POTASSIUM CITRATE tablet, extended release
Potassium Citrate by
Drug Labeling and Warnings
Potassium Citrate by is a Prescription medication manufactured, distributed, or labeled by American Health Packaging. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use Potassium Citrate Extended-release Tablets safely and effectively. See full prescribing information for Potassium Citrate Extended-release Tablets.
POTASSIUM citrate extended-release tablets for oral use
Initial U.S. Approval: 1985INDICATIONS AND USAGE
DOSAGE AND ADMINISTRATION
Objective: To restore normal urinary citrate (greater than 320 mg/day and as close to the normal mean of 640 mg/day as possible), and to increase urinary pH to a level of 6 to 7.
- Severe hypocitraturia (urinary citrate < 150 mg/day): therapy should be initiated at 60 mEq per day; a dose of 30 mEq two times per day or 20 mEq three times per day with meals or within 30 minutes after meals or bedtime snack (2.2)
- Mild to moderate hypocitraturia (urinary citrate > 150 mg/day): therapy should be initiated at 30 mEq per day; a dose of 15 mEq two times per day or 10 mEq three times per day with meals or within 30 minutes after meals or bedtime snack (2.3)
DOSAGE FORMS AND STRENGTHS
Tablets: 5 mEq, 10 mEq and 15 mEq (3)
CONTRAINDICATIONS
- Patients with hyperkalemia (or who have conditions predisposing them to hyperkalemia). Such conditions include chronic renal failure, uncontrolled diabetes mellitus, acute dehydration, strenuous physical exercise in unconditioned individuals, adrenal insufficiency, extensive tissue breakdown (4)
- Patients for whom there is cause for arrest or delay in tablet passage through the gastrointestinal tract such as those suffering from delayed gastric emptying, esophageal compression, intestinal obstruction or stricture (4)
- Patients with peptic ulcer disease (4)
- Patients with active urinary tract infection (4)
- Patients with renal insufficiency (glomerular filtration rate of less than 0.7 ml/kg/min) (4)
WARNINGS AND PRECAUTIONS
- Hyperkalemia: In patients with impaired mechanisms for excreting potassium, potassium citrate administration can produce hyperkalemia and cardiac arrest. Potentially fatal hyperkalemia can develop rapidly and be asymptomatic. The use of potassium citrate in patients with chronic renal failure, or any other condition which impairs potassium excretion such as severe myocardial damage or heart failure, should be avoided (5.1)
- Gastrointestinal lesions: if there is severe vomiting, abdominal pain or gastrointestinal bleeding, potassium citrate should be discontinued immediately and the possibility of bowel perforation or obstruction investigated (5.2)
ADVERSE REACTIONS
Some patients may develop minor gastrointestinal complaints such as abdominal discomfort, vomiting, diarrhea, loose bowel movements or nausea. These may be alleviated by taking the dose with meals or snacks or by reducing the dosage (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Zydus Pharmaceuticals USA Inc. at 1-877-993-8779 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
DRUG INTERACTIONS
The following drug interactions may occur with potassium citrate:
- Potassium-sparing diuretics: concomitant administration should be avoided since the simultaneous administration of these agents can produce severe hyperkalemia (7.1)
- Drugs that slow gastrointestinal transit time: These agents (such as anticholinergics) can be expected to increase the gastrointestinal irritation produced by potassium salts (7.2)
USE IN SPECIFIC POPULATIONS
- Pregnant women: Pregnancy Category C; animal reproduction studies have not been conducted. It is not known whether potassium citrate can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Potassium citrate should be given to a pregnant woman only if clearly needed (8.1)
- Nursing mothers: The normal potassium ion content of human milk is about 13 mEq/L. It is not known if potassium citrate has an effect on this content. Potassium citrate should be given to a woman who is breastfeeding only if clearly needed (8.3)
- Pediatric Use: Safety and effectiveness in children have not been established (8.4)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 4/2018
