Woosh Antibacterial Pet Wipes by Freshening Paper Products(Hui Zhou)Co.Ltd 84215-002
Woosh Antibacterial Pet Wipes by
Drug Labeling and Warnings
Woosh Antibacterial Pet Wipes by is a Otc medication manufactured, distributed, or labeled by Freshening Paper Products(Hui Zhou)Co.Ltd. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
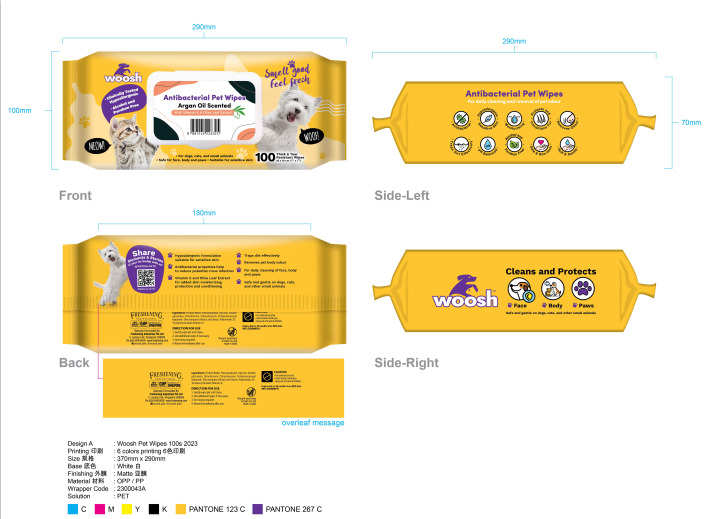
WOOSH ANTIBACTERIAL PET WIPES- wet wipes cloth
Freshening Paper Products(Hui Zhou)Co.Ltd
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
84215-002
Use
Hypoallergenic formulation suitable for sensitive skin.
Traps dirt effectively.
Removes pet body odour.
Antibacterial properties help to reduce potential cross infection.
For daily cleaning of face, body and paws.
Vitamin E and Olive Leaf Extract.
for added skin moisturising,protection and conditioning.
Safe and gentle on dogs, cats,and other small animals.
Directions
Gently wipe pet until clean.
Use additional wipes if necessary.
No rinsing required.
Reseal immediately after use.
| WOOSH ANTIBACTERIAL PET WIPES
wet wipes cloth |
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| Labeler - Freshening Paper Products(Hui Zhou)Co.Ltd (723246894) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Freshening Paper Products(Hui Zhou)Co.Ltd | 723246894 | manufacture(84215-002) | |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.