7) 17 (0.7) Figures 7 and 8 are plots of the time from randomization to the occurrence of the first primary efficacy endpoint event in the two treatment groups in EINSTEIN DVT and EINSTEIN PE studies, respectively.
Figure 7: Time to First Occurrence of the Composite of Recurrent DVT or Non-fatal or Fatal PE by Treatment Group (Intent-to-Treat Population) – EINSTEIN DVT Study
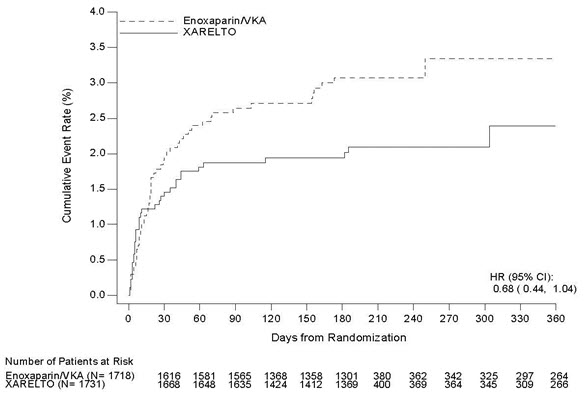
Figure 8: Time to First Occurrence of the Composite of Recurrent DVT or Non-fatal or Fatal PE by Treatment Group (Intent-to-Treat Population) – EINSTEIN PE Study
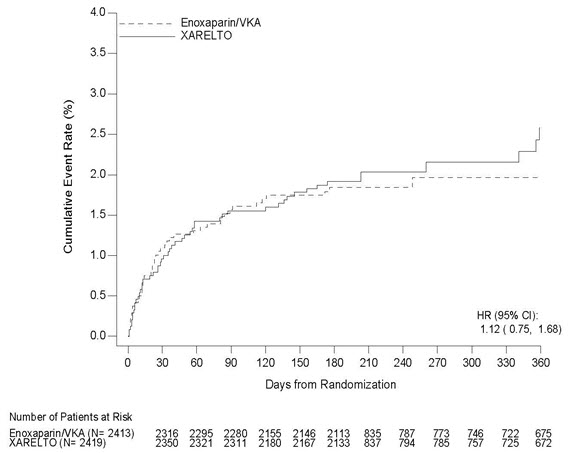
14.3 Reduction in the Risk of Recurrence of DVT and/or PE
EINSTEIN CHOICE Study
XARELTO for reduction in the risk of recurrence of DVT and of PE was evaluated in the EINSTEIN CHOICE study [NCT02064439], a multi-national, double-blind, superiority study comparing XARELTO (10 or 20 mg once daily with food) to 100 mg acetylsalicylic acid (aspirin) once daily in patients who had completed 6 to 12 months of anticoagulant treatment for DVT and/or PE following the acute event. The intended treatment duration in the study was up to 12 months. Patients with an indication for continued therapeutic-dose anticoagulation were excluded.
Because the benefit-risk assessment favored the 10 mg dose versus aspirin compared to the 20 mg dose versus aspirin, only the data concerning the 10 mg dose is discussed below.
A total of 2275 patients were randomized and followed on study treatment for a mean of 290 days for the XARELTO and aspirin treatment groups. The mean age was approximately 59 years. The population was 56% male, 70% Caucasian, 14% Asian and 3% Black. In the EINSTEIN CHOICE study, 51% of patients had DVT only, 33% had PE only, and 16% had PE and DVT combined. Other risk factors included idiopathic VTE (43%), previous episode of DVT/PE (17%), recent surgery or trauma (12%), prolonged immobilization (10%), use of estrogen containing drugs (5%), known thrombophilic conditions (6%), Factor V Leiden gene mutation (4%), or active cancer (3%).
In the EINSTEIN CHOICE study, XARELTO 10 mg was demonstrated to be superior to aspirin 100 mg for the primary composite endpoint of time to first occurrence of recurrent DVT or non-fatal or fatal PE.
Table 13 displays the overall results for the primary composite endpoint and its components.
Table 13: Primary Composite Endpoint and its Components Results* in EINSTEIN CHOICE Study – Full Analysis Set Event XARELTO
10 mg
N=1,127
n (%)Acetylsalicylic Acid (Aspirin)
100 mg
N=1,131
n (%)XARELTO 10 mg vs. Aspirin
100 mg
Hazard Ratio
(95% CI)- * For the primary efficacy analysis, all confirmed events were considered from randomization up to the end of intended treatment duration (12 months) irrespective of the actual treatment duration. The individual component of the primary endpoint represents the first occurrence of the event.
Primary Composite Endpoint 13 (1.2) 50 (4.4) 0.26
(0.14, 0.47)
p<0.0001Symptomatic recurrent DVT 8 (0.7) 29 (2.6) Symptomatic recurrent PE 5 (0.4) 19 (1.7) Death (PE) 0 1 (<0.1) Death (PE cannot be excluded) 0 1 (<0.1) Figure 9 is a plot of the time from randomization to the occurrence of the first primary efficacy endpoint event in the two treatment groups.
Figure 9: Time to First Occurrence of the Composite of Recurrent DVT or Non-fatal or Fatal PE by Treatment Group (Full Analysis Set) – EINSTEIN CHOICE Study
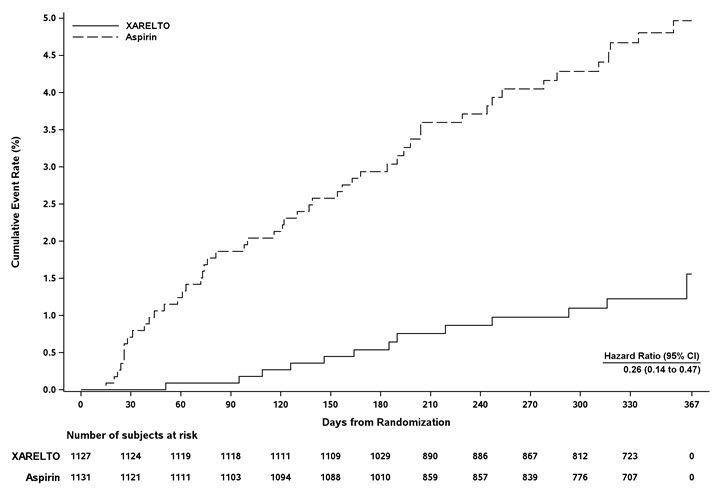
14.4 Prophylaxis of Deep Vein Thrombosis Following Hip or Knee Replacement Surgery
XARELTO was studied in 9011 patients (4487 XARELTO-treated, 4524 enoxaparin-treated patients) in the REgulation of Coagulation in ORthopedic Surgery to Prevent DVT and PE, Controlled, Double-blind, Randomized Study of BAY 59-7939 in the Extended Prevention of VTE in Patients Undergoing Elective Total Hip or Knee Replacement (RECORD 1, 2, and 3) [NCT00329628, NCT0033, NCT00361894] studies.