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
1.1 Renal tubular acidosis (RTA) with calcium stones
1.2 Hypocitraturic calcium oxalate nephrolithiasis of any etiology
1.3 Uric acid lithiasis with or without calcium stones
2 DOSAGE AND ADMINISTRATION
2.1 Dosing Instructions
2.2 Severe Hypocitraturia
2.3 Mild to Moderate Hypocitraturia
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Hyperkalemia
5.2 Gastrointestinal Lesions
6 ADVERSE REACTIONS
6.1 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Potential Effects of Potassium citrate on Other Drugs
7.2 Potential Effects of Other Drugs on Potassium citrate
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.3 Nursing Mothers
8.4 Pediatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
14 CLINICAL STUDIES
14.1 Renal tubular acidosis (RTA) with calcium stones
14.2 Hypocitraturic calcium oxalate nephrolithiasis of any etiology
14.3 Uric acid lithiasis with or without calcium stones
15 REFERENCES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
17.1 Administration of Drug
- * Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
1.1 Renal tubular acidosis (RTA) with calcium stones
Potassium citrate is indicated for the management of renal tubular acidosis [see Clinical Studies (14.1)].
1.2 Hypocitraturic calcium oxalate nephrolithiasis of any etiology
Potassium citrate is indicated for the management of Hypocitraturic calcium oxalate nephrolithiasis [see Clinical Studies (14.2)].
1.3 Uric acid lithiasis with or without calcium stones
Potassium citrate is indicated for the management of Uric acid lithiasis with or without calcium stones [see Clinical Studies (14.3)].
-
2 DOSAGE AND ADMINISTRATION
2.1 Dosing Instructions
Treatment with extended release potassium citrate should be added to a regimen that limits salt intake (avoidance of foods with high salt content and of added salt at the table) and encourages high fluid intake (urine volume should be at least two liters per day). The objective of treatment with potassium citrate extended-release tablets is to provide potassium citrate in sufficient dosage to restore normal urinary citrate (greater than 320 mg/day and as close to the normal mean of 640 mg/day as possible), and to increase urinary pH to a level of 6 or 7.
Monitor serum electrolytes (sodium, potassium, chloride and carbon dioxide), serum creatinine and complete blood counts every four months and more frequently in patients with cardiac disease, renal disease or acidosis. Perform electrocardiograms periodically. Treatment should be discontinued if there is hyperkalemia, a significant rise in serum creatinine or a significant fall in blood hemocrit or hemoglobin.
2.2 Severe Hypocitraturia
In patients with severe hypocitraturia (urinary citrate < 150 mg/day), therapy should be initiated at a dosage of 60 mEq/day (30 mEq two times/day or 20 mEq three times/day with meals or within 30 minutes after meals or bedtime snack). Twenty-four hour urinary citrate and/or urinary pH measurements should be used to determine the adequacy of the initial dosage and to evaluate the effectiveness of any dosage change. In addition, urinary citrate and/or pH should be measured every four months. Doses of potassium citrate extended-release tablets greater than 100 mEq/day have not been studied and should be avoided.
2.3 Mild to Moderate Hypocitraturia
In patients with mild to moderate hypocitraturia (urinary citrate > 150 mg/day) therapy should be initiated at 30 mEq/day (15 mEq two times/day or 10 mEq three times/day with meals or within 30 minutes after meals or bedtime snack). Twenty-four hour urinary citrate and/or urinary pH measurements should be used to determine the adequacy of the initial dosage and to evaluate the effectiveness of any dosage change. Doses of potassium citrate extended-release tablet greater than 100 mEq/day have not been studied and should be avoided.
-
3 DOSAGE FORMS AND STRENGTHS
- 5 mEq tablets are tan to yellowish color, round shaped, biconvex uncoated tablets debossed with "536" on one side and plain on the other side.
