IPRATROPIUM BROMIDE INHALATION aerosol
Ipratropium Bromide Inhalation by
Drug Labeling and Warnings
Ipratropium Bromide Inhalation by is a Prescription medication manufactured, distributed, or labeled by Armstrong Pharmaceuticals, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use IPRATROPIUM BROMIDE HFA INHALATION AEROSOL safely and effectively.
See full prescribing information for IPRATROPIUM BROMIDE HFA INHALATION AEROSOL.
IPRATROPIUM BROMIDE HFA inhalation aerosol, for oral inhalation use
Initial U.S. Approval: 2004INDICATIONS AND USAGE
Ipratropium bromide HFA inhalation aerosol is an anticholinergic indicated for the maintenance treatment of bronchospasm associated with chronic obstructive pulmonary disease (COPD), including chronic bronchitis and emphysema (1)
DOSAGE AND ADMINISTRATION
For oral inhalation only
Two inhalations four times a day, not to exceed 12 inhalations in 24 hours (2)DOSAGE FORMS AND STRENGTHS
CONTRAINDICATIONS
Hypersensitivity to ipratropium bromide or other ipratropium bromide HFA inhalation aerosol components (4)
Hypersensitivity to atropine or any of its derivativesWARNINGS AND PRECAUTIONS
Not indicated for the initial treatment of acute episodes of bronchospasm where rescue therapy is required for rapid response (5.1)
Hypersensitivity reactions including anaphylaxis: Discontinue ipratropium bromide HFA inhalation aerosol at once and consider alternative treatments (5.2)
Paradoxical bronchospasm: Discontinue ipratropium bromide HFA inhalation aerosol and consider other treatments if paradoxical bronchospasm occurs (5.3)
Ocular effects: Use with caution in patients with narrow-angle glaucoma and instruct patients to consult a physician immediately if signs or symptoms of narrow-angle glaucoma develop (5.4)
Urinary retention: Use with caution in patients with prostatic hyperplasia or bladder-neck obstruction and instruct patients to consult a physician immediately if signs or symptoms of urinary retention develop (5.5)ADVERSE REACTIONS
Most common adverse reactions (>5% incidence in the 12-week placebo- controlled trials) were bronchitis, COPD exacerbation, dyspnea, and headache (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Armstrong Pharmaceuticals, Inc. at 1-800-423-4136, or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
Anticholinergics: May interact additively with concomitantly used anticholinergic medications. Avoid administration of ipratropium bromide HFA inhalation aerosol with other anticholinergic-containing drugs (7.1)
Revised: 4/2025
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Use for Maintenance Treatment Only
5.2 Hypersensitivity Reactions, Including Anaphylaxis
5.3 Paradoxical Bronchospasm
5.4 Ocular Effects
5.5 Urinary Retention
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Post-Marketing Experience
7 DRUG INTERACTIONS
7.1 Anticholinergic Agents
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
- * Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
The usual starting dose of ipratropium bromide HFA inhalation aerosol is two inhalations four times a day. Patients may take additional inhalations as required; however, the total number of inhalations should not exceed 12 in 24 hours.
Ipratropium bromide HFA inhalation aerosol is a solution aerosol that does not require shaking. However, as with any other metered-dose inhaler, some coordination is required between actuating the canister and inhaling the medication.
Patients should “prime” or actuate ipratropium bromide HFA inhalation aerosol before using for the first time by releasing 2 test sprays into the air away from the face. In cases where the inhaler has not been used for more than 3 days, prime the inhaler again by releasing 2 test sprays into the air away from the face. Patients should avoid spraying ipratropium bromide HFA inhalation aerosol into their eyes.
Each inhaler provides sufficient medication for 200 actuations. The inhaler should be discarded after the labeled number of actuations has been used. The amount of medication in each actuation cannot be assured after this point, even though the canister is not completely empty.
Patients should be instructed on the proper use of their inhaler [see Patient Counseling Information (17)]. -
3 DOSAGE FORMS AND STRENGTHS
Ipratropium bromide HFA inhalation aerosol is an inhalation aerosol supplied in a pressurized fluorocarbon polymer-coated aluminum canister as a metered-dose inhaler with a blue mouthpiece and a grey protective cap. Each pressurized metered-dose aerosol unit for oral inhalation contains a 12.9 g solution of ipratropium bromide (monohydrate) that provides sufficient medication for 200 actuations. After priming, each actuation of the inhaler delivers 21 mcg of ipratropium bromide (monohydrate) from the valve and delivers 17 mcg of ipratropium bromide (monohydrate) from the mouthpiece.
-
4 CONTRAINDICATIONS
Ipratropium bromide HFA inhalation aerosol is contraindicated in the following conditions [see Warnings and Precautions (5.2)].
Hypersensitivity to ipratropium bromide or other ipratropium bromide HFA inhalation aerosol components
Hypersensitivity to atropine or any of its derivatives -
5 WARNINGS AND PRECAUTIONS
5.1 Use for Maintenance Treatment Only
Ipratropium bromide HFA inhalation aerosol is a bronchodilator for the maintenance treatment of bronchospasm associated with COPD and is not indicated for the initial treatment of acute episodes of bronchospasm where rescue therapy is required for rapid response.