The two randomized, double-blind, clinical studies (RECORD 1 and 2) in patients undergoing elective total hip replacement surgery compared XARELTO 10 mg once daily starting at least 6 to 8 hours (about 90% of patients dosed 6 to 10 hours) after wound closure versus enoxaparin 40 mg once daily started 12 hours preoperatively. In RECORD 1 and 2, a total of 6727 patients were randomized and 6579 received study drug. The mean age [± standard deviation (SD)] was 63 ± 12.2 (range 18 to 93) years with 49% of patients ≥65 years and 55% of patients were female. More than 82% of patients were White, 7% were Asian, and less than 2% were Black. The studies excluded patients undergoing staged bilateral total hip replacement, patients with severe renal impairment defined as an estimated creatinine clearance <30 mL/min, or patients with significant liver disease (hepatitis or cirrhosis). In RECORD 1, the mean exposure duration (± SD) to active XARELTO and enoxaparin was 33.3 ± 7.0 and 33.6 ± 8.3 days, respectively. In RECORD 2, the mean exposure duration to active XARELTO and enoxaparin was 33.5 ± 6.9 and 12.4 ± 2.9 days, respectively. After Day 13, oral placebo was continued in the enoxaparin group for the remainder of the double-blind study duration. The efficacy data for RECORD 1 and 2 are provided in Table 14.
Table 14: Summary of Key Efficacy Analysis Results for Patients Undergoing Total Hip Replacement Surgery - Modified Intent-to-Treat Population RECORD 1 RECORD 2 Treatment Dosage and Duration XARELTO
10 mg once dailyEnoxaparin
40 mg once dailyRRR*,
p-valueXARELTO
10 mg once dailyEnoxaparin†
40 mg once dailyRRR*,
p-value- * Relative Risk Reduction; CI = confidence interval
- † Includes the placebo-controlled period of RECORD 2
- ‡ Proximal DVT, nonfatal PE or VTE-related death
Number of Patients N=1513 N=1473 N=834 N=835 Total VTE 17 (1.1%) 57 (3.9%) 71%
(95% CI: 50, 83),
p<0.00117 (2.0%) 70 (8.4%) 76%
(95% CI: 59, 86),
p<0.001Components of Total VTE Proximal DVT 1 (0.1%) 31 (2.1%) 5 (0.6%) 40 (4.8%) Distal DVT 12 (0.8%) 26 (1.8%) 11 (1.3%) 43 (5.2%) Non-fatal PE 3 (0.2%) 1 (0.1%) 1 (0.1%) 4 (0.5%) Death (any cause) 4 (0.3%) 4 (0.3%) 2 (0.2%) 4 (0.5%) Number of Patients N=1600 N=1587 N=928 N=929 Major VTE‡ 3 (0.2%) 33 (2.1%) 91% (95% CI: 71, 97),
p<0.0016 (0.7%) 45 (4.8%) 87% (95% CI: 69, 94),
p<0.001Number of Patients N=2103 N=2119 N=1178 N=1179 Symptomatic VTE 5 (0.2%) 11 (0.5%) 3 (0.3%) 15 (1.3%) One randomized, double-blind, clinical study (RECORD 3) in patients undergoing elective total knee replacement surgery compared XARELTO 10 mg once daily started at least 6 to 8 hours (about 90% of patients dosed 6 to 10 hours) after wound closure versus enoxaparin. In RECORD 3, the enoxaparin regimen was 40 mg once daily started 12 hours preoperatively. The mean age (± SD) of patients in the study was 68 ± 9.0 (range 28 to 91) years with 66% of patients ≥65 years. Sixty-eight percent (68%) of patients were female. Eighty-one percent (81%) of patients were White, less than 7% were Asian, and less than 2% were Black. The study excluded patients with severe renal impairment defined as an estimated creatinine clearance <30 mL/min or patients with significant liver disease (hepatitis or cirrhosis). The mean exposure duration (± SD) to active XARELTO and enoxaparin was 11.9 ± 2.3 and 12.5 ± 3.0 days, respectively. The efficacy data are provided in Table 15.
Table 15: Summary of Key Efficacy Analysis Results for Patients Undergoing Total Knee Replacement Surgery - Modified Intent-to-Treat Population RECORD 3 Treatment Dosage and Duration XARELTO
10 mg once dailyEnoxaparin
40 mg once dailyRRR*,
p-value- * Relative Risk Reduction; CI = confidence interval
- † Proximal DVT, nonfatal PE or VTE-related death
Number of Patients N=813 N=871 Total VTE 79 (9.7%) 164 (18.8%) 48%
(95% CI: 34, 60),
p<0.001Components of events contributing to Total VTE Proximal DVT 9 (1.1%) 19 (2.2%) Distal DVT 74 (9.1%) 154 (17.7%) Non-fatal PE 0 4 (0.5%) Death (any cause) 0 2 (0.2%) Number of Patients N=895 N=917 Major VTE† 9 (1.0%) 23 (2.5%) 60% (95% CI: 14, 81),
p = 0.024Number of Patients N=1206 N=1226 Symptomatic VTE 8 (0.7%) 24 (2.0%) 14.5 Prophylaxis of Venous Thromboembolism in Acutely Ill Medical Patients at Risk for Thromboembolic Complications Not at High Risk of Bleeding
The efficacy and safety of XARELTO for prophylaxis of venous thromboembolism in acutely ill medical patients at risk for thromboembolic complications not at high risk of bleeding was evaluated in the MAGELLAN study (Multicenter, rAndomized, parallel Group Efficacy and safety study for the prevention of venous thromboembolism in hospitalized medically iLL patients comparing rivaroxabaN with enoxaparin [NCT00571649]). MAGELLAN was a multicenter, randomized, double-blind, parallel-group efficacy and safety study comparing XARELTO to enoxaparin, in the prevention of VTE in hospitalized acutely ill medical patients during the in-hospital and post-hospital discharge period. Eligible patients included adults who were at least 40 years of age, hospitalized for an acute medical illness, at risk of VTE due to moderate or severe immobility, and had additional risk factors for VTE. The population at risk of VTE was required to have one or more of the following VTE risk factors, i.e. prolonged immobilization, age ≥75 years, history of cancer, history of VTE, history of heart failure, thrombophilia, acute infectious disease contributing to the hospitalization and BMI ≥35 kg/m2). The causes for hospitalization included heart failure, active cancer, acute ischemic stroke, acute infectious and inflammatory disease and acute respiratory insufficiency. Patients were randomized to receive either XARELTO 10 mg once daily for 35 ±4 days starting in hospital and continuing post hospital discharge (n=4050) or enoxaparin 40 mg once daily for 10 ±4 days starting in hospital followed by placebo post-discharge (n=4051).
The major efficacy outcome in the MAGELLAN trial was a composite endpoint that included asymptomatic proximal deep venous thrombosis (DVT) in lower extremity, symptomatic proximal or distal DVT in the lower extremity, symptomatic non-fatal pulmonary embolism (PE), and death related to venous thromboembolism (VTE).