- 10 mEq tablets are tan to yellowish color, oval shaped, biconvex uncoated tablets debossed with "537" on one side and plain on the other side.
- 15 mEq tablets are tan to yellowish color, oblong shaped, biconvex uncoated tablets debossed with "538" on one side and plain on the other side.
-
4 CONTRAINDICATIONS
Potassium citrate extended-release tablets are contraindicated:
- In patients with hyperkalemia (or who have conditions pre-disposing them to hyperkalemia), as a further rise in serum potassium concentration may produce cardiac arrest. Such conditions include: chronic renal failure, uncontrolled diabetes mellitus, acute dehydration, strenuous physical exercise in unconditioned individuals, adrenal insufficiency, extensive tissue breakdown or the administration of a potassium-sparing agent (such as triamterene, spironolactone or amiloride).
- In patients in whom there is cause for arrest or delay in tablet passage through the gastrointestinal tract, such as those suffering from delayed gastric emptying, esophageal compression, intestinal obstruction or stricture, or those taking anticholinergic medication.
- In patients with peptic ulcer disease because of its ulcerogenic potential.
- In patients with active urinary tract infection (with either urea-splitting or other organisms, in association with either calcium or struvite stones). The ability of potassium citrate extended-release tablet to increase urinary citrate may be attenuated by bacterial enzymatic degradation of citrate. Moreover, the rise in urinary pH resulting from potassium citrate extended-release tablet therapy might promote further bacterial growth.
- In patients with renal insufficiency (glomerular filtration rate of less than 0.7 ml/kg/min), because of the danger of soft tissue calcification and increased risk for the development of hyperkalemia.
-
5 WARNINGS AND PRECAUTIONS
5.1 Hyperkalemia
In patients with impaired mechanisms for excreting potassium, potassium citrate administration can produce hyperkalemia and cardiac arrest. Potentially fatal hyperkalemia can develop rapidly and be asymptomatic. The use of potassium citrate in patients with chronic renal failure, or any other condition which impairs potassium excretion such as severe myocardial damage or heart failure, should be avoided. Closely monitor for signs of hyperkalemia with periodic blood tests and ECGs.
5.2 Gastrointestinal Lesions
Because of reports of upper gastrointestinal mucosal lesions following administration of potassium-chloride (wax-matrix), an endoscopic examination of the upper gastrointestinal mucosa was performed in 30 normal volunteers after they had taken glycopyrrolate 2 mg p.o. t.i.d., potassium citrate 95 mEq/day, wax-matrix potassium chloride 96 mEq/day or wax-matrix placebo, in thrice daily schedule in the fasting state for one week. Potassium citrate and the wax-matrix formulation of potassium chloride were indistinguishable but both were significantly more irritating than the wax-matrix placebo. In a subsequent, similar study, lesions were less severe when glycopyrrolate was omitted.
Solid dosage forms of potassium chlorides have produced stenotic and/or ulcerative lesions of the small bowel and deaths. These lesions are caused by a high local concentration of potassium ions in the region of the dissolving tablets, which injured the bowel. In addition, perhaps because wax-matrix preparations are not enteric-coated and release some of their potassium content in the stomach, there have been reports of upper gastrointestinal bleeding associated with these products. The frequency of gastrointestinal lesions with wax-matrix potassium chloride products is estimated at one per 100,000 patient-years. Experience with potassium citrate is limited, but a similar frequency of gastrointestinal lesions should be anticipated.
If there is severe vomiting, abdominal pain or gastrointestinal bleeding, potassium citrate should be discontinued immediately and the possibility of bowel perforation or obstruction investigated.
-
6 ADVERSE REACTIONS
6.1 Postmarketing Experience
Some patients may develop minor gastrointestinal complaints during potassium citrate therapy, such as abdominal discomfort, vomiting, diarrhea, loose bowel movements or nausea. These symptoms are due to the irritation of the gastrointestinal tract, and may be alleviated by taking the dose with meals or snacks, or by reducing the dosage. Patients may find intact matrices in their feces.
