Hydroxychloroquine Sulfate Tablets, USP 200 mg*
Hydroxychloroquine Sulfate by
Drug Labeling and Warnings
Hydroxychloroquine Sulfate by is a Prescription medication manufactured, distributed, or labeled by Aphena Pharma Solutions - Tennessee, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
HYDROXYCHLOROQUINE SULFATE - hydroxychloroquine sulfate tablet, film coated
Aphena Pharma Solutions - Tennessee, LLC
----------
Hydroxychloroquine Sulfate Tablets, USP 200 mg*
PHYSICIANS SHOULD COMPLETELY FAMILIARIZE THEMSELVES WITH THE COMPLETE CONTENTS OF THIS LEAFLET BEFORE PRESCRIBING HYDROXYCHLOROQUINE SULFATE TABLETS, USP.
DESCRIPTION
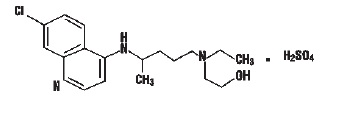
Hydroxychloroquine sulfate, USP is a colorless crystalline solid, soluble in water to at least 20 percent; chemically the drug is 2-[[4-[(7-Chloro-4-quinolyl) amino]pentyl] ethylamino] ethanol sulfate (1:1). Hydroxychloroquine sulfate, USP has the following structural formula:
Molecular Formula: C18H26ClN3O.H2SO4
Each tablet, for oral administration, contains 200 mg hydroxychloroquine sulfate, USP (equivalent to 155 mg base). In addition, each tablet contains the following Inactive Ingredients: colloidal silicon dioxide, dibasic calcium phosphate, hypromellose, macrogol/PEG 3350, magnesium stearate, polysorbate 80, pregelatinized starch, talc and titanium dioxide.
CLINICAL PHARMACOLOGY
The drug possesses antimalarial actions and also exerts a beneficial effect in lupus erythematosus (chronic discoid or systemic) and acute or chronic rheumatoid arthritis. The precise mechanism of action is not known.
INDICATIONS AND USAGE
Hydroxychloroquine sulfate tablets, USP are indicated for the suppressive treatment and treatment of acute attacks of malaria due to Plasmodium vivax, P. malariae, P. ovale, and susceptible strains of P. falciparum. Hydroxychloroquine sulfate tablets, USP are also indicated for the treatment of discoid and systemic lupus erythematosus, and rheumatoid arthritis.
CONTRAINDICATIONS
Use of this drug is contraindicated (1) in the presence of retinal or visual field changes attributable to any 4-aminoquinoline compound, (2) in patients with known hypersensitivity to 4-aminoquinoline compounds, and (3) for long-term therapy in children.
WARNINGS
General
Hydroxychloroquine sulfate is not effective against chloroquine-resistant strains of P. falciparum.
Children are especially sensitive to the 4-aminoquinoline compounds. A number of fatalities have been reported following the accidental ingestion of chloroquine, sometimes in relatively small doses (0.75 g or 1 g in one 3-year-old child). Patients should be strongly warned to keep these drugs out of the reach of children.
Use of hydroxychloroquine sulfate in patients with psoriasis may precipitate a severe attack of psoriasis. When used in patients with porphyria the condition may be exacerbated. The preparation should not be used in these conditions unless in the judgment of the physician the benefit to the patient outweighs the possible hazard.
Usage in Pregnancy
Usage of this drug during pregnancy should be avoided except in the suppression or treatment of malaria when in the judgment of the physician the benefit outweighs the possible hazard. It should be noted that radioactively-tagged chloroquine administered intravenously to pregnant, pigmented CBA mice passed rapidly across the placenta. It accumulated selectively in the melanin structures of the fetal eyes and was retained in the ocular tissues for five months after the drug had been eliminated from the rest of the body.
PRECAUTIONS
General
Antimalarial compounds should be used with caution in patients with hepatic disease or alcoholism or in conjunction with known hepatotoxic drugs.
Periodic blood cell counts should be made if patients are given prolonged therapy. If any severe blood disorder appears which is not attributable to the disease under treatment, discontinuation of the drug should be considered. The drug should be administered with caution in patients having G-6-PD (glucose-6-phosphate dehydrogenase) deficiency.