A total of 6024 patients were evaluable for the major efficacy outcome analysis (2967 on XARELTO 10 mg once daily and 3057 on enoxaparin/placebo). The mean age was 68.9 years, with 37.1% of the subject population ≥ 75 years. VTE risk factors included severe immobilization at study entry (99.9%), D-dimer > 2X ULN (43.7%), history of heart failure (35.6%), BMI ≥ 35 kg/m2 (15.2%), chronic venous insufficiency (14.9%), acute infectious disease (13.9%), severe varicosis (12.5%), history of cancer (16.2%), history of VTE (4.5%), hormone replacement therapy (1.1%), and thrombophilia (0.3%), recent major surgery (0.8%) and recent serious trauma (0.2%). The population was 54.7% male, 68.2% White, 20.4% Asian, 1.9% Black and 5.3% Other. Admitting diagnoses for hospitalization were acute infectious diseases (43.8%) followed by congestive heart failure NYHA class III or IV (33.2%), acute respiratory insufficiency (26.4%), acute ischemic stroke (18.5%) and acute inflammatory diseases (3.4%).
Table 16 shows the overall results from the prespecified, modified intent-to-treat (mITT) analysis for the efficacy outcomes and their components. This analysis excludes approximately 25% of the patients mainly due to no ultrasonographic assessment (13.5%), inadequate assessment at day 35 (8.1%), or lack of intake of study medication (1.3%).
Table 16: Efficacy Results at Day 35 (modified Intent-to-Treat) and at Day 10 (per protocol) in the MAGELLAN Study mITT: modified intent-to-treat; PP: per protocol; DVT: Deep vein thrombosis; PE: pulmonary embolism; VTE: venous thromboembolism; CI: Confidence Interval; RR: Relative Risk Events from Day 1 to Day 35, mITT analysis set XARELTO
10 mg
N=2967
n (%)
Enoxaparin 40 mg/placebo
N=3057
n (%)
RR
(95% CI)Primary Composite Endpoint at Day 35 131 (4.4%) 175 (5.7%) 0.77
(0.62, 0.96)Symptomatic non-fatal PE 10 (0.3) 14 (0.5) Symptomatic DVT in lower extremity 13 (0.4) 15 (0.5) Asymptomatic proximal DVT in lower extremity 103 (3.5) 133 (4.4) VTE related death 19 (0.6) 30 (1.0) Events from Day 1 to Day 10, PP analysis set XARELTO
10 mg
N=2938
n (%)Enoxaparin
40 mg
N=2993
n (%)RR
(95% CI)Primary Composite Endpoint at Day 10 78 (2.7) 82 (2.7) 0.97
(0.71, 1.31)Symptomatic non-fatal PE 6 (0.2) 2 (<0.1) Symptomatic DVT in lower extremity 7 (0.2) 6 (0.2) Asymptomatic proximal DVT in lower extremity 71 (2.4) 71 (2.4) VTE related death 3 (0.1) 6 (0.2) mITT analysis set plus all-cause mortality N=3096
n (%)N=3169
n (%)RR
(95% CI)Other Composite Endpoint at Day 35 266 (8.6) 293 (9.2) 0.93
(0.80, 1.09)Symptomatic non-fatal PE 10 (0.3) 14 (0.4) Symptomatic DVT in lower extremity 13 (0.4) 15 (0.5) Asymptomatic proximal DVT in lower extremity 103 (3.3) 133 (4.2) All-cause mortality 159 (5.1) 153 (4.8) Patients with bronchiectasis/pulmonary cavitation, active cancer, dual antiplatelet therapy or active gastroduodenal ulcer or any bleeding in the previous three months (19.4%) all had an excess of bleeding with XARELTO compared with enoxaparin/placebo. Therefore, patients meeting these criteria were excluded from the following analyses presented below.
Table 17 provides the efficacy results for the subgroup of patients not at a high risk of bleeding.
Table 17: Efficacy Results at Day 35 (modified Intent-to-Treat) and at Day 10 (per protocol) in patients not at a high risk of bleeding in the MAGELLAN Study* mITT: modified intent-to-treat; PP: per protocol; DVT: Deep vein thrombosis; PE: pulmonary embolism; VTE: venous thromboembolism; CI: Confidence Interval; RR: Relative Risk - * Patients at high risk of bleeding (i.e. bronchiectasis/pulmonary cavitation, active cancer, dual antiplatelet therapy or active gastroduodenal ulcer or any bleeding in the previous three months) were excluded.
Events from Day 1 to Day 35, mITT analysis set XARELTO
10 mg
N=2419
n (%)
Enoxaparin 40 mg/placebo
N=2506
n (%)
RR
(95% CI)Primary Composite Endpoint at Day 35 94 (3.9) 143 (5.7) 0.68
(0.53, 0.88)Symptomatic non-fatal PE 7 (0.3) 10 (0.4) Symptomatic DVT in lower extremity 9 (0.4) 10 (0.4) Asymptomatic proximal DVT in lower extremity 73 (3.0) 110 (4.4) VTE related death 15 (0.6) 26 (1.0) Events from Day 1 to Day 10, PP analysis set XARELTO
10 mg
N=2385
n (%)Enoxaparin
40 mg
N=2433
n (%)RR
(95% CI)Primary Composite Endpoint at Day 10 58 (2.4) 72 (3.0) 0.82
(0.58, 1.15)Symptomatic non-fatal PE 5 (0.2) 2 (<0.1) Symptomatic DVT in lower extremity 6 (0.3) 4 (0.2) Asymptomatic proximal DVT in lower extremity 52 (2.2) 62 (2.5) VTE related death 2 (<0.1) 6 (0.2) mITT analysis set plus all-cause mortality N=2504
n (%)N=2583
n (%)RR
(95% CI)Other Composite Endpoint at Day 35 184 (7.3) 225 (8.7) 0.84
(0.70, 1.02)Symptomatic non-fatal PE 7 (0.3) 10 (0.4) Symptomatic DVT in lower extremity 9 (0.4) 10 (0.4) Asymptomatic proximal DVT in lower extremity 73 (2.9) 110 (4.3) All-cause mortality 107 (4.3) 112 (4.3) 14.6 Reduction of Risk of Major Cardiovascular Events in Patients with Chronic CAD or PAD
The evidence for the efficacy and safety of XARELTO for the reduction in the risk of stroke, myocardial infarction, or cardiovascular death in patients with coronary artery disease (CAD) or peripheral artery disease (PAD) was derived from the double-blind Cardiovascular OutcoMes for People using Anticoagulation StrategieS trial (COMPASS) [NCT10776424]. A total of 27,395 patients were evenly randomized to rivaroxaban 2.5 mg orally twice daily plus aspirin 100 mg once daily, rivaroxaban 5 mg orally twice daily alone, or aspirin 100 mg once daily alone. Because the 5 mg dose alone was not superior to aspirin alone, only the data concerning the 2.5 mg dose plus aspirin are discussed below.