-
7 DRUG INTERACTIONS
7.1 Potential Effects of Potassium citrate on Other Drugs
Potassium-sparing Diuretics: Concomitant administration of potassium citrate and a potassium-sparing diuretic (such as triamterene, spironolactone or amiloride) should be avoided since the simultaneous administration of these agents can produce severe hyperkalemia.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Category C
Animal reproduction studies have not been conducted. It is also not known whether potassium citrate can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Potassium citrate should be given to a pregnant woman only if clearly needed. -
10 OVERDOSAGE
Treatment of Overdosage: The administration of potassium salts to persons without predisposing conditions for hyperkalemia rarely causes serious hyperkalemia at recommended dosages. It is important to recognize that hyperkalemia is usually asymptomatic and may be manifested only by an increased serum potassium concentration and characteristic electrocardiographic changes (peaking of T-wave, loss of P-wave, depression of S-T segment and prolongation of the QT interval). Late manifestations include muscle paralysis and cardiovascular collapse from cardiac arrest.
Treatment measures for hyperkalemia include the following:
- Patients should be closely monitored for arrhythmias and electrolyte changes.
- Elimination of medications containing potassium and of agents with potassium sparing properties such as potassium-sparing diuretics, ARBs, ACE inhibitors, NSAIDs, certain nutritional supplements and many others.
- Elimination of foods containing high levels of potassium such as almonds, apricots, bananas, beans (lima, pinto, white), cantaloupe, carrot juice (canned), figs, grapefruit juice, halibut, milk, oat bran, potato (with skin), salmon, spinach, tuna and many others.
- Intravenous calcium gluconate if the patient is at no risk or low risk of developing digitalis toxicity.
- Intravenous administration of 300 to 500 mL/hr of 10% dextrose solution containing 10 to 20 units of crystalline insulin per 1,000 mL.
- Correction of acidosis, if present, with intravenous sodium bicarbonate.
- Hemodialysis or peritoneal dialysis.
- Exchange resins may be used. However, this measure alone is not sufficient for the acute treatment of hyperkalemia.
Lowering potassium levels too rapidly in patients taking digitalis can produce digitalis toxicity.
-
11 DESCRIPTION
Potassium citrate is a citrate salt of potassium. Its molecular formula is K 3C 6H 5O 7 H 2O, and it has the following chemical structure:
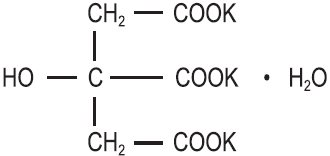
Potassium citrate, USP is a transparent crystals or white, granular powder. It is freely soluble in water and slightly soluble in alcohol.
Potassium citrate extended-release tablets, USP intended for oral administration, each contains 5 mEq (540 mg), 10 mEq (1080 mg) or 15 mEq (1620 mg) of potassium citrate, USP as monohydrate. In addition, each tablet contains the following inactive ingredients: carnauba wax and magnesium stearate.
Meets USP Dissolution Test 2
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
When potassium citrate is given orally, the metabolism of absorbed citrate produces an alkaline load. The induced alkaline load in turn increases urinary pH and raises urinary citrate by augmenting citrate clearance without measurably altering ultrafilterable serum citrate. Thus, potassium citrate therapy appears to increase urinary citrate principally by modifying the renal handling of citrate, rather than by increasing the filtered load of citrate. The increased filtered load of citrate may play some role, however, as in small comparisons of oral citrate and oral bicarbonate, citrate had a greater effect on urinary citrate.
In addition to raising urinary pH and citrate, potassium citrate increases urinary potassium by approximately the amount contained in the medication. In some patients, potassium citrate causes a transient reduction in urinary calcium.
