RESPITORY TONIC F (echinacea (angustifolia), uva-ursi, baptisia tinctoria, convallaria majalis, kali muriaticum, pinus sylvestris, thuja occidentalis, bryonia- alba, condurango, crotalus horridus, ginkgo biloba, lachesis mutus, selenium metallicum, tabebuia impetiginosa, viscum album liquid
Respitory Tonic F by
Drug Labeling and Warnings
Respitory Tonic F by is a Homeopathic medication manufactured, distributed, or labeled by BioActive Nutritional, Inc., Apotheca Company. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
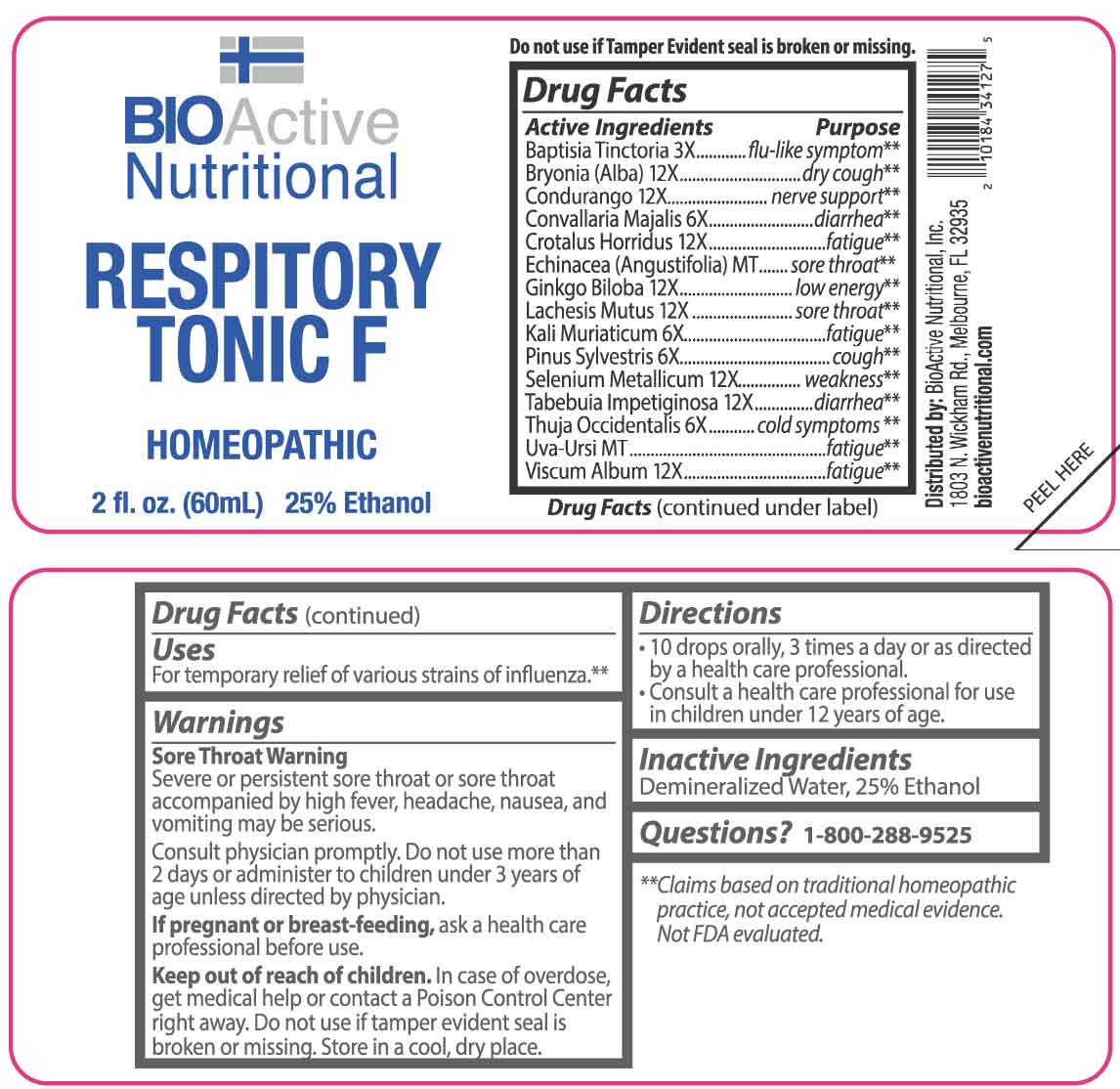
ACTIVE INGREDIENTS:
Baptisia Tinctoria 3X, Bryonia (Alba) 12X, Condurango 12X, Convallaria Majalis 6X, Crotalus Horridus 12X, Echinacea (Angustifolia) MT, Ginkgo Biloba 12X, Lachesis Mutus 12X, Kali Muriaticum 6X, Pinus Sylvestris 6X, Selenium Metallicum 12X, Tabebuia Impetiginosa 12X, Thuja Occidentalis 6X, Uva-Ursi MT, Viscum Album 12X.