Patients with established CAD or PAD were eligible. Patients with CAD who were younger than 65 years of age were also required to have documentation of atherosclerosis involving at least two vascular beds or to have at least two additional cardiovascular risk factors (current smoking, diabetes mellitus, an estimated glomerular filtration rate [eGFR] <60 mL per minute, heart failure, or non-lacunar ischemic stroke ≥1 month earlier). Patients with PAD were either symptomatic with ankle brachial index <0.90 or had asymptomatic carotid artery stenosis ≥50%, a previous carotid revascularization procedure, or established ischemic disease of one or both lower extremities. Patients were excluded for use of dual antiplatelet, other non-aspirin antiplatelet, or oral anticoagulant therapies, ischemic, non-lacunar stroke within 1 month, hemorrhagic or lacunar stroke at any time, or eGFR <15 mL/min. [see Warnings and Precautions (5.2)].
The mean age was 68 years and 21% of the subject population were ≥75 years. Of the included patients, 91% had CAD, 27% had PAD, and 18% had both CAD and PAD. Of the patients with CAD, 69% had prior MI, 60% had prior percutaneous transluminal coronary angioplasty (PTCA)/atherectomy/ percutaneous coronary intervention (PCI), and 26% had history of coronary artery bypass grafting (CABG) prior to study. Of the patients with PAD, 49% had intermittent claudication, 27% had peripheral artery bypass surgery or peripheral percutaneous transluminal angioplasty, 26% had asymptomatic carotid artery stenosis > 50%, and 4% had limb or foot amputation for arterial vascular disease.
The mean duration of follow-up was 23 months. Relative to aspirin alone, XARELTO plus aspirin reduced the rate of the primary composite outcome of stroke, myocardial infarction or cardiovascular death. The benefit was observed early with a constant treatment effect over the entire treatment period (see Table 18 and Figure 11).
A benefit-risk analysis of the data from COMPASS was performed by comparing the number of CV events (CV deaths, myocardial infarctions and non-hemorrhagic strokes) prevented to the number of fatal or life-threatening bleeding events (fatal bleeds + symptomatic non-fatal bleeds into a critical organ) in the XARELTO plus aspirin group versus the aspirin group. Compared to aspirin alone, during 10,000 patient-years of treatment, XARELTO plus aspirin would be expected to result in 70 fewer CV events and 12 additional life-threatening bleeds, indicating a favorable balance of benefits and risks.
The results in patients with PAD, CAD, and both CAD and PAD were consistent with the overall efficacy and safety results (see Figure 10).
Figure 10 shows the risk of primary efficacy outcome across major subgroups.
Figure 10: Risk of Primary Efficacy Outcome by Baseline Characteristics in COMPASS (Intent-to-Treat Population)
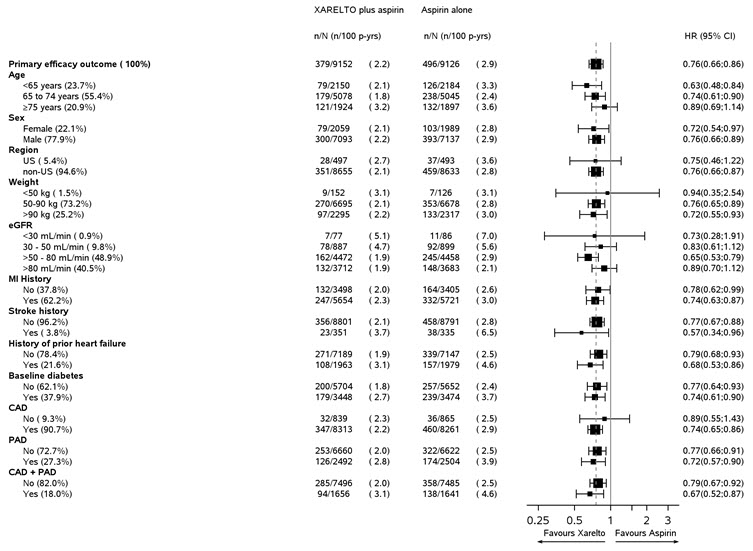
Table 18: Efficacy results from COMPASS study Study Population Patients with CAD or PAD* Event XARELTO plus aspirin†
N=9152Aspirin alone†
N=9126Hazard Ratio
(95% CI) ‡n (%) Event Rate
(%/year)n (%) Event Rate
(%/year)CHD: coronary heart disease, CI: confidence interval; CV: cardiovascular; MI: myocardial infarction - * intention to treat analysis set, primary analyses.
- † Treatment schedule: XARELTO 2.5 mg twice daily plus aspirin 100 mg once daily, or aspirin 100 mg once daily.
- ‡ vs. aspirin 100 mg
- § Coronary heart disease death: death due to acute MI, sudden cardiac death, or CV procedure.
- ¶ Acute limb ischemia is defined as limb-threatening ischemia leading to an acute vascular intervention (i.e., pharmacologic, peripheral arterial surgery/reconstruction, peripheral angioplasty/stent, or amputation).
- # CV death includes CHD death, or death due to other CV causes or unknown death.
