FEXOFENADINE HYDROCHLORIDE tablet, film coated
Fexofenadine hydrochloride by
Drug Labeling and Warnings
Fexofenadine hydrochloride by is a Otc medication manufactured, distributed, or labeled by Amerisource Bergen, Aurolife Pharma, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
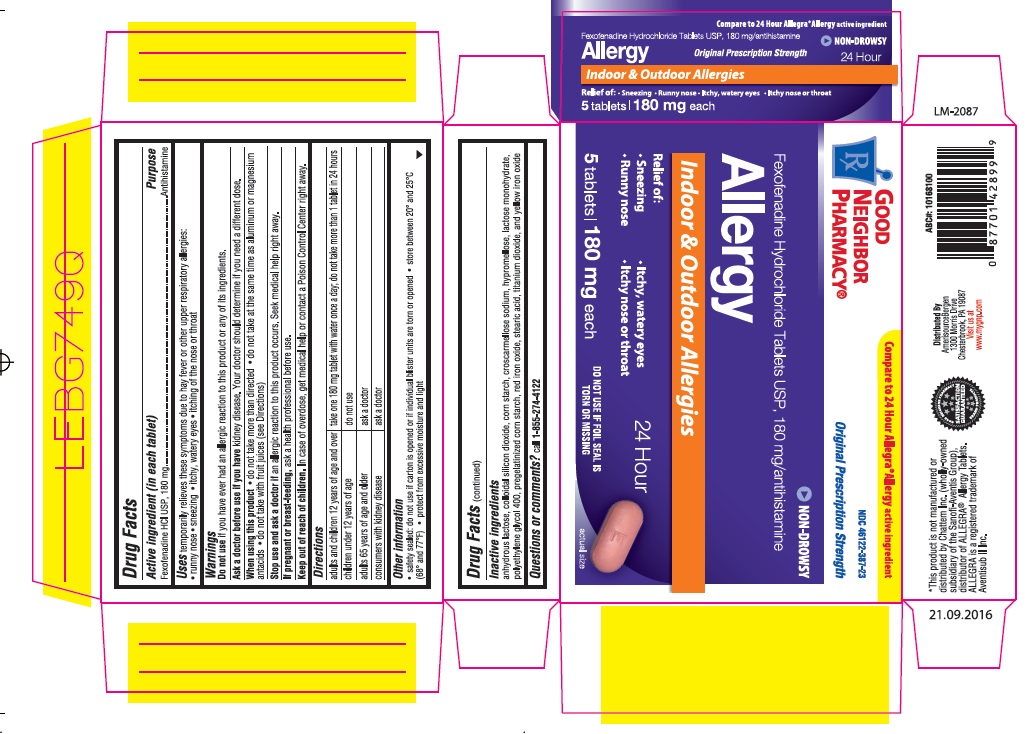
- Active ingredient(in each tablet)
- Purpose
- Uses
- Warnings
- Do not use
- Ask a doctor before use if you have
- When using this product
- Stop use and ask doctor if
- If pregnant or breast-feeding
- Keep out of reach of children
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
Principal Display Panel
NDC: 46122-387-23
Original Prescription Strength
Non-Drowsy
Fexofenadine Hydrochloride Tablets USP, 180 mg/antihistamine
Allergy
Indoor & Outdoor Allergies
Relief of: 24 Hour
Sneezing
Runny nose
Itchy, Watery Eyes
Itchy Nose or Throat
DO NOT USE IF FOIL SEAL IS TORN OR MISSING
5 Tablets 180 mg each
-
INGREDIENTS AND APPEARANCE
FEXOFENADINE HYDROCHLORIDE
fexofenadine hydrochloride tablet, film coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 46122-387 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength FEXOFENADINE HYDROCHLORIDE (UNII: 2S068B75ZU) (FEXOFENADINE - UNII:E6582LOH6V) FEXOFENADINE HYDROCHLORIDE 180 mg Inactive Ingredients Ingredient Name Strength ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) HYPROMELLOSES (UNII: 3NXW29V3WO) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) STARCH, PREGELATINIZED CORN (UNII: O8232NY3SJ) STEARIC ACID (UNII: 4ELV7Z65AP) FERRIC OXIDE RED (UNII: 1K09F3G675) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color ORANGE (Peach) Score no score Shape CAPSULE (Bevel Edge, Biconvex) Size 17mm Flavor Imprint Code E;44 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 46122-387-23 1 in 1 CARTON; Type 0: Not a Combination Product 01/15/2015 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA202039 01/15/2015 Labeler - Amerisource Bergen (007914906) Establishment Name Address ID/FEI Business Operations Aurolife Pharma, LLC 829084461 MANUFACTURE(46122-387)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.