The changes induced by potassium citrate produce urine that is less conducive to the crystallization of stone-forming salts (calcium oxalate, calcium phosphate and uric acid). Increased citrate in the urine, by complexing with calcium, decreases calcium ion activity and thus the saturation of calcium oxalate. Citrate also inhibits the spontaneous nucleation of calcium oxalate and calcium phosphate (brushite).
The increase in urinary pH also decreases calcium ion activity by increasing calcium complexation to dissociated anions. The rise in urinary pH also increases the ionization of uric acid to the more soluble urate ion.
Potassium citrate therapy does not alter the urinary saturation of calcium phosphate, since the effect of increased citrate complexation of calcium is opposed by the rise in pH-dependent dissociation of phosphate. Calcium phosphate stones are more stable in alkaline urine.
In the setting of normal renal function, the rise in urinary citrate following a single dose begins by the first hour and lasts for 12 hours. With multiple doses the rise in citrate excretion reaches its peak by the third day and averts the normally wide circadian fluctuation in urinary citrate, thus maintaining urinary citrate at a higher, more constant level throughout the day. When the treatment is withdrawn, urinary citrate begins to decline toward the pretreatment level on the first day.
The rise in citrate excretion is directly dependent on the potassium citrate dosage. Following long-term treatment, potassium citrate at a dosage of 60 mEq/day raises urinary citrate by approximately 400 mg/day and increases urinary pH by approximately 0.7 units.
In patients with severe renal tubular acidosis or chronic diarrheal syndrome where urinary citrate may be very low (<100 mg/day), potassium citrate may be relatively ineffective in raising urinary citrate. A higher dose of potassium citrate may therefore be required to produce a satisfactory citraturic response. In patients with renal tubular acidosis in whom urinary pH may be high, potassium citrate produces a relatively small rise in urinary pH.
-
14 CLINICAL STUDIES
The pivotal potassium citrate trials were non-randomized and non-placebo controlled where dietary management may have changed coincidentally with pharmacological treatment. Therefore, the results as presented in the following sections may overstate the effectiveness of the product.
14.1 Renal tubular acidosis (RTA) with calcium stones
The effect of oral potassium citrate therapy in a non-randomized, non-placebo controlled clinical study of five men and four women with calcium oxalate/calcium phosphate nephrolithiasis and documented incomplete distal renal tubular acidosis was examined. The main inclusion criterion was a history of stone passage or surgical removal of stones during the 3 years prior to initiation of potassium citrate therapy. All patients began alkali treatment with 60 to 80 mEq potassium citrate daily in 3 or 4 divided doses. Throughout treatment, patients were instructed to stay on a sodium restricted diet (100 mEq/day) and to reduce oxalate intake (limited intake of nuts, dark roughage, chocolate and tea). A moderate calcium restriction (400 to 800 mg/day) was imposed on patients with hypercalciuria.
X-rays of the urinary tract, available in all patients, were reviewed to determine presence of pre-existing stones, appearance of new stones, or change in the number of stones.
Potassium citrate therapy was associated with inhibition of new stone formation in patients with distal tubular acidosis. Three of the nine patients continued to pass stones during the on-treatment phase. While it is likely that these patients passed preexisting stones during therapy, the most conservative assumption is that the passed stones were newly formed. Using this assumption, the stone-passage remission rate was 67%. All patients had a reduced stone formation rate. Over the first 2 years of treatment, the on-treatment stone formation rate was reduced from 13±27 to 1±2 per year.