Stroke, MI or CV death 379 (4.1) 2.2 496 (5.4) 2.9 0.76 (0.66, 0.86) - Stroke 83 (0.9) 0.5 142 (1.6) 0.8 0.58 (0.44, 0.76) - MI 178 (1.9) 1.0 205 (2.2) 1.2 0.86 (0.70, 1.05) - CV death 160 (1.7) 0.9 203 (2.2) 1.2 0.78 (0.64, 0.96) Coronary heart disease death, MI, ischemic stroke, acute limb ischemia 329 (3.6) 1.9 450 (4.9) 2.6 0.72 (0.63, 0.83) - Coronary heart disease death§ 86 (0.9) 0.5 117 (1.3) 0. 7 0.73 (0.55, 0.96) - Ischemic stroke 64 (0.7) 0.4 125 (1.4) 0.7 0.51 (0.38, 0.69) - Acute limb ischemia¶ 22 (0.2) 0.1 40 (0.4) 0.2 0.55 (0.32, 0.92) CV death#, MI, ischemic stroke, acute limb ischemia 389 (4.3) 2.2 516 (5.7) 3.00 0.74 (0.65, 0.85) All-cause mortality 313 (3.4) 1. 8 378 (4.1) 2.2 0.82 (0.71, 0.96) Lower extremity amputations for CV reasons 15 (0.2) <0.1 31 (0.3) 0.2 0.48 (0.26, 0.89) Patients with PAD Acute limb ischemia 19 (0.8) 0.4 34 (1.4) 0.8 0.56 (0.32, 0.99) Figure 11: Time to first occurrence of primary efficacy outcome (stroke, myocardial infarction, cardiovascular death) in COMPASS
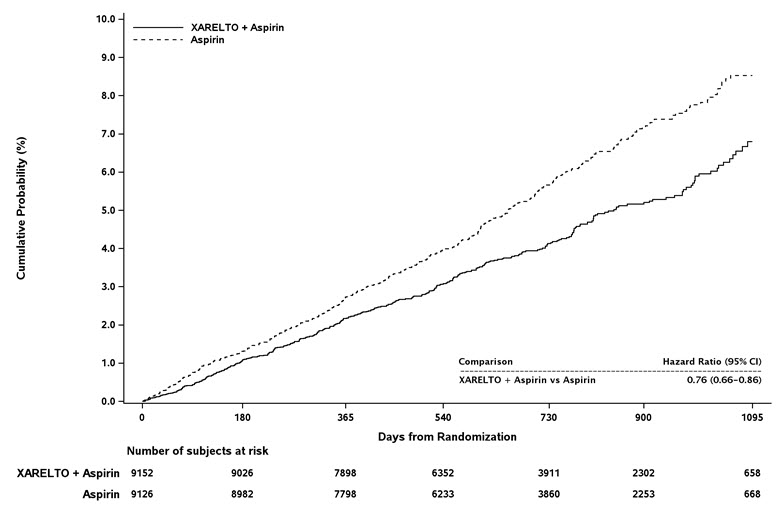
CI: confidence interval
-
16 HOW SUPPLIED/STORAGE AND HANDLING
XARELTO® (rivaroxaban) Tablets are available in the strengths and packages listed below:
- 2.5 mg tablets are round, light yellow, and film-coated with a triangle pointing down above a "2.5" marked on one side and "Xa" on the other side. The tablets are supplied in the packages listed:
NDC: 50458-577-60 Bottle containing 60 tablets NDC: 50458-577-18 Bottle containing 180 tablets NDC: 50458-577-10 Blister package containing 100 tablets (10 blister cards containing 10 tablets each) - 10 mg tablets are round, light red, biconvex film-coated tablets marked with a triangle pointing down above a "10" on one side, and "Xa" on the other side. The tablets are supplied in the packages listed:
NDC: 50458-580-30 Bottle containing 30 tablets NDC: 50458-580-90 Bottle containing 90 tablets NDC: 50458-580-10 Blister package containing 100 tablets (10 blister cards containing 10 tablets each) - 15 mg tablets are round, red, biconvex film-coated tablets with a triangle pointing down above a "15" marked on one side and "Xa" on the other side. The tablets are supplied in the packages listed:
NDC: 50458-578-30 Bottle containing 30 tablets NDC: 50458-578-90 Bottle containing 90 tablets NDC: 50458-578-10 Blister package containing 100 tablets (10 blister cards containing 10 tablets each) - 20 mg tablets are triangle-shaped, dark red film-coated tablets with a triangle pointing down above a "20" marked on one side and "Xa" on the other side. The tablets are supplied in the packages listed:
NDC: 50458-579-30 Bottle containing 30 tablets NDC: 50458-579-90 Bottle containing 90 tablets NDC: 50458-579-89 Bulk bottle containing 1000 tablets NDC: 50458-579-10 Blister package containing 100 tablets (10 blister cards containing 10 tablets each) - Starter Pack for treatment of deep vein thrombosis and treatment of pulmonary embolism:
NDC: 50458-584-51 30-day starter blister pack containing 51 tablets: 42 tablets of 15 mg and 9 tablets of 20 mg
- 2.5 mg tablets are round, light yellow, and film-coated with a triangle pointing down above a "2.5" marked on one side and "Xa" on the other side. The tablets are supplied in the packages listed:
-
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Instructions for Patient Use
- Advise patients to take XARELTO only as directed.
- Remind patients to not discontinue XARELTO without first talking to their healthcare professional.
- Advise patients with atrial fibrillation to take XARELTO once daily with the evening meal.
- Advise patients for initial treatment of DVT and/or PE to take XARELTO 15 mg or 20 mg tablets with food at approximately the same time every day [see Dosage and Administration (2.1)].
- Advise patients who are at a continued risk of recurrent DVT and/or PE after at least 6 months of initial treatment, to take XARELTO 10 mg once daily with or without food [see Dosage and Administration (2.1)].
- Advise patients who cannot swallow the tablet whole to crush XARELTO and combine with a small amount of applesauce followed by food [see Dosage and Administration (2.5)].
- For patients requiring an NG tube or gastric feeding tube, instruct the patient or caregiver to crush the XARELTO tablet and mix it with a small amount of water before administering via the tube [see Dosage and Administration (2.5)].
- If a dose is missed, advise the patient to take XARELTO as soon as possible on the same day and continue on the following day with their recommended daily dose regimen [see Dosage and Administration (2.4)].
Bleeding Risks
- Advise patients to report any unusual bleeding or bruising to their physician. Inform patients that it might take them longer than usual to stop bleeding, and that they may bruise and/or bleed more easily when they are treated with XARELTO [see Warnings and Precautions (5.2)].
- If patients have had neuraxial anesthesia or spinal puncture, and particularly, if they are taking concomitant NSAIDs or platelet inhibitors, advise patients to watch for signs and symptoms of spinal or epidural hematoma, such as back pain, tingling, numbness (especially in the lower limbs), muscle weakness, and stool or urine incontinence. If any of these symptoms occur, advise the patient to contact his or her physician immediately [see Boxed Warning].
Invasive or Surgical Procedures
Instruct patients to inform their healthcare professional that they are taking XARELTO before any invasive procedure (including dental procedures) is scheduled.
Concomitant Medication and Herbals
Advise patients to inform their physicians and dentists if they are taking, or plan to take, any prescription or over-the-counter drugs or herbals, so their healthcare professionals can evaluate potential interactions [see Drug Interactions (7)].
Pregnancy and Pregnancy-Related Hemorrhage
- Advise patients to inform their physician immediately if they become pregnant or intend to become pregnant during treatment with XARELTO [see Use in Specific Populations (8.1)].
- Advise pregnant women receiving XARELTO to immediately report to their physician any bleeding or symptoms of blood loss [see Warnings and Precautions (5.7)].
Lactation
Advise patients to discuss with their physician the benefits and risks of XARELTO for the mother and for the child if they are nursing or intend to nurse during anticoagulant treatment [see Use in Specific Populations (8.2)].
Females and Males of Reproductive Potential
Advise patients who can become pregnant to discuss pregnancy planning with their physician [see Use in Specific Populations (8.3)].
- SPL UNCLASSIFIED SECTION
-
MEDICATION GUIDE
This Medication Guide has been approved by the U.S. Food and Drug Administration Revised: 03/ MEDICATION GUIDE
XARELTO® (zah-REL-toe)
(rivaroxaban)
tabletsWhat is the most important information I should know about XARELTO?
XARELTO may cause serious side effects, including:-
Increased risk of blood clots if you stop taking XARELTO. People with atrial fibrillation (a type of irregular heart beat) that is not caused by a heart valve problem (non-valvular) are at an increased risk of forming a blood clot in the heart, which can travel to the brain, causing a stroke, or to other parts of the body. XARELTO lowers your chance of having a stroke by helping to prevent clots from forming. If you stop taking XARELTO, you may have increased risk of forming a clot in your blood.