5.2 Hypersensitivity Reactions, Including Anaphylaxis
Hypersensitivity reactions including urticaria, angioedema, rash, bronchospasm, anaphylaxis, and oropharyngeal edema may occur after the administration of ipratropium bromide HFA inhalation aerosol. In clinical trials and postmarketing experience with ipratropium-containing products, hypersensitivity reactions such as skin rash, pruritus, angioedema of tongue, lips and face, urticaria (including giant urticaria), laryngospasm and anaphylactic reactions have been reported [see Adverse Reactions (6.1, 6.2)]. If such a reaction occurs, therapy with ipratropium bromide HFA inhalation aerosol should be stopped at once and alternative treatment should be considered [see Contraindications (4)].
5.3 Paradoxical Bronchospasm
Ipratropium bromide HFA inhalation aerosol can produce paradoxical bronchospasm that can be life threatening. If this occurs, treatment with ipratropium bromide HFA inhalation aerosol should be stopped and other treatments considered.
5.4 Ocular Effects
Ipratropium bromide HFA inhalation aerosol is an anticholinergic and its use may increase intraocular pressure. This may result in precipitation or worsening of narrow-angle glaucoma. Therefore, ipratropium bromide HFA inhalation aerosol should be used with caution in patients with narrow-angle glaucoma [see Drug Interactions (7.1)].
Patients should avoid spraying ipratropium bromide HFA inhalation aerosol into their eyes. If a patient sprays ipratropium bromide HFA inhalation aerosol into their eyes, they may cause eye pain or discomfort, temporary blurring of vision, mydriasis, visual halos or colored images in association with red eyes from conjunctival and corneal congestion. Advise patients to consult their physician immediately if any of these symptoms develop while using ipratropium bromide HFA inhalation aerosol.5.5 Urinary Retention
Ipratropium bromide HFA inhalation aerosol is an anticholinergic and may cause urinary retention. Therefore caution is advised when administering ipratropium bromide HFA inhalation aerosol to patients with prostatic hyperplasia, or bladder-neck obstruction [see Drug Interactions (7.1)].
-
6 ADVERSE REACTIONS
The following adverse reactions are described, or described in greater detail, in other sections:
Hypersensitivity Reactions, Including Anaphylaxis [see Contraindications (4) and Warnings and Precautions (5.2)]
Paradoxical Bronchospasm [see Warnings and Precautions (5.3)]
Ocular Effects [see Warnings and Precautions (5.4)]
Urinary Retention [see Warnings and Precautions (5.5)]
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in patients.6.1 Clinical Trials Experience
The adverse reaction information concerning ipratropium bromide HFA inhalation aerosol is derived from two 12-week, double-blind, parallel-group studies and one 1-year open-label, parallel group study. These studies compared ipratropium bromide HFA inhalation aerosol, ipratropium bromide CFC inhalation aerosol, and placebo (in one study only) in 1,010 COPD patients. The following table lists the incidence of adverse reactions that occurred at a rate of greater than or equal to 3% in any ipratropium bromide group and greater than placebo in the 12-week study. The frequency of corresponding reactions in the 1-year open label study is included for comparison.
TABLE 1 Adverse Reactions (% Patients) in Ipratropium Bromide HFA Inhalation Aerosol Clinical Trials Placebo-controlled 12-week study 244.1405 and Active-controlled 12-week Study 244.1408
Active-controlled 1-year Study 244.2453
Ipratropium Bromide HFA
Inhalation Aerosol
(N=243)
%
Ipratropium Bromide CFC
Inhalation Aerosol
(N=183)
%
Placebo
(N=128)
%
Ipratropium Bromide HFA
Inhalation Aerosol
(N=305)
%
Ipratropium Bromide CFC
Inhalation Aerosol
(N=151)
%
BODY AS A WHOLE - GENERAL DISORDERS Back Pain 2 3 2 7 3 Headache 6 9 8 7 5 Influenza-like symptoms 4 2 2 8 5 CENTRAL & PERIPHERAL NERVOUS SYSTEM DISORDERS Dizziness 3 3 2 3 1 GASTROINTESTINAL SYSTEM DISORDERS Dyspepsia 1 3 1 5 3 Mouth dry 4 2 2 2 3 Nausea 4 1 2 4 4 RESPIRATORY SYSTEM DISORDERS Bronchitis 10 11 6 23 19 COPD exacerbation 8 14 13 23 23 Dyspnea 8 8 4 7 4 Sinusitis 1 4 3 11 14 URINARY SYSTEM DISORDER Urinary tract infection 2 3 1 10 8 Cough, rhinitis, and upper respiratory infection occurred in greater than or equal to 3% of patients in either ipratropium treatment group but not greater than placebo in the 12-week study.
In the one open-label controlled study in 456 COPD patients, the overall incidence of adverse events was also similar between ipratropium bromide HFA inhalation aerosol and ipratropium bromide CFC inhalation aerosol formulations.