-
PURPOSE:
Baptisia Tinctoria – flu-like symptoms,** Bryonia (Alba) – dry cough,** Condurango – nerve support,** Convallaria Majalis - diarrhea,** Crotalus Horridus - fatigue,** Echinacea (Angustifolia) – sore throat,** Ginkgo Biloba – low energy,** Lachesis Mutus – sore throat,** Kali Muriaticum - fatigue,** Pinus Sylvestris - cough,** Selenium Metallicum - weakness,** Tabebuia Impetiginosa - diarrhea,** Thuja Occidentalis – cold symptoms,** Uva-Ursi - fatigue,** Viscum Album – fatigue**
**Claims based on traditional homeopathic practice, not accepted medical evidence. Not FDA evaluated.
- USES:
-
WARNINGS:
Sore Throat Warning
Severe or persistent sore throat or sore throat accompanied by high fever, headache, nausea, and vomiting may be serious.
Consult physician promptly. Do not use more than 2 days or administer to children under 3 years of age unless directed by physician.
If pregnant or breast-feeding, ask a health care professional before use.
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Do not use if tamper evident seal is broken or missing.
Store in a cool, dry place.
- KEEP OUT OF REACH OF CHILDREN:
- DIRECTIONS:
- INACTIVE INGREDIENTS:
- QUESTIONS:
- PACKAGE LABEL DISPLAY:
-
INGREDIENTS AND APPEARANCE
RESPITORY TONIC F
echinacea (angustifolia), uva-ursi, baptisia tinctoria, convallaria majalis, kali muriaticum, pinus sylvestris, thuja occidentalis, bryonia (alba), condurango, crotalus horridus, ginkgo biloba, lachesis mutus, selenium metallicum, tabebuia impetiginosa, viscum album liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 43857-0583 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ECHINACEA ANGUSTIFOLIA WHOLE (UNII: VB06AV5US8) (ECHINACEA ANGUSTIFOLIA - UNII:VB06AV5US8) ECHINACEA ANGUSTIFOLIA WHOLE 1 [hp_X] in 1 mL ARCTOSTAPHYLOS UVA-URSI LEAF (UNII: 3M5V3D1X36) (ARCTOSTAPHYLOS UVA-URSI LEAF - UNII:3M5V3D1X36) ARCTOSTAPHYLOS UVA-URSI LEAF 1 [hp_X] in 1 mL BAPTISIA TINCTORIA ROOT (UNII: 5EF0HWI5WU) (BAPTISIA TINCTORIA ROOT - UNII:5EF0HWI5WU) BAPTISIA TINCTORIA ROOT 3 [hp_X] in 1 mL CONVALLARIA MAJALIS (UNII: QHH4HVF5QE) (CONVALLARIA MAJALIS - UNII:QHH4HVF5QE) CONVALLARIA MAJALIS 6 [hp_X] in 1 mL POTASSIUM CHLORIDE (UNII: 660YQ98I10) (POTASSIUM CATION - UNII:295O53K152) POTASSIUM CATION 6 [hp_X] in 1 mL PINUS SYLVESTRIS LEAFY TWIG (UNII: Q1RGP4UB73) (PINUS SYLVESTRIS LEAFY TWIG - UNII:Q1RGP4UB73) PINUS SYLVESTRIS LEAFY TWIG 6 [hp_X] in 1 mL THUJA OCCIDENTALIS LEAFY TWIG (UNII: 1NT28V9397) (THUJA OCCIDENTALIS LEAFY TWIG - UNII:1NT28V9397) THUJA OCCIDENTALIS LEAFY TWIG 6 [hp_X] in 1 mL BRYONIA ALBA ROOT (UNII: T7J046YI2B) (BRYONIA ALBA ROOT - UNII:T7J046YI2B) BRYONIA ALBA ROOT 12 [hp_X] in 1 mL MARSDENIA CONDURANGO BARK (UNII: R23QIR6YBA) (MARSDENIA CONDURANGO BARK - UNII:R23QIR6YBA) MARSDENIA CONDURANGO BARK 12 [hp_X] in 1 mL CROTALUS HORRIDUS HORRIDUS VENOM (UNII: YHA2XLJ956) (CROTALUS HORRIDUS HORRIDUS VENOM - UNII:YHA2XLJ956) CROTALUS HORRIDUS HORRIDUS VENOM 12 [hp_X] in 1 mL GINKGO (UNII: 19FUJ2C58T) (GINKGO - UNII:19FUJ2C58T) GINKGO 12 [hp_X] in 1 mL LACHESIS MUTA VENOM (UNII: VSW71SS07I) (LACHESIS MUTA VENOM - UNII:VSW71SS07I) LACHESIS MUTA VENOM 12 [hp_X] in 1 mL SELENIUM (UNII: H6241UJ22B) (SELENIUM - UNII:H6241UJ22B) SELENIUM 12 [hp_X] in 1 mL TABEBUIA IMPETIGINOSA BARK (UNII: 6GLA1946WX) (HANDROANTHUS IMPETIGINOSUS BARK - UNII:6GLA1946WX) TABEBUIA IMPETIGINOSA BARK 12 [hp_X] in 1 mL VISCUM ALBUM FRUITING TOP (UNII: BK9092J5MP) (VISCUM ALBUM FRUITING TOP - UNII:BK9092J5MP) VISCUM ALBUM FRUITING TOP 12 [hp_X] in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) ALCOHOL (UNII: 3K9958V90M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 43857-0583-1 60 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product 05/10/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 05/10/2021 Labeler - BioActive Nutritional, Inc. (624980496) Registrant - Apotheca Company (844330915) Establishment Name Address ID/FEI Business Operations Apotheca Company 844330915 manufacture(43857-0583) , api manufacture(43857-0583) , label(43857-0583) , pack(43857-0583)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.