STAY AWAKE- caffeine tablet
Stay Awake by
Drug Labeling and Warnings
Stay Awake by is a Otc medication manufactured, distributed, or labeled by SUPERVALU INC., LNK International, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Active ingredient (in each tablet)
- Purpose
- Uses
-
Warnings
For occasional use only
Caffeine warning: The recommended dose of this product contains about as much caffeine as a cup of coffee. Limit the use of caffeine-containing medications, foods, or beverages while taking this product because too much caffeine may cause nervousness, irritability, sleeplessness, and, occasionally, rapid heartbeat.
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
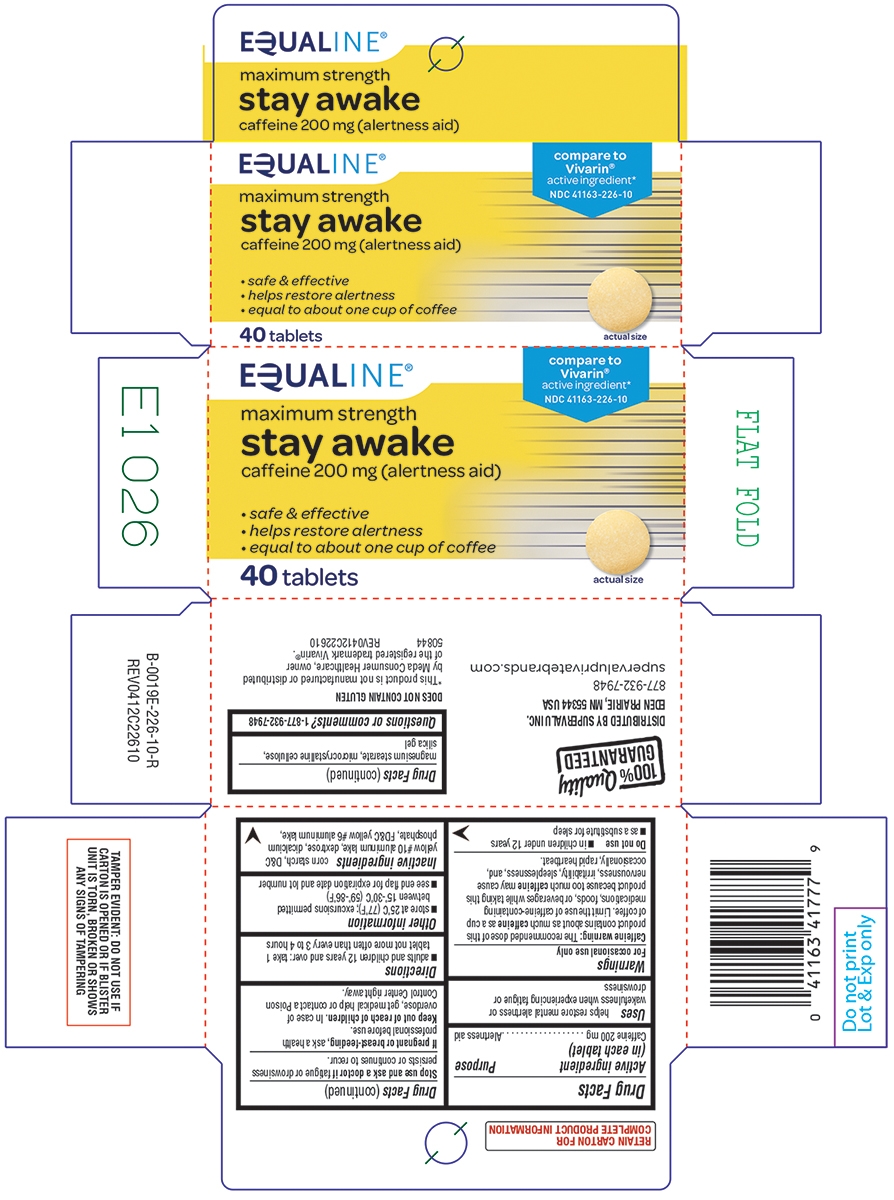
Principal Display Panel
EQUALINE®
compare to
Vivarin®
active ingredient*
NDC: 41163-226-10maximum strength
stay awake
caffeine 200 mg (alertness aid)
helps restore alertness
equal to about one cup of coffee40 tablets
actual size
TAMPER EVIDENT: DO NOT USE IF CARTON IS OPENED OR IF BLISTER UNIT IS TORN, BROKEN OR SHOWS ANY SIGNS OF TAMPERING
DOES NOT CONTAIN GLUTEN
*This product is not manufactured or distributed by Meda AB, owner of the registered trademark Vivarin®.
50844 REV1219D22610DISTRIBUTED BY UNFI
PROVIDENCE, RI 02908 USA855-423-2630
44-226
-
INGREDIENTS AND APPEARANCE
STAY AWAKE
caffeine tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 41163-226 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CAFFEINE (UNII: 3G6A5W338E) (CAFFEINE - UNII:3G6A5W338E) CAFFEINE 200 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS DIBASIC CALCIUM PHOSPHATE (UNII: L11K75P92J) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) STARCH, CORN (UNII: O8232NY3SJ) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) DEXTROSE MONOHYDRATE (UNII: LX22YL083G) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) Product Characteristics Color YELLOW Score no score Shape ROUND Size 11mm Flavor Imprint Code 44;226 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 41163-226-10 5 in 1 CARTON 11/21/1996 1 8 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part340 11/21/1996 Labeler - SUPERVALU INC. (006961411) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 038154464 PACK(41163-226) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867894 MANUFACTURE(41163-226) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 967626305 PACK(41163-226) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867837 PACK(41163-226) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 868734088 PACK(41163-226)
Trademark Results [Stay Awake]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 STAY AWAKE 85047007 not registered Dead/Abandoned |
A.N.N.A. LLC 2010-05-25 |
 STAY AWAKE 76575252 not registered Dead/Abandoned |
Pharmaceutical Formulations, Inc. 2004-02-11 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.