Overall, in the above mentioned studies, 9.3% of the patients taking 42 mcg ipratropium bromide HFA inhalation aerosol and 8.7% of the patients taking 42 mcg ipratropium bromide CFC inhalation aerosol reported at least one adverse event that was considered by the investigator to be related to the study drug. The most common drug-related adverse events were dry mouth (1.6% of ipratropium bromide HFA inhalation aerosol and 0.9% of ipratropium bromide CFC inhalation aerosol patients), and taste perversion (bitter taste) (0.9% of ipratropium bromide HFA inhalation aerosol and 0.3% of ipratropium bromide CFC inhalation aerosol patients).
As an anticholinergic drug, cases of precipitation or worsening of narrow-angle glaucoma, glaucoma, halo vision, conjunctival hyperemia, corneal edema, mydriasis, acute eye pain, dry throat, hypotension, palpitations, urinary retention, tachycardia, constipation, bronchospasm, including paradoxical bronchospasm have been reported with the use of ipratropium bromide HFA inhalation aerosol. Additional adverse reactions identified for ipratropium bromide HFA inhalation aerosol seen in clinical trials include throat irritation, stomatitis, mouth edema, and vision blurred.
Allergic-type reactions such as skin rash, pruritus, angioedema including that of tongue, lips and face, urticaria (including giant urticaria), laryngospasm and anaphylactic reactions have been reported [see Warnings and Precautions (5.2)].6.2 Post-Marketing Experience
In a 5-year, placebo-controlled trial, hospitalizations for supraventricular tachycardia and/or atrial fibrillation occurred with an incidence rate of 0.5% in COPD patients receiving ipratropium bromide CFC inhalation aerosol.
In addition to the adverse reactions reported in the controlled clinical trials, adverse reactions have been identified during post-approval use of ipratropium bromide. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Allergic-type reactions such as skin rash, angioedema including that of tongue, lips and face, urticaria (including giant urticaria), laryngospasm, and anaphylactic reactions have been reported, with positive rechallenge in some cases.
Additionally, urinary retention, mydriasis, gastrointestinal distress (diarrhea, nausea, vomiting), cough and bronchospasm, including paradoxical bronchospasm, hypersensitivity reactions, intraocular pressure increased, accommodation disorder, heart rate increased, pharyngeal edema, and gastrointestinal motility disorders have been reported during the post-marketing period with use of ipratropium bromide. -
7 DRUG INTERACTIONS
Ipratropium bromide HFA inhalation aerosol has been used concomitantly with other drugs, including sympathomimetic bronchodilators, methylxanthines, oral and inhaled steroids commonly used in the treatment of COPD. With the exception of albuterol, there are no formal studies fully evaluating the interaction effects of ipratropium bromide HFA inhalation aerosol and these drugs with respect to safety and effectiveness.
7.1 Anticholinergic Agents
There is potential for an additive interaction with concomitantly used anticholinergic medications. Therefore, avoid coadministration of ipratropium bromide HFA inhalation aerosol with other anticholinergic-containing drugs as this may lead to an increase in anticholinergic adverse effects [see Warnings and Precautions (5.4, 5.5)].
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Ipratropium is negligibly absorbed systemically following oral inhalation; therefore, maternal use is not expected to result in fetal exposure to the drug [see Clinical Pharmacology (12.3)]. There is limited experience with ipratropium bromide use in pregnant women. Published literature, including cohort studies, case control studies and case series, over several decades have not identified a drug associated risk of major birth defects, miscarriage or adverse maternal or fetal outcomes. Based on animal reproduction animal studies, no evidence of structural alternations was observed when ipratropium bromide was administered to pregnant mice, rats and rabbits during organogenesis at doses up to approximately 200, 40,000, and 10,000 times, respectively, the maximum recommended human daily inhalation dose (MRHDID) in adults (see Data).
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.Data
Animal Data
In animal reproduction studies, oral and inhalation administration of ipratropium bromide to pregnant mice, rats, and rabbits during the period of organogenesis did not show evidence of fetal structural alterations. The ipratropium bromide dose in oral studies in mice, rats, and rabbits was up to approximately 200, 40,000, and 10,000 times, respectively, the MRHDID in adults (on a mg/m2 basis at maternal doses of 10, 1000, and 125 mg/kg/day, respectively). The ipratropium bromide dose in inhalation studies in rats and rabbits was up to approximately 60 and 140 times, respectively, the MRHDID in adults (on a mg/m2 basis at maternal doses of 1.5 and 1.8 mg/kg/day, respectively). Embryotoxicity was observed as increased resorption in rats at oral doses approximately 3,600 times the MRHDID in adults (on a mg/m2 basis at maternal doses of 90 mg/kg/day and above). This effect is not considered relevant to human use due to the large doses at which it was observed and the difference in route of administration.