OVERDOSAGE
The 4-aminoquinoline compounds are very rapidly and completely absorbed after ingestion, and in accidental overdosage, or rarely with lower doses in hypersensitive patients, toxic symptoms may occur within 30 minutes. These consist of headache, drowsiness, visual disturbances, cardiovascular collapse, and convulsions, followed by sudden and early respiratory and cardiac arrest. The electrocardiogram may reveal atrial standstill, nodal rhythm, prolonged intraventricular conduction time, and progressive bradycardia leading to ventricular fibrillation and/or arrest. Treatment is symptomatic and must be prompt with immediate evacuation of the stomach by emesis (at home, before transportation to the hospital) or gastric lavage until the stomach is completely emptied. If finely powdered, activated charcoal is introduced by the stomach tube, after lavage, and within 30 minutes after ingestion of the tablets, it may inhibit further intestinal absorption of the drug. To be effective, the dose of activated charcoal should be at least five times the estimated dose of hydroxychloroquine sulfate tablets that were ingested. Convulsions, if present, should be controlled before attempting gastric lavage. If due to cerebral stimulation, cautious administration of an ultrashort-acting barbiturate may be tried but, if due to anoxia, it should be corrected by oxygen administration, artificial respiration or, in shock with hypotension, by vasopressor therapy. Because of the importance of supporting respiration, tracheal intubation or tracheostomy, followed by gastric lavage, may also be necessary. Exchange transfusions have been used to reduce the level of 4-aminoquinoline drug in the blood.
A patient who survives the acute phase and is asymptomatic should be closely observed for at least six hours. Fluids may be forced, and sufficient ammonium chloride (8 g daily in divided doses for adults) may be administered for a few days to acidify the urine to help promote urinary excretion in cases of both overdosage and sensitivity.
| MALARIA |
Like chloroquine phosphate, USP, hydroxychloroquine sulfate is highly active against the erythrocytic forms of P. vivax and malariae and most strains of P. falciparum (but not the gametocytes of P. falciparum).
Hydroxychloroquine sulfate does not prevent relapses in patients with vivax or malariae malaria because it is not effective against exo-erythrocytic forms of the parasite, nor will it prevent vivax or malariae infection when administered as a prophylactic. It is highly effective as a suppressive agent in patients with vivax or malariae malaria, in terminating acute attacks, and significantly lengthening the interval between treatment and relapse. In patients with falciparum malaria, it abolishes the acute attack and effects complete cure of the infection, unless due to a resistant strain of P. falciparum.
Hydroxychloroquine sulfate tablets, USP are indicated for the treatment of acute attacks and suppression of malaria.
In recent years, it has been found that certain strains of P. falciparum have become resistant to 4-aminoquinoline compounds (including hydroxychloroquine) as shown by the fact that normally adequate doses have failed to prevent or cure clinical malaria or parasitemia. Treatment with quinine or other specific forms of therapy is therefore advised for patients infected with a resistant strain of parasites.
ADVERSE REACTIONS
Following the administration in doses adequate for the treatment of an acute malarial attack, mild and transient headache, dizziness, and gastrointestinal complaints (diarrhea, anorexia, nausea, abdominal cramps and, on rare occasions, vomiting) may occur. Cardiomyopathy has been rarely reported with high daily dosages of hydroxychloroquine sulfate tablets.
DOSAGE AND ADMINISTRATION
One tablet of 200 mg of hydroxychloroquine sulfate, USP is equivalent to 155 mg base.
Malaria: Suppression: In adults, 400 mg (=310 mg base) on exactly the same day of each week. In infants and children, the weekly suppressive dosage is 5 mg, calculated as base, per kg of body weight, but should not exceed the adult dose regardless of weight.
If circumstances permit, suppressive therapy should begin two weeks prior to exposure. However, failing this, in adults an initial double (loading) dose of 800 mg (=620 mg base), or in children 10 mg base/kg may be taken in two divided doses, six hours apart. The suppressive therapy should be continued for eight weeks after leaving the endemic area.
Treatment of the acute attack: In adults, an initial dose of 800 mg (=620 mg base) followed by 400 mg (=310 mg base) in six to eight hours and 400 mg (=310 mg base) on each of two consecutive days (total 2 g hydroxychloroquine sulfate or 1.55 g base). An alternative method, employing a single dose of 800 mg (=620 mg base), has also proved effective.
The dosage for adults may also be calculated on the basis of body weight; this method is preferred for infants and children. A total dose representing 25 mg of base per kg of body weight is administered in three days, as follows:
First dose: 10 mg base per kg (but not exceeding a single dose of 620 mg base).
