EYE ALLERGY ITCH RELIEF- olopatadine hcl solution/ drops
Eye Allergy Itch Relief by
Drug Labeling and Warnings
Eye Allergy Itch Relief by is a Otc medication manufactured, distributed, or labeled by CARDINAL HEALTH 110, LLC. DBA LEADER. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- ACTIVE INGREDIENT
- PURPOSE
- USE
- WARNINGS
- DO NOT USE
- WHEN USING THIS PRODUCT
- STOP USE AND ASK DOCTOR IF
- KEEP OUT OF REACH OF CHILDREN
- DIRECTIONS
- OTHER INFORMATION
- INACTIVE INGREDIENTS
- QUESTIONS?
-
SPL UNCLASSIFIED SECTION
DISTRIBUTED BY:
CARDINAL HEALTH
DUBLIN, OHIO 43017
www.myleader.com
1-800-200-6313
Made in India
Code: AP/DRUGS/103/97
Rev. 11/25
CIN 5659222
©2025 CARDINAL HEALTH, LEADER, and LOGOs are trademarks of Cardinal Health. All other marks are the property of their respective owners.
*This product is not manufactured or distributed by Alcon Laboratories Inc., distributor of Pataday® Once Daily Relief. Pataday® is a registered trademark of Novartis AG.
100% Money Back Guarantee
Return to place of purchase if not satisfied.
-
PRINCIPAL DISPLAY PANEL
Carton Label:
NDC: 70000-0053-1
Original Prescription Strength
Eye Allergy Itch Relief
Olopatadine HCl Opthalmic Solution USP, 0.2%
Antihistamine
Once Daily Relief
Works in Minutes
Once Daily
Sterile
2.5 mL (0.085 fl oz)
For Ages 2 and Older
30 DAY SUPPLY
TAMPER EVIDENT: Packaged with Tamper-Evident bottle cap. Do Not Use if breakable ring is separated or missing.
COMPARE TO PATADAY® ONCE DAILY RELIEF active ingredient*
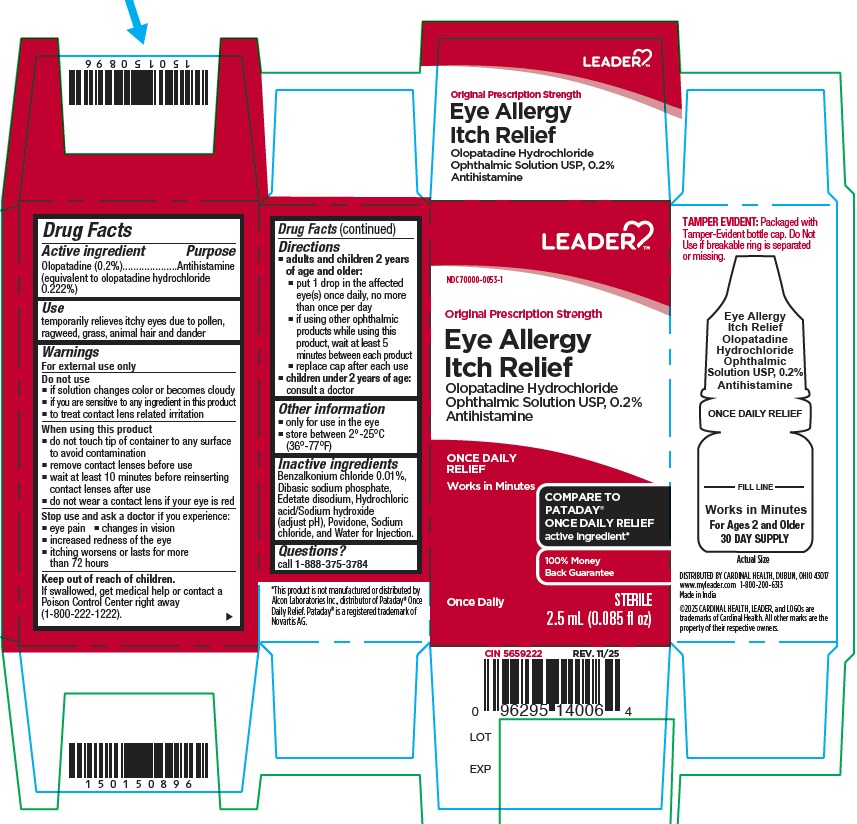
Container Label:
NDC: 70000-0053-1
Eye Allergy Itch Relief
Olopatadine Hydrochloride
Ophthalmic Solution USP, 0.2%
Antihistamine
ONCE DAILY RELIEF
Sterile
2.5 mL (0.085 fl oz)
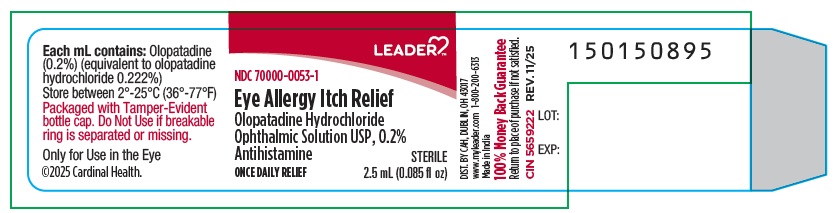
-
INGREDIENTS AND APPEARANCE
EYE ALLERGY ITCH RELIEF
olopatadine hcl solution/ dropsProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 70000-0053 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OLOPATADINE HYDROCHLORIDE (UNII: 2XG66W44KF) (OLOPATADINE - UNII:D27V6190PM) OLOPATADINE 2 mg in 1 mL Inactive Ingredients Ingredient Name Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) SODIUM PHOSPHATE, DIBASIC, ANHYDROUS (UNII: 22ADO53M6F) EDETATE DISODIUM (UNII: 7FLD91C86K) HYDROCHLORIC ACID (UNII: QTT17582CB) POVIDONE K30 (UNII: U725QWY32X) SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM HYDROXIDE (UNII: 55X04QC32I) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 70000-0053-1 1 in 1 CARTON 01/15/2021 1 2.5 mL in 1 BOTTLE, DROPPER; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA209752 01/15/2021 Labeler - CARDINAL HEALTH 110, LLC. DBA LEADER (063997360)