8.2 Lactation
Risk Summary
There are no data on the presence of ipratropium in either human or animal milk, the effects on the breastfed infant, or the effects on milk production. Although lipid-insoluble quaternary cations pass into breast milk, ipratropium concentrations in plasma after inhaled therapeutic doses are low, therefore, ipratropium levels in human breast milk are expected to be low [see Clinical Pharmacology (12.3)]. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for ipratropium bromide HFA inhalation aerosol and any potential adverse effects on the breastfed child from ipratropium bromide HFA inhalation aerosol or from the underlying maternal condition.
8.5 Geriatric Use
In the pivotal 12-week study, both ipratropium bromide HFA inhalation aerosol and ipratropium bromide CFC inhalation aerosol formulations were equally effective in patients over 65 years of age and under 65 years of age. Of the total number of subjects in clinical studies of ipratropium bromide HFA inhalation aerosol, 57% were ≥65 years of age. No overall differences in safety or effectiveness were observed between these subjects and younger subjects.
- 10 OVERDOSAGE
-
11 DESCRIPTION
The active ingredient in ipratropium bromide HFA inhalation aerosol is ipratropium bromide (monohydrate). It is an anticholinergic bronchodilator chemically described as 8-azoniabicyclo[3.2.1]octane, 3-(3-hydroxy-1-oxo-2-phenylpropoxy)-8-methyl-8-(1-methylethyl)-,bromide monohydrate, (3-endo, 8-syn)-: a synthetic quaternary ammonium compound, chemically related to atropine. The structural formula for ipratropium bromide is:
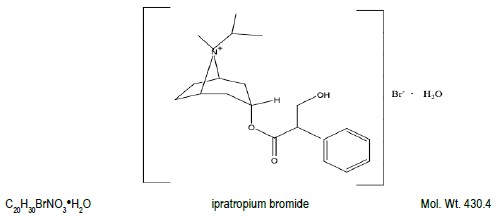
Ipratropium bromide is a white to off-white crystalline substance, freely soluble in water and methanol, sparingly soluble in ethanol, and insoluble in lipophilic solvents such as ether, chloroform, and fluorocarbons.
Ipratropium bromide HFA inhalation aerosol is a pressurized metered-dose aerosol unit for oral inhalation that contains a solution of ipratropium bromide. The 200 inhalation unit has a net weight of 12.9 grams. After priming, each actuation of the inhaler delivers 21 mcg of ipratropium bromide (monohydrate) from the valve in 56 mg of solution and delivers 17 mcg of ipratropium bromide (monohydrate) from the mouthpiece. The actual amount of drug delivered to the lung may depend on patient factors, such as the coordination between the actuation of the device and inspiration through the delivery system. The excipients are anhydrous citric acid, dehydrated alcohol, HFA-134a (1,1,1,2-tetrafluoroethane) as propellant, and sterile water for irrigation. This product does not contain chlorofluorocarbons (CFCs) as propellants.
Ipratropium bromide HFA inhalation aerosol should be primed before using for the first time by releasing 2 test sprays into the air away from the face. In cases where the inhaler has not been used for more than 3 days, prime the inhaler again by releasing 2 test sprays into the air away from the face. -
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Ipratropium bromide is an anticholinergic (parasympatholytic) agent which, based on animal studies, appears to inhibit vagally-mediated reflexes by antagonizing the action of acetylcholine, the transmitter agent released at the neuromuscular junctions in the lung. Anticholinergics prevent the increases in intracellular concentration of Ca++ which is caused by interaction of acetylcholine with the muscarinic receptors on bronchial smooth muscle.
12.2 Pharmacodynamics
Cardiovascular effects
At recommended doses, ipratropium bromide does not produce clinically significant changes in pulse rate or blood pressure.
12.3 Pharmacokinetics
Following administration by oral inhalation from a metered-dose inhaler, the majority of the delivered dose is deposited in the gastrointestinal tract and, to a lesser extent, in the lung, the intended site of action. Ipratropium bromide is a quaternary amine and hence is not readily absorbed into the systemic circulation either from the surface of the lung or from the gastrointestinal tract as confirmed by blood level and renal excretion studies.
The half-life of elimination is about 2 hours after inhalation or intravenous administration. Ipratropium bromide is minimally bound (0% to 9% in vitro) to plasma albumin and α1-acid glycoprotein. It is partially metabolized to inactive ester hydrolysis products. Following intravenous administration, approximately one-half of the dose is excreted unchanged in the urine.
A pharmacokinetic study with 29 chronic obstructive pulmonary disease (COPD) patients (48-79 years of age) demonstrated that mean peak plasma ipratropium concentrations of 59±20 pg/mL were obtained following a single administration of 4 inhalations of ipratropium bromide HFA inhalation aerosol (84 mcg). Plasma ipratropium concentrations declined to 24±15 pg/mL by six hours. When these patients were administered 4 inhalations QID (16 inhalations/day=336 mcg) for one week, the mean peak plasma ipratropium concentration increased to 82±39 pg/mL with a trough (6 hour) concentration of 28±12 pg/mL at steady state.Specific Populations
Geriatric Patients
In the pharmacokinetic study with 29 COPD patients, a subset of 14 patients were >65 years of age. Mean peak plasma ipratropium concentrations of 56±24 pg/mL were obtained following a single administration of 4 inhalations (21 mcg/puff) of ipratropium bromide HFA inhalation aerosol (84 mcg). When these 14 patients were administered 4 inhalations four times a day (16 inhalations/day) for one week, the mean peak plasma ipratropium concentration only increased to 84±50 pg/mL indicating that the pharmacokinetic behavior of ipratropium bromide in the geriatric population is consistent with younger patients.