Second dose: 5 mg base per kg (but not exceeding a single dose of 310 mg base) 6 hours after first dose.
Third dose: 5 mg base per kg 18 hours after second dose.
Fourth dose: 5 mg base per kg 24 hours after third dose.
For radical cure of vivax and malariae malaria concomitant therapy with an 8-aminoquinoline compound is necessary.
| LUPUS ERYTHEMATOSUS AND RHEUMATOID ARTHRITIS |
Hydroxychloroquine sulfate tablets, USP are useful in patients with the following disorders who have not responded satisfactorily to drugs with less potential for serious side effects: lupus erythematosus (chronic discoid and systemic) and acute or chronic rheumatoid arthritis.
PHYSICIANS SHOULD COMPLETELY FAMlLlARlZE THEMSELVES WITH THE COMPLETE CONTENTS OF THIS LEAFLET BEFORE PRESCRlBlNG HYDROXYCHLOROQUINE SULFATE TABLETS, USP.
Irreversible retinal damage has been observed in some patients who had received long-term or high-dosage 4-aminoquinoline therapy for discoid and systemic lupus erythematosus, or rheumatoid arthritis. Retinopathy has been reported to be dose-related.
When prolonged therapy with any antimalarial compound is contemplated, initial (base line) and periodic (every three months) ophthalmologic examinations (including visual acuity, expert slit-lamp, funduscopic, and visual field tests) should be performed.
If there is any indication of abnormality in the visual acuity, visual field, or retinal macular areas (such as pigmentary changes, loss of foveal reflex), or any visual symptoms (such as light flashes and streaks) which are not fully explainable by difficulties of accommodation or corneal opacities, the drug should be discontinued immediately and the patient closely observed for possible progression. Retinal changes (and visual disturbances) may progress even after cessation of therapy.
All patients on long-term therapy with this preparation should be questioned and examined periodically, including the testing of knee and ankle reflexes, to detect any evidence of muscular weakness. If weakness occurs, discontinue the drug.
In the treatment of rheumatoid arthritis, if objective improvement (such as reduced joint swelling, increased mobility) does not occur within six months, the drug should be discontinued. Safe use of the drug in the treatment of juvenile arthritis has not been established.
Dermatologic reactions to hydroxychloroquine sulfate tablets may occur and, therefore, proper care should be exercised when they were administered to any patient receiving a drug with a significant tendency to produce dermatitis.
The methods recommended for early diagnosis of "chloroquine retinopathy" consist of (1) funduscopic examination of the macula for fine pigmentary disturbances or loss of the foveal reflex and (2) examination of the central visual field with a small red test object for pericentral or paracentral scotoma or determination of retinal thresholds to red. Any unexplained visual symptoms, such as light flashes or streaks should also be regarded with suspicion as possible manifestations of retinopathy.
If serious toxic symptoms occur from overdosage or sensitivity, it has been suggested that ammonium chloride (8 g daily in divided doses for adults) be administered orally three or four days a week for several months after therapy has been stopped, as acidification of the urine increases renal excretion of the 4-aminoquinoline compounds by 20 to 90 percent. However, caution must be exercised in patients with impaired renal function and/or metabolic acidosis.
Not all of the following reactions have been observed with every 4-aminoquinoline compound during long-term therapy, but they have been reported with one or more and should be borne in mind when drugs of this class are administered. Adverse effects with different compounds vary in type and frequency.
CNS Reactions: Irritability, nervousness, emotional changes, nightmares, psychosis, headache, dizziness, vertigo, tinnitus, nystagmus, nerve deafness, convulsions, ataxia.
Neuromuscular Reactions: Skeletal muscle palsies or skeletal muscle myopathy or neuromyopathy leading to progressive weakness and atrophy of proximal muscle groups which may be associated with mild sensory changes, depression of tendon reflexes and abnormal nerve conduction.
Ocular Reactions:
- Ciliary body: Disturbance of accommodation with symptoms of blurred vision. This reaction is dose-related and reversible with cessation of therapy.
- Cornea: Transient edema, punctate to lineal opacities, decreased corneal sensitivity. The corneal changes, with or without accompanying symptoms (blurred vision, halos around lights, photophobia), are fairly common, but reversible. Corneal deposits may appear as early as three weeks following initiation of therapy.
The incidence of corneal changes and visual side effects appears to be considerably lower with hydroxychloroquine than with chloroquine.