Do not stop taking XARELTO without talking to the doctor who prescribes it for you. Stopping XARELTO increases your risk of having a stroke. If you have to stop taking XARELTO, your doctor may prescribe another blood thinner medicine to prevent a blood clot from forming. -
Increased risk of bleeding. XARELTO can cause bleeding which can be serious and may lead to death. This is because XARELTO is a blood thinner medicine (anticoagulant) that lowers blood clotting. During treatment with XARELTO you are likely to bruise more easily, and it may take longer for bleeding to stop. You may have a higher risk of bleeding if you take XARELTO and have certain other medical problems.
You may have a higher risk of bleeding if you take XARELTO and take other medicines that increase your risk of bleeding, including:- aspirin or aspirin containing products
- long-term (chronic) use of non-steroidal anti-inflammatory drugs (NSAIDs)
- warfarin sodium (Coumadin®, Jantoven®)
- any medicine that contains heparin
- clopidogrel (Plavix®)
- selective serotonin reuptake inhibitors (SSRIs) or serotonin norepinephrine reuptake inhibitors (SNRIs)
- other medicines to prevent or treat blood clots
Call your doctor or get medical help right away if you develop any of these signs or symptoms of bleeding:- unexpected bleeding or bleeding that lasts a long time, such as:
- nose bleeds that happen often
- unusual bleeding from the gums
- menstrual bleeding that is heavier than normal or vaginal bleeding
- bleeding that is severe or you cannot control
- red, pink or brown urine
- bright red or black stools (looks like tar)
- cough up blood or blood clots
- vomit blood or your vomit looks like "coffee grounds"
- headaches, feeling dizzy or weak
- pain, swelling, or new drainage at wound sites
-
Spinal or epidural blood clots (hematoma). People who take a blood thinner medicine (anticoagulant) like XARELTO, and have medicine injected into their spinal and epidural area, or have a spinal puncture have a risk of forming a blood clot that can cause long-term or permanent loss of the ability to move (paralysis). Your risk of developing a spinal or epidural blood clot is higher if:
- a thin tube called an epidural catheter is placed in your back to give you certain medicine
- you take NSAIDs or a medicine to prevent blood from clotting
- you have a history of difficult or repeated epidural or spinal punctures
- you have a history of problems with your spine or have had surgery on your spine
XARELTO is not for use in people with antiphospholipid syndrome (APS), especially with positive triple antibody testing.What is XARELTO?
XARELTO is a prescription medicine used to:- reduce the risk of stroke and blood clots in people who have a medical condition called atrial fibrillation that is not caused by a heart valve problem. With atrial fibrillation, part of the heart does not beat the way it should. This can lead to the formation of blood clots, which can travel to the brain, causing a stroke, or to other parts of the body.
- treat blood clots in the veins of your legs (deep vein thrombosis or DVT) or lungs (pulmonary embolism or PE)
- reduce the risk of blood clots happening again in people who continue to be at risk for DVT or PE after receiving treatment for blood clots for at least 6 months.
- help prevent a blood clot in the legs and lungs of people who have just had hip or knee replacement surgery.
- help prevent blood clots in certain people hospitalized for an acute illness and after discharge who are at risk of getting blood clots because of the loss of or decreased ability to move around (mobility) and other risks for getting blood clots and who do not have a high risk of bleeding.
- reduce the risk of serious heart problems, heart attack and stroke in people with coronary artery disease (a condition where the blood supply to the heart is reduced or blocked) or peripheral artery disease (a condition where the blood flow to the legs is reduced).
Do not take XARELTO if you: - currently have certain types of abnormal bleeding. Talk to your doctor before taking XARELTO if you currently have unusual bleeding.
- are allergic to rivaroxaban or any of the ingredients in XARELTO. See the end of this Medication Guide for a complete list of ingredients in XARELTO.
Before taking XARELTO, tell your doctor about all of your medical conditions, including if you: - have or ever had bleeding problems
- have liver or kidney problems
- have antiphospholipid syndrome (APS)
- are pregnant or plan to become pregnant. It is not known if XARELTO will harm your unborn baby.
- Tell your doctor right away if you become pregnant during treatment with XARELTO. Taking XARELTO while you are pregnant may increase the risk of bleeding in you or in your unborn baby.
- If you take XARELTO during pregnancy tell your doctor right away if you have any signs or symptoms of bleeding or blood loss. See "What is the most important information I should know about XARELTO?" for signs and symptoms of bleeding.
- are breastfeeding or plan to breastfeed. XARELTO can pass into your breast milk. Talk to your doctor about the best way to feed your baby during treatment with XARELTO.
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
Some of your other medicines may affect the way XARELTO works, causing side effects. Certain medicines may increase your risk of bleeding. See "What is the most important information I should know about XARELTO?"
Especially tell your doctor if you take:- ketoconazole
- erythromycin
- phenytoin
- St. John's wort
- ritonavir
- carbamazepine
- rifampin
How should I take XARELTO? - Take XARELTO exactly as prescribed by your doctor.
- Do not change your dose or stop taking XARELTO unless your doctor tells you to. Your doctor may change your dose if needed.
- Your doctor will decide how long you should take XARELTO.
- XARELTO may need to be stopped for one or more days before any surgery or medical or dental procedure. Your doctor will tell you when to stop taking XARELTO and when to start taking XARELTO again after your surgery or procedure.
- If you need to stop taking XARELTO for any reason, talk to the doctor who prescribed XARELTO to you to find out when you should stop taking it. Do not stop taking XARELTO without first talking to the doctor who prescribes it to you.
- If you have difficulty swallowing XARELTO tablets whole, talk to your doctor about other ways to take XARELTO.
- Do not run out of XARELTO. Refill your prescription of XARELTO before you run out. When leaving the hospital following a hip or knee replacement, be sure that you will have XARELTO available to avoid missing any doses.
- If you take too much XARELTO, go to the nearest hospital emergency room or call your doctor right away.
-
Atrial fibrillation that is not caused by a heart valve problem:
- Take XARELTO 1 time a day with your evening meal.
- If you miss a dose of XARELTO, take it as soon as you remember on the same day. Take your next dose at your regularly scheduled time.
-
Blood clots in the veins of your legs or lungs:
- Take XARELTO 1 or 2 times a day as prescribed by your doctor.
- For the 10 mg dose, take XARELTO with or without food.
- For the 15 mg and 20 mg doses, take XARELTO with food at the same time each day.
- If you miss a dose:
- ➢ If you take the 15 mg dose of XARELTO 2 times a day (a total of 30 mg of XARELTO in 1 day): Take XARELTO as soon as you remember on the same day. You may take 2 doses at the same time to make up for the missed dose. Take your next dose at your regularly scheduled time.
- ➢ If you take XARELTO 1 time a day: Take XARELTO as soon as you remember on the same day. Take your next dose at your regularly scheduled time.
-
Hip or knee replacement surgery:
- Take XARELTO 1 time a day with or without food.
- If you miss a dose of XARELTO, take it as soon as you remember on the same day. Take your next dose at your regularly scheduled time.
-
Blood clots in people hospitalized for an acute illness:
- Take XARELTO 1 time a day, with or without food, while you are in the hospital and after you are discharged as prescribed by your doctor.