Renally Impaired Patients
The pharmacokinetics of ipratropium bromide HFA inhalation aerosol have not been studied in patients with renal insufficiency.
Hepatically Impaired Patients
The pharmacokinetics of ipratropium bromide HFA inhalation aerosol have not been studied in patients with hepatic insufficiency.
Drug-Drug Interaction
No specific pharmacokinetic studies were conducted to evaluate potential drug-drug interactions with other medications.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Two-year oral carcinogenicity studies in rats and mice have revealed no carcinogenic activity at doses up to 6 mg/kg (approximately 240 and 120 times the maximum recommended human daily inhalation dose (MRHDID) in adults on a mg/m2 basis, respectively). Results of various mutagenicity/clastogenicity studies (Ames test, mouse dominant lethal test, mouse micronucleus test and chromosome aberrations of bone marrow in Chinese hamsters) were negative.
Fertility of male or female rats at oral doses up to 50 mg/kg (approximately 2000 times the MRHDID in adults on a mg/m2 basis) was unaffected by ipratropium bromide administration. At an oral dose of 500 mg/kg (approximately 20,000 times the MRHDID in adults on a mg/m2 basis), ipratropium bromide produced a decrease in the conception rate. -
14 CLINICAL STUDIES
Conclusions regarding the efficacy of ipratropium bromide HFA inhalation aerosol were derived from two randomized, double-blind, controlled clinical studies. These studies enrolled males and females ages 40 years and older, with a history of COPD, a smoking history of >10 pack-years, an FEV1 <65% and an FEV1/FVC <70%.
One of the studies was a 12-week randomized, double-blind active, and placebo-controlled study in which 505 of the 507 randomized COPD patients were evaluated for the safety and efficacy of 42 mcg (n=124) and 84 mcg (n=126) ipratropium bromide HFA inhalation aerosol in comparison to 42 mcg (n=127) ipratropium bromide CFC inhalation aerosol and their respective placebos (HFA n=62, CFC n=66). Data for both placebo HFA and placebo CFC were combined in the evaluation.
Serial FEV1 (shown in Figure 1, below, as means adjusted for center and baseline effects on test day 1 and test day 85 (primary endpoint)) demonstrated that 1 dose (2 inhalations/21 mcg each) of ipratropium bromide HFA inhalation aerosol produced significantly greater improvement in pulmonary function than placebo. During the six hours immediately post-dose on day 1, the average hourly improvement in adjusted mean FEV1 was 0.148 liters for ipratropium bromide HFA inhalation aerosol (42 mcg) and 0.013 liters for placebo. The mean peak improvement in FEV1, relative to baseline, was 0.295 liters, compared to 0.138 liters for placebo. During the six hours immediately post-dose on day 85, the average hourly improvement in adjusted mean FEV1 was 0.141 liters for ipratropium bromide HFA inhalation aerosol (42 mcg) and 0.014 liters for placebo. The mean peak improvement in FEV1, relative to baseline, was 0.295 liters, compared to 0.140 liters for placebo.
Ipratropium bromide HFA inhalation aerosol (42 mcg) was shown to be clinically comparable to ipratropium bromide CFC inhalation aerosol (42 mcg).Figure 1 Day 1 and Day 85 (Primary Endpoint) Results
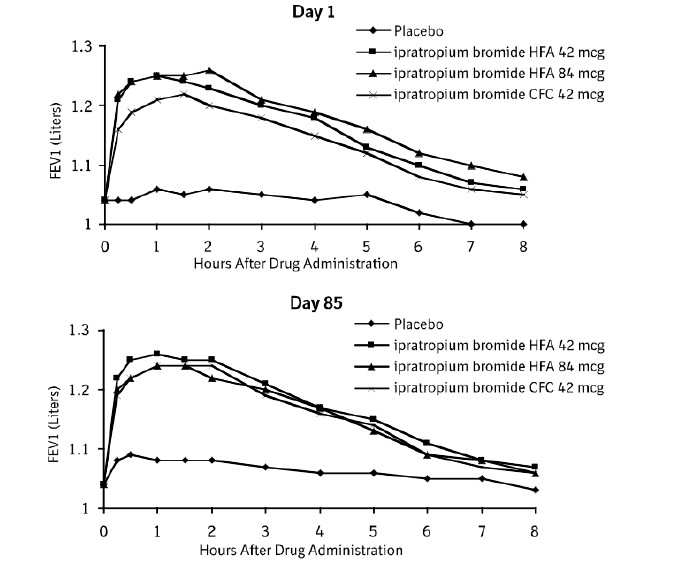
In this study, both ipratropium bromide HFA inhalation aerosol and ipratropium bromide CFC inhalation aerosol formulations were equally effective in patients over 65 years of age and under 65 years of age.