14.2 Hypocitraturic calcium oxalate nephrolithiasis of any etiology
Eighty-nine patients with hypocitraturic calcium nephrolithiasis or uric acid lithiasis with or without calcium nephrolithiasis participated in this non-randomized, non-placebo controlled clinical study. Four groups of patients were treated with potassium citrate: Group 1 was comprised of 19 patients, 10 with renal tubular acidosis and 9 with chronic diarrheal syndrome, Group 2 was comprised of 37 patients, 5 with uric acid stones alone, 6 with uric acid lithiasis and calcium stones, 3 with type 1 absorptive hypercalciuria, 9 with type 2 absorptive hypercalciuria and 14 with hypocitraturia. Group 3 was comprised of 15 patients with history of relapse on other therapy and Group 4 was comprised of 18 patients, 9 with type 1 absorptive hypercalciuria and calcium stones, 1 with type 2 absorptive hypercalciuria and calcium stones, 2 with hyperuricosuric calcium oxalate nephrolithiasis, 4 with uric acid lithiasis accompanied by calcium stones and 2 with hypocitraturia and hyperuricemia accompanied by calcium stones. The dose of potassium citrate ranged from 30 to 100 mEq per day, and usually was 20 mEq administered orally 3 times daily. Patients were followed in an outpatient setting every 4 months during treatment and were studied over a period from 1 to 4.33 years. A three-year retrospective pre-study history for stone passage or removal was obtained and corroborated by medical records. Concomitant therapy (with thiazide or allopurinol) was allowed if patients had hypercalciuria, hyperuricosuria or hyperuricemia. Group 2 was treated with potassium citrate alone.
In all groups, treatment that included potassium citrate was associated with a sustained increase in urinary citrate excretion from subnormal values to normal values (400 to 700 mg/day), and a sustained increase in urinary pH from 5.6 to 6 to approximately 6.5. The stone formation rate was reduced in all groups as shown in Table 1.
Table 1 Effect of Potassium Citrate In Patients With Calcium Oxalate Nephrolithiasis. Stones Formed Per Year - * Remission defined as “the percentage of patients remaining free of newly formed stones during treatment”.
Group
Baseline
On Treatment
Remission*
Any Decrease
I (n=19)
12 ± 30
0.9 ± 1.3
58%
95%
II (n=37)
1.2 ± 2
0.4 ± 1.5
89%
97%
III (n=15)
4.2 ± 7
0.7 ± 2
67%
100%
IV (n=18)
3.4 ± 8
0.5 ± 2
94%
100%
Total (n=89)
4.3 ± 15
0.6 ± 2
80%
98%
14.3 Uric acid lithiasis with or without calcium stones
A long-term non-randomized, non-placebo controlled clinical trial with eighteen adult patients with uric acid lithiasis participated in the study. Six patients formed only uric acid stones, and the remaining 12 patients formed mixed stones containing both uric acid and calcium salts or formed both uric acid stones (without calcium salts) and calcium stones (without uric acid) on separate occasions.
Eleven of the 18 patients received potassium citrate alone. Six of the 7 other patients also received allopurinol for hyperuricemia with gouty arthritis, symptomatic hyperuricemia, or hyperuricosuria. One patient also received hydrochlorothiazide because of unclassified hypercalciuria. The main inclusion criterion was a history of stone passage or surgical removal of stones during the 3 years prior to initiation of potassium citrate therapy. All patients received potassium citrate at a dosage of 30 to 80 mEq/day in three-to-four divided doses and were followed every four months for up to 5 years.
While on potassium citrate treatment, urinary pH rose significantly from a low value of 5.3 ± 0.3 to within normal limits (6.2 to 6.5). Urinary citrate which was low before treatment rose to the high normal range and only one stone was formed in the entire group of 18 patients.
-
15 REFERENCES
- Pak, C. (1987). Citrate and Renal Calculi. Mineral and Electrolyte Metabolism 13, 257-266.
- Pak, C. (1985). Long-Term Treatment of Calcium Nephrolithiasis with Potassium Citrate. The Journal of Urology 134, 11-19.
- Preminger, G.M., K. Sakhaee, C. Skurla and C.Y.C. Pak. (1985). Prevention of Recurrent Calcium Stone Formation with Potassium Citrate therapy in Patients with Distal Renal Tubular Acidosis. The Journal of Urology 134, 20-23.
