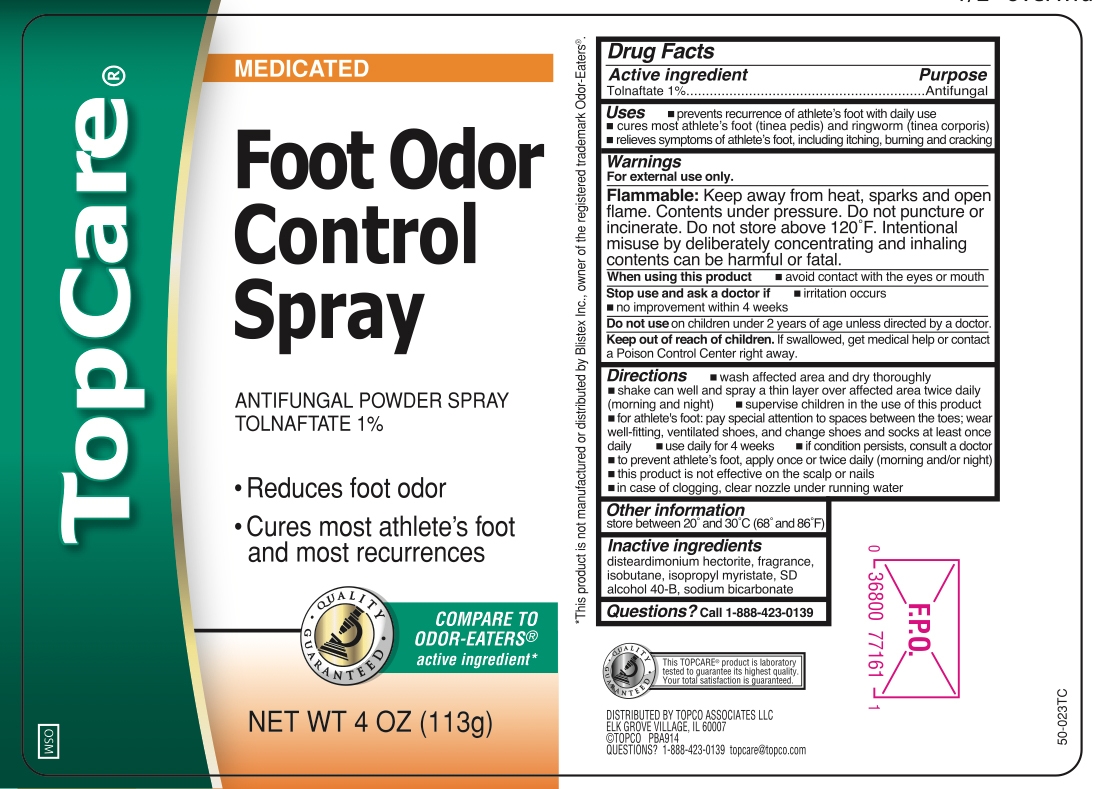
TOLNAFTATE- foot odor control spray aerosol, spray
Tolnaftate by
Drug Labeling and Warnings
Tolnaftate by is a Otc medication manufactured, distributed, or labeled by Topco Associates LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Active ingredient
- Purpose
- Uses
- Warnings
-
Directions
- wash affected area and dry thoroughly
- shake can well and spray a thin layer over affected area twice daily (morning and night)
- supervise children in the use of this product
- for athlete's foot: pay special attention to spaces between the toes, wear well-fitting, ventilated shoes and change shoes and socks at least once daily
- use daily for 4 weeks
- if conditions persist, consult a doctor
- to prevent athlete's foot, apply once or twice daily (morning and/or night)
- in case of clogging, clear nozzle under running water
- Other information
- Inactive ingredients
- Questions?
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
TOLNAFTATE
foot odor control spray aerosol, sprayProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 36800-023 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TOLNAFTATE (UNII: 06KB629TKV) (TOLNAFTATE - UNII:06KB629TKV) TOLNAFTATE 1.13 g in 113 g Inactive Ingredients Ingredient Name Strength DISTEARDIMONIUM HECTORITE (UNII: X687XDK09L) ISOBUTANE (UNII: BXR49TP611) ISOPROPYL MYRISTATE (UNII: 0RE8K4LNJS) ALCOHOL (UNII: 3K9958V90M) SODIUM BICARBONATE (UNII: 8MDF5V39QO) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 36800-023-04 113 g in 1 CAN; Type 0: Not a Combination Product 01/05/2012 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part333C 01/05/2012 Labeler - Topco Associates LLC (006935977)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.