The median time to improvement in pulmonary function (FEV1 increase of 15% or more) was within approximately 15 minutes, reached a peak in 1 to 2 hours, and persisted for 2 to 4 hours in the majority of the patients. Improvements in Forced Vital Capacity (FVC) were also demonstrated.
The other study was a 12-week, randomized, double-blind, active-controlled clinical study in 174 adults with COPD, in which ipratropium bromide HFA inhalation aerosol 42 mcg (n=118) was compared to ipratropium bromide CFC inhalation aerosol 42 mcg (n=56). Safety and efficacy of HFA and CFC formulations were shown to be comparable.
The bronchodilatory efficacy and comparability of ipratropium bromide HFA inhalation aerosol vs ipratropium bromide CFC inhalation aerosol were also studied in a one-year open-label safety and efficacy study in 456 COPD patients. The safety and efficacy of HFA and CFC formulations were shown to be comparable. -
16 HOW SUPPLIED/STORAGE AND HANDLING
Ipratropium bromide HFA inhalation aerosol is supplied in a pressurized aluminum canister as a metered-dose inhaler with a blue mouthpiece and a grey protective cap (NDC: 17270-0401-0). The mouthpiece has an actuation indicator visible through a small window. The indicator typically moves during every 5 to 7 actuations. It displays the approximate number of actuations remaining in increments of 20, starting at “200” and decreasing until it reaches “0”.
The ipratropium bromide HFA inhalation aerosol canister is to be used only with the accompanying ipratropium bromide HFA inhalation aerosol mouthpiece. This mouthpiece should not be used with other aerosol medications. Similarly, the canister should not be used with other mouthpieces. After priming, each actuation of ipratropium bromide HFA inhalation aerosol delivers 21 mcg of ipratropium bromide (monohydrate) from the valve and 17 mcg from the mouthpiece. Each canister has a net weight of 12.9 grams and provides sufficient medication for 200 actuations. The inhaler should be discarded after the labeled number of actuations has been used when the indicator displays “0”. The amount of medication in each actuation cannot be assured after this point, even though the canister is not completely empty.
Storage
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. For optimal results, the canister should be at room temperature before use.
Contents Under Pressure: Do not puncture. Do not use or store near heat or open flame. Exposure to temperatures above 120°F may cause bursting. Never throw the inhaler into a fire or incinerator.
Keep out of reach of children. Avoid spraying in eyes. -
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Instructions for Use).
Appropriate and safe use of ipratropium bromide HFA inhalation aerosol includes providing the patient with the information listed below and an understanding of the way it should be administered.Advise patients that ipratropium bromide HFA inhalation aerosol is a bronchodilator for the maintenance treatment of bronchospasm associated with COPD and is not indicated for the initial treatment of acute episodes of bronchospasm where rescue therapy is required for rapid response.
Hypersensitivity Reactions
Inform patients that hypersensitivity reactions, including urticaria, angioedema, rash, bronchospasm, anaphylaxis, and oropharyngeal edema, may occur after the administration of ipratropium bromide HFA inhalation aerosol. Advise patients to immediately discontinue ipratropium bromide HFA inhalation aerosol and consult a physician [see Warnings and Precautions (5.2)].
Paradoxical Bronchospasm
Inform patients that ipratropium bromide HFA inhalation aerosol can produce paradoxical bronchospasm that can be life-threatening. If paradoxical bronchospasm occurs, patients should discontinue using ipratropium bromide HFA inhalation aerosol.
Ocular Effects
Caution patients to avoid spraying the aerosol into their eyes and be advised that this may result in precipitation or worsening of narrow-angle glaucoma, mydriasis, increased intraocular pressure, acute eye pain or discomfort, temporary blurring of vision, visual halos or colored images in association with red eyes from conjunctival and corneal congestion. Patients should also be advised that should any combination of these symptoms develop, they should consult their physician immediately.
Since dizziness, accommodation disorder, mydriasis, and blurred vision may occur with use of ipratropium bromide HFA inhalation aerosol, patients should be cautioned about engaging in activities requiring balance and visual acuity such as driving a car or operating appliances or machinery.
Urinary Retention
Inform patients that ipratropium bromide HFA inhalation aerosol may cause urinary retention and should be advised to consult their physicians if they experience difficulty with urination.
Frequency of Use
The action of ipratropium bromide HFA inhalation aerosol should last 2 to 4 hours. Advise patients not to increase the dose or frequency of ipratropium bromide HFA inhalation aerosol without patients consulting their physician. Advise patients to seek immediate medical attention if treatment with ipratropium bromide HFA inhalation aerosol becomes less effective for symptomatic relief, their symptoms become worse, and/or patients need to use the product more frequently than usual.
Concomitant Drug Use
Advise patients on the use of ipratropium bromide HFA inhalation aerosol in relation to other inhaled drugs [see Drug Interactions (7.1)].