- Pak, C.Y.C., K. Sakhaee and C. Fuller. (1986). Successful Management of Uric Acid Nephrolithiasis with Potassium Citrate. Kidney International 30, 422-428.
- Hollander-Rodriguez, J et al. (2006). Hyperkalemia, American Family Physician, Vol.73/No. 2.
- Greenberg, A et al. (1998). Hyperkalemia: treatment options. Semen Nephrol. Jan; 18 (1): 46-57.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
Potassium Citrate Extended-release Tablets, 10 mEq are tan to yellowish color, oval shaped, biconvex uncoated tablets debossed with "537" on one side and plain on the other side and are supplied as follows:
Unit dose packages of 20 (5 x 4) NDC: 68084-850-32Storage: Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
FOR YOUR PROTECTION: Do not use if blister is torn or broken.
-
17 PATIENT COUNSELING INFORMATION
17.1 Administration of Drug
Tell patients to take each dose without crushing, chewing or sucking the tablet.
Tell patients to take this medicine only as directed. This is especially important if the patient is also taking both diuretics and digitalis preparations.
Tell patients to check with the doctor if there is trouble swallowing tablets or if the tablet seems to stick in the throat.
Tell patients to check with the doctor at once if tarry stools or other evidence of gastrointestinal bleeding is noticed.
Tell patients that their doctor will perform regular blood tests and electrocardiograms to ensure safety.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
-
PACKAGING INFORMATION
American Health Packaging unit dose blisters (see How Supplied section) contain drug product from Zydus Pharmaceuticals (USA) Inc. as follows:
(10 mEq / 20 UD) NDC: 68084-850-32 packaged from NDC: 68382-537Distributed by:
American Health Packaging
Columbus, OH 432178285032/0918F
-
Package/Label Display Panel – Carton – 10 mEq

NDC 68084- 850-32
Potassium Citrate
Extended-Release Tablets USP10 mEq
(1080 mg per tablet)20 Tablets (5 x 4) Rx Only
PHARMACIST: Dispense with the accompanying Package
Insert to each patient.Each Extended-Release Tablet Contains:
10 mEq (1080 mg) of potassium citrate, USP as monohydrate.Each 10 mEq tablet is debossed with "537" on one side and
plain on other side.Potassium Citrate Extended-release Tablets USP, 10 mEq are
tan to yellowish color, oval shaped, biconvex uncoated tablets.Usual Dosage: See package insert for full prescribing
information.Store at 20º to 25ºC (68º to 77ºF); excursions permitted
between 15° to 30°C (59° to 86°F) [see USP Controlled Room
Temperature].Keep this and all drugs out of reach of children.
FOR YOUR PROTECTION: Do not use if blister is torn or
broken.The drug product contained in this package is from
NDC # 68382-537, Zydus Pharmaceuticals (USA) Inc.Distributed by:
American Health Packaging
Columbus, Ohio 43217085032
0285032/0918OS - Package/Label Display Panel – Blister – 10 mEq
-
INGREDIENTS AND APPEARANCE
POTASSIUM CITRATE
potassium citrate tablet, extended releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 68084-850(NDC:68382-537) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POTASSIUM CITRATE (UNII: EE90ONI6FF) (ANHYDROUS CITRIC ACID - UNII:XF417D3PSL) POTASSIUM CITRATE 10 meq Inactive Ingredients Ingredient Name Strength CARNAUBA WAX (UNII: R12CBM0EIZ) MAGNESIUM STEARATE (UNII: 70097M6I30) Product Characteristics Color yellow (tan to yellowish) Score no score Shape OVAL (Oval) Size 19mm Flavor Imprint Code 537 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 68084-850-32 20 in 1 BOX, UNIT-DOSE 09/15/2014 1 NDC: 68084-850-33 1 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA203546 09/15/2014 Labeler - American Health Packaging (929561009) Establishment Name Address ID/FEI Business Operations American Health Packaging 929561009 repack(68084-850)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.