- If you miss a dose of XARELTO, take it as soon as you remember on the same day. Take your next dose at your regularly scheduled time.
-
Reducing the risk of serious heart problems, heart attack and stroke in coronary artery disease or peripheral artery disease:
- Take XARELTO 2.5 mg 2 times a day with or without food.
- If you miss a dose of XARELTO, take your next dose at your regularly scheduled time.
- Take aspirin 75 to 100 mg once daily as instructed by your doctor.
What are the possible side effects of XARELTO?
XARELTO may cause serious side effects: The most common side effect of XARELTO was bleeding.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1 800-FDA-1088.How should I store XARELTO? - Store XARELTO at room temperature between 68°F to 77°F (20°C to 25°C).
General information about the safe and effective use of XARELTO.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use XARELTO for a condition for which it was not prescribed. Do not give XARELTO to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or doctor for information about XARELTO that is written for health professionals.What are the ingredients in XARELTO?
Active ingredient: rivaroxaban
Inactive ingredients: croscarmellose sodium, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, and sodium lauryl sulfate.
The proprietary film coating mixture for XARELTO 2.5 mg tablets is Opadry® Light Yellow and contains: ferric oxide yellow, hypromellose, polyethylene glycol 3350, and titanium dioxide.
The proprietary film coating mixture for XARELTO 10 mg tablets is Opadry® Pink and contains: ferric oxide red, hypromellose, polyethylene glycol 3350, and titanium dioxide.
The proprietary film coating mixture for XARELTO 15 mg tablets is Opadry® Red and contains: ferric oxide red, hypromellose, polyethylene glycol 3350, and titanium dioxide.
The proprietary film coating mixture for XARELTO 20 mg tablets is Opadry® II Dark Red and contains: ferric oxide red, polyethylene glycol 3350, polyvinyl alcohol (partially hydrolyzed), talc, and titanium dioxide.
Finished Product Manufactured by: Janssen Ortho LLC Gurabo, PR 00778 or Bayer AG 51368 Leverkusen, Germany
Manufactured for: Janssen Pharmaceuticals, Inc. Titusville, NJ 08560 Licensed from: Bayer HealthCare AG 51368 Leverkusen, Germany
© 2011-2019 Janssen Pharmaceutical Companies
Trademarks are property of their respective owners.
For more information go to www.XARELTO-US.com or call 1-800-526-7736. -
Increased risk of blood clots if you stop taking XARELTO. People with atrial fibrillation (a type of irregular heart beat) that is not caused by a heart valve problem (non-valvular) are at an increased risk of forming a blood clot in the heart, which can travel to the brain, causing a stroke, or to other parts of the body. XARELTO lowers your chance of having a stroke by helping to prevent clots from forming. If you stop taking XARELTO, you may have increased risk of forming a clot in your blood.
- PRINCIPAL DISPLAY PANEL - 2.5 mg Tablet Bottle Label
- PRINCIPAL DISPLAY PANEL - 10 mg Tablet Bottle Label
- PRINCIPAL DISPLAY PANEL - 15 mg Tablet Bottle Label
- PRINCIPAL DISPLAY PANEL - 20 mg Tablet Bottle Label
-
PRINCIPAL DISPLAY PANEL - Kit Carton
Xarelto®
(rivaroxaban) TabletsNDC: 50458-584-51
Rx onlyPlease see full Prescribing Information, including
BOXED WARNINGS, and Medication Guide inside.Starter Pack
for treatment of deep vein thrombosis and treatment of pulmonary embolismDays 1-21
15 mg per tablet, twice daily
42 tabletsDays 22-30
20 mg per tablet, once daily
9 tabletsFirst 30-day supply
LIFT HERE TO OPEN

-
INGREDIENTS AND APPEARANCE
XARELTO
rivaroxaban tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 50458-577 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength rivaroxaban (UNII: 9NDF7JZ4M3) (rivaroxaban - UNII:9NDF7JZ4M3) rivaroxaban 2.5 mg Inactive Ingredients Ingredient Name Strength croscarmellose sodium (UNII: M28OL1HH48) hypromellose, unspecified (UNII: 3NXW29V3WO) lactose monohydrate (UNII: EWQ57Q8I5X) magnesium stearate (UNII: 70097M6I30) microcrystalline cellulose (UNII: OP1R32D61U) sodium lauryl sulfate (UNII: 368GB5141J) ferric oxide yellow (UNII: EX438O2MRT) polyethylene glycol 3350 (UNII: G2M7P15E5P) titanium dioxide (UNII: 15FIX9V2JP) Product Characteristics Color YELLOW (light yellow) Score no score Shape ROUND Size 6mm Flavor Imprint Code 2;5;Xa Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 50458-577-60 60 in 1 BOTTLE; Type 0: Not a Combination Product 10/09/2018 2 NDC: 50458-577-18 180 in 1 BOTTLE; Type 0: Not a Combination Product 10/09/2018 3 NDC: 50458-577-14 14 in 1 BOTTLE; Type 0: Not a Combination Product 10/09/2018 4 NDC: 50458-577-10 10 in 1 BOX, UNIT-DOSE 10/09/2018 4 NDC: 50458-577-01 10 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA202439 10/09/2018 XARELTO
rivaroxaban tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 50458-580 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength rivaroxaban (UNII: 9NDF7JZ4M3) (rivaroxaban - UNII:9NDF7JZ4M3) rivaroxaban 10 mg Inactive Ingredients Ingredient Name Strength microcrystalline cellulose (UNII: OP1R32D61U) croscarmellose sodium (UNII: M28OL1HH48) hypromellose, unspecified (UNII: 3NXW29V3WO) lactose monohydrate (UNII: EWQ57Q8I5X) magnesium stearate (UNII: 70097M6I30) sodium lauryl sulfate (UNII: 368GB5141J) polyethylene glycol 3350 (UNII: G2M7P15E5P) titanium dioxide (UNII: 15FIX9V2JP) ferric oxide red (UNII: 1K09F3G675) Product Characteristics Color RED (light red) Score no score Shape ROUND (biconvex) Size 6mm Flavor Imprint Code 10;Xa Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 50458-580-10 10 in 1 CARTON 07/01/2011 1 NDC: 50458-580-01 10 in 1 BLISTER PACK; Type 0: Not a Combination Product 2 NDC: 50458-580-30 30 in 1 BOTTLE; Type 0: Not a Combination Product 07/01/2011 3 NDC: 50458-580-07 7 in 1 BOTTLE; Type 0: Not a Combination Product 11/01/2017 4 NDC: 50458-580-90 90 in 1 BOTTLE; Type 0: Not a Combination Product 11/01/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA022406 07/01/2011 XARELTO