Use Only as Prescribed
Remind patients that ipratropium bromide HFA inhalation aerosol should be used consistently as prescribed throughout the course of therapy.
Preparation for Use and Priming
Instruct patients that priming ipratropium bromide HFA inhalation aerosol is essential to ensure appropriate content of the medication in each actuation. Patients do not have to shake the ipratropium bromide HFA inhalation aerosol canister before use.
Distributed by:
Armstrong Pharmaceuticals, Inc.
Canton, Massachusetts, U.S.A.
F0401C -
INSTRUCTIONS FOR USEIpratropium Bromide HFAInhalation Aerosol, for oral inhalation use
Read this Instructions for Use before using your ipratropium bromide HFA inhalation aerosol and each time you get a refill. There may be new information. This leaflet does not take the place of talking with your healthcare provider about your medical condition or your treatment.
Use ipratropium bromide HFA inhalation aerosol exactly as your healthcare provider tells you to. Do not change your dose or how often you use ipratropium bromide HFA inhalation aerosol without talking with your healthcare provider.
Tell your doctor about all the medicines you take. Ipratropium bromide HFA inhalation aerosol may affect the way some other medicines work and some other medicines may affect the way ipratropium bromide HFA inhalation aerosol works.
Important information about using ipratropium bromide HFA inhalation aerosol
You do not have to shake ipratropium bromide HFA inhalation aerosol before using it.
Ipratropium bromide HFA inhalation aerosol should be “primed” 2 times before you use the first dose of a new ipratropium bromide HFA inhalation aerosol inhaler or when the inhaler has not been used for more than 3 days.
o To prime, push the canister against the mouthpiece (see Figure 1), allowing the medicine to spray into the air.
o Do not spray the medicine into your eyes while priming ipratropium bromide HFA inhalation aerosol.Inhaler Description
Ipratropium bromide HFA inhalation aerosol (Figure 1) consists of a metal canister containing the medicine and a mouthpiece that releases the medicine from the canister. The mouthpiece includes a blue plastic portion and a grey protective dust cap.
The inhaler comes with a dose indicator you can see through a small window on the plastic mouthpiece (see Figure 1). A new inhaler first shows “200” in the dose indicator window. The dose indicator will show the approximate number of actuations (sprays) of medicine remaining in the inhaler. As you use the inhaler, the dose indicator will typically rotate during every 5 to 7 actuations (sprays) towards the next decreasing number (see Figure 2).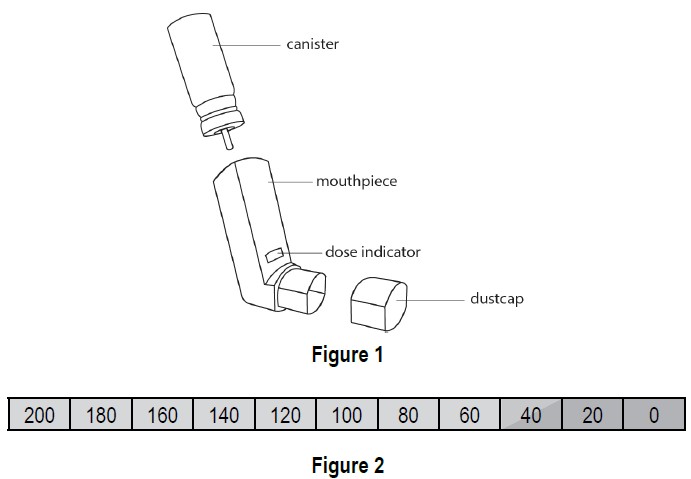
Instructions for Use:
1. Insert the metal canister into the end of the mouthpiece (see Figure 1).
Make sure the canister is fully and firmly inserted into the mouthpiece.The ipratropium bromide HFA inhalation aerosol canister is to be used only with the ipratropium bromide HFA inhalation aerosol mouthpiece.
Do not use the ipratropium bromide HFA inhalation aerosol mouthpiece with other inhaled medicines.
2. Remove the grey protective dust cap. If the cap is not on the mouthpiece, make sure there is nothing in the mouthpiece before use. For best results, the canister should be at room temperature before use.
3. Breathe out (exhale) deeply through your mouth. Hold the inhaler upright (see Figure 3). Put the mouthpiece in your mouth and close your lips.
Keep your eyes closed so that no medicine will be sprayed into your eyes. If sprayed into the eyes, ipratropium bromide HFA inhalation aerosol can cause blurry vision and other vision abnormalities, eye pain or discomfort, dilated pupils, or narrow-angle glaucoma or worsening of this condition. If any combination of these symptoms develops, you should consult your physician immediately.
4. Breathe in (inhale) slowly through your mouth and at the same time spray the ipratropium bromide HFA inhalation aerosol into your mouth.