rivaroxaban tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 50458-578 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength rivaroxaban (UNII: 9NDF7JZ4M3) (rivaroxaban - UNII:9NDF7JZ4M3) rivaroxaban 15 mg Inactive Ingredients Ingredient Name Strength microcrystalline cellulose (UNII: OP1R32D61U) croscarmellose sodium (UNII: M28OL1HH48) hypromellose, unspecified (UNII: 3NXW29V3WO) lactose monohydrate (UNII: EWQ57Q8I5X) magnesium stearate (UNII: 70097M6I30) sodium lauryl sulfate (UNII: 368GB5141J) polyethylene glycol 3350 (UNII: G2M7P15E5P) titanium dioxide (UNII: 15FIX9V2JP) ferric oxide red (UNII: 1K09F3G675) Product Characteristics Color RED Score no score Shape ROUND (biconvex) Size 6mm Flavor Imprint Code 15;Xa Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 50458-578-10 10 in 1 CARTON 11/04/2011 1 NDC: 50458-578-01 10 in 1 BLISTER PACK; Type 0: Not a Combination Product 2 NDC: 50458-578-30 30 in 1 BOTTLE; Type 0: Not a Combination Product 11/04/2011 3 NDC: 50458-578-90 90 in 1 BOTTLE; Type 0: Not a Combination Product 11/04/2011 4 NDC: 50458-578-14 14 in 1 BOTTLE; Type 0: Not a Combination Product 11/04/2011 5 NDC: 50458-578-07 7 in 1 BOTTLE; Type 0: Not a Combination Product 09/20/2016 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA202439 11/04/2011 XARELTO
rivaroxaban tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 50458-579 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength rivaroxaban (UNII: 9NDF7JZ4M3) (rivaroxaban - UNII:9NDF7JZ4M3) rivaroxaban 20 mg Inactive Ingredients Ingredient Name Strength microcrystalline cellulose (UNII: OP1R32D61U) croscarmellose sodium (UNII: M28OL1HH48) hypromellose, unspecified (UNII: 3NXW29V3WO) lactose monohydrate (UNII: EWQ57Q8I5X) magnesium stearate (UNII: 70097M6I30) sodium lauryl sulfate (UNII: 368GB5141J) polyethylene glycol 3350 (UNII: G2M7P15E5P) titanium dioxide (UNII: 15FIX9V2JP) ferric oxide red (UNII: 1K09F3G675) Product Characteristics Color RED (dark red) Score no score Shape TRIANGLE Size 7mm Flavor Imprint Code 20;Xa Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 50458-579-10 10 in 1 CARTON 11/05/2011 1 NDC: 50458-579-01 10 in 1 BLISTER PACK; Type 0: Not a Combination Product 2 NDC: 50458-579-30 30 in 1 BOTTLE; Type 0: Not a Combination Product 11/05/2011 3 NDC: 50458-579-90 90 in 1 BOTTLE; Type 0: Not a Combination Product 11/05/2011 4 NDC: 50458-579-99 5 in 1 BOTTLE; Type 0: Not a Combination Product 11/05/2011 5 NDC: 50458-579-07 7 in 1 BOTTLE; Type 0: Not a Combination Product 09/20/2016 6 NDC: 50458-579-89 1000 in 1 BOTTLE; Type 0: Not a Combination Product 01/06/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA202439 11/05/2011 XARELTO
rivaroxaban kitProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 50458-584 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 50458-584-51 1 in 1 CARTON 09/16/2014 2 NDC: 50458-584-52 1 in 1 CARTON 11/01/2016 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 BLISTER PACK 42 Part 2 1 BLISTER PACK 9 Part 1 of 2 XARELTO
rivaroxaban tablet, film coatedProduct Information Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength rivaroxaban (UNII: 9NDF7JZ4M3) (rivaroxaban - UNII:9NDF7JZ4M3) rivaroxaban 15 mg Inactive Ingredients Ingredient Name Strength microcrystalline cellulose (UNII: OP1R32D61U) croscarmellose sodium (UNII: M28OL1HH48) hypromellose, unspecified (UNII: 3NXW29V3WO) lactose monohydrate (UNII: EWQ57Q8I5X) magnesium stearate (UNII: 70097M6I30) sodium lauryl sulfate (UNII: 368GB5141J) polyethylene glycol 3350 (UNII: G2M7P15E5P) titanium dioxide (UNII: 15FIX9V2JP) ferric oxide red (UNII: 1K09F3G675) Product Characteristics Color RED Score no score Shape ROUND (biconvex) Size 6mm Flavor Imprint Code 15;Xa Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 42 in 1 BLISTER PACK; Type 1: Convenience Kit of Co-Package Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA022406 09/16/2014 Part 2 of 2 XARELTO
rivaroxaban tablet, film coatedProduct Information Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength rivaroxaban (UNII: 9NDF7JZ4M3) (rivaroxaban - UNII:9NDF7JZ4M3) rivaroxaban 20 mg Inactive Ingredients Ingredient Name Strength microcrystalline cellulose (UNII: OP1R32D61U) croscarmellose sodium (UNII: M28OL1HH48) hypromellose, unspecified (UNII: 3NXW29V3WO) lactose monohydrate (UNII: EWQ57Q8I5X) magnesium stearate (UNII: 70097M6I30) sodium lauryl sulfate (UNII: 368GB5141J) polyethylene glycol 3350 (UNII: G2M7P15E5P) titanium dioxide (UNII: 15FIX9V2JP) ferric oxide red (UNII: 1K09F3G675) POLYVINYL ALCOHOL, UNSPECIFIED (UNII: 532B59J990) talc (UNII: 7SEV7J4R1U) Product Characteristics Color RED (dark red) Score no score Shape TRIANGLE Size 7mm Flavor Imprint Code 20;Xa Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 9 in 1 BLISTER PACK; Type 1: Convenience Kit of Co-Package Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA022406 09/16/2014 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA022406 09/16/2014 Labeler - Janssen Pharmaceuticals, Inc. (063137772) Establishment Name Address ID/FEI Business Operations Bayer AG 314947622 MANUFACTURE(50458-580, 50458-578, 50458-579, 50458-584) , API MANUFACTURE(50458-577, 50458-580, 50458-578, 50458-579, 50458-584) Establishment Name Address ID/FEI Business Operations Bayer Aktiengesellschaft 342872971 API MANUFACTURE(50458-577, 50458-580, 50458-578, 50458-579, 50458-584) Establishment Name Address ID/FEI Business Operations Bayer AG 314398484 ANALYSIS(50458-577, 50458-580, 50458-578, 50458-579, 50458-584) Establishment Name Address ID/FEI Business Operations Janssen Ortho LLC 805887986 MANUFACTURE(50458-577, 50458-580, 50458-578, 50458-579, 50458-584) , ANALYSIS(50458-577, 50458-580, 50458-578, 50458-579, 50458-584) Establishment Name Address ID/FEI Business Operations AndersonBrecon Inc. 053217022 PACK(50458-577, 50458-578, 50458-579, 50458-580, 50458-584)
Trademark Results [XARELTO]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 XARELTO 78152154 2804753 Live/Registered |
Bayer Aktiengesellschaft 2002-08-08 |
 XARELTO 77752463 4105934 Live/Registered |
Bayer Aktiengesellschaft 2009-06-04 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.