To spray ipratropium bromide HFA inhalation aerosol firmly press the canister against the mouthpiece 1 time (see Figure 4). Keep breathing in deeply.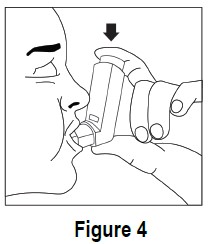
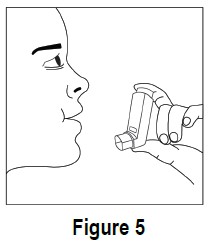
5. Hold your breath for ten seconds and then take the mouthpiece out of your mouth and breathe out slowly (see Figure 5).
6. Wait at least 15 seconds and repeat steps 3 to 5 again.
7. Replace the grey protective dust cap after use.
8. Keep the mouthpiece clean. At least once a week, wash the mouthpiece, shake it to remove excess water and let it air dry all the way (see Mouthpiece Cleaning Instructions).Mouthpiece Cleaning Instructions:
Step A. Remove and set aside the canister and dust cap from the mouthpiece (see Figure 1).
Step B. Wash the mouthpiece through the top and bottom with warm running water for at least 30 seconds (see Figure 6). Do not use anything other than water to wash the mouthpiece.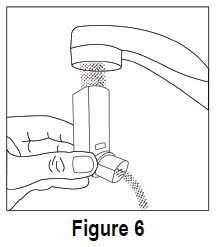 Step C. Dry the mouthpiece by shaking off the excess water and allow it to air dry all the way.
Step C. Dry the mouthpiece by shaking off the excess water and allow it to air dry all the way.
Step D. When the mouthpiece is dry, replace the canister. Make sure the canister is fully and firmly inserted into the mouthpiece.
Step E. Replace the grey protective dust cap.If little or no medicine comes out of the mouthpiece, wash the mouthpiece as described in Steps A to E under the “Mouthpiece Cleaning Instructions”.
9. When to get a new ipratropium bromide HFA inhalation aerosol inhaler.
There are approximately 40 actuations (sprays) left when the dose indicator displays “40,” where the background changes from green to red (see Figure 7a). This is when you need to refill your prescription or ask your doctor if you need another prescription for ipratropium bromide HFA inhalation aerosol.
The background color will be all red when the indicator approaches 20. The indicator will stop moving at “0”. Discard the inhaler once the dose indicator displays “0” (see Figure 7b). Even though the canister may not be empty, you cannot be sure of the amount of medicine in each actuation (spray) once the dose indicator displays “0”.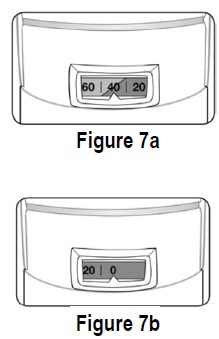
This product does not contain any chlorofluorocarbon (CFC) propellants.
The contents of ipratropium bromide HFA inhalation aerosol are under pressure. Do not puncture the canister. Do not use or store near heat or open flame. Exposure to temperatures above 120°F may cause bursting. Never throw the container into a fire or incinerator.
Keep ipratropium bromide HFA inhalation aerosol and all medicines out of the reach of children.
Store ipratropium bromide HFA inhalation aerosol at room temperature 68°F to 77°F (20°C to 25°C). Short-term exposure to higher or lower temperatures [from 59ºF (15ºC) to 86ºF (30ºC)] is acceptable.For more information about ipratropium bromide HFA inhalation aerosol including current prescribing information and Instructions for Use, go to www.Amphastar.com or call Amphastar Pharmaceuticals, Inc. at 1-800-423-4136.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Distributed by:
Armstrong Pharmaceuticals, Inc.
Canton, Massachusetts U.S.A.Revised: 4/2025 F0401C
-
Carton Label
Principal Display Panel Text:
NDC: 17270-0401-0
Ipratropium
Bromide HFA
Inhalation Aerosol17 mcg/actuation
Each actuation delivers 17 mcg ipratropium bromide (monohydrate) from the mouthpiece.Rx Only
with Dose Indicator
12.9 grams/200 metered actuations
U0401C/4-25

-
INGREDIENTS AND APPEARANCE
IPRATROPIUM BROMIDE INHALATION
ipratropium bromide inhalation aerosolProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 17270-0401 Route of Administration RESPIRATORY (INHALATION) Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength IPRATROPIUM BROMIDE (UNII: J697UZ2A9J) (IPRATROPIUM - UNII:GR88G0I6UL) IPRATROPIUM BROMIDE 17 ug Inactive Ingredients Ingredient Name Strength 1,1,1,2-TETRACHLOROETHANE (UNII: 0S5MKE574X) WATER (UNII: 059QF0KO0R) DEHYDRATED ALCOHOL (UNII: 3K9958V90M) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 17270-0401-0 1 in 1 CARTON 04/16/2026 1 200 in 1 CANISTER; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA217953 04/16/2026 Labeler - Armstrong Pharmaceuticals, Inc. (809773794) Establishment Name Address ID/FEI Business Operations Armstrong Pharmaceuticals, Inc. 809773794 analysis(17270-0401) , distribute(17270-0401) , label(17270-0401) , manufacture(17270-0401) , pack(17270-0401)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.