Macula: Edema, atrophy, abnormal pigmentation (mild pigment stippling to a "bull's-eye" appearance), loss of foveal reflex, increased macular recovery time following exposure to a bright light (photo-stress test), elevated retinal threshold to red light in macular, paramacular, and peripheral retinal areas.
Other fundus changes include optic disc pallor and atrophy, attenuation of retinal arterioles, fine granular pigmentary disturbances in the peripheral retina and prominent choroidal patterns in advanced stage.
D. Visual field defects: Pericentral or paracentral scotoma, central scotoma with decreased visual acuity, rarely field constriction, abnormal color vision.
The most common visual symptoms attributed to the retinopathy are: reading and seeing difficulties (words, letters, or parts of objects missing), photophobia, blurred distance vision, missing or blacked out areas in the central or peripheral visual field, light flashes and streaks.
Retinopathy appears to be dose related and has occurred within several months (rarely) to several years of daily therapy; a small number of cases have been reported several years after antimalarial drug therapy was discontinued. It has not been noted during prolonged use of weekly doses of the 4-aminoquinoline compounds for suppression of malaria.
Patients with retinal changes may have visual symptoms or may be asymptomatic (with or without visual field changes). Rarely scotomatous vision or field defects may occur without obvious retinal change.
Retinopathy may progress even after the drug is discontinued. In a number of patients, early retinopathy (macular pigmentation sometimes with central field defects) diminished or regressed completely after therapy was discontinued. Paracentral scotoma to red targets (sometimes called "premaculopathy") is indicative of early retinal dysfunction which is usually reversible with cessation of therapy.
A small number of cases of retinal changes have been reported as occurring in patients who received only hydroxychloroquine sulfate tablets. These usually consisted of alteration in retinal pigmentation which was detected on periodic ophthalmologic examination; visual field defects were also present in some instances. A case of delayed retinopathy has been reported with loss of vision starting one year after administration of hydroxychloroquine sulfate tablets had been discontinued.
Dermatologic Reactions: Bleaching of hair, alopecia, pruritus, skin and mucosal pigmentation, photosensitivity, and skin eruptions (urticarial, morbilliform, Iichenoid, maculopapular, purpuric, erythema annulare centrifugum, Stevens Johnson-syndrome, acute generalized exanthematous pustulosis, and exfoliative dermatitis).
Hematologic Reactions: Various blood dyscrasias such as aplastic anemia, agranulocytosis, leukopenia, thrombocytopenia (hemolysis in individuals with glucose-6-phosphate dehydrogenase (G-6-PD) deficiency).
Gastrointestinal Reactions: Anorexia, nausea, vomiting, diarrhea, and abdominal cramps. Isolated cases of abnormal liver function and fulminant hepatic failure.
Allergic reactions: Urticaria, angiodema and bronchospasm have been reported.
Miscellaneous Reactions: Weight loss, lassitude, exacerbation or precipitation of porphyria and nonlight-sensitive psoriasis.
Cardiomyopathy has been rarely reported with high daily dosages of hydroxychloroquine sulfate tablets.
One tablet of hydroxychloroquine sulfate, USP 200 mg, is equivalent to 155 mg base.
Lupus erythematosus: Initially, the average adult dose is 400 mg (=310 mg base) once or twice daily. This may be continued for several weeks or months, depending on the response of the patient. For prolonged maintenance therapy, a smaller dose, from 200 mg to 400 mg (=155 mg to 310 mg base) daily will frequently suffice.
The incidence of retinopathy has been reported to be higher when this maintenance dose is exceeded.
Rheumatoid arthritis: The compound is cumulative in action and will require several weeks to exert its beneficial therapeutic effects, whereas minor side effects may occur relatively early. Several months of therapy may be required before maximum effects can be obtained. If objective improvement (such as reduced joint swelling, increased mobility) does not occur within six months, the drug should be discontinued. Safe use of the drug in the treatment of juvenile rheumatoid arthritis has not been established.
Initial dosage - In adults, from 400 mg to 600 mg (=310 mg to 465 mg base) daily, each dose to be taken with a meal or a glass of milk. In a small percentage of patients, troublesome side effects may require temporary reduction of the initial dosage. Later (usually from five to ten days), the dose may gradually be increased to the optimum response level, often without return of side effects.
Maintenance dosage - When a good response is obtained (usually in four to twelve weeks), the dosage is reduced by 50 percent and continued at a usual maintenance level of 200 mg to 400 mg (=155 mg to 310 mg base) daily, each dose to be taken with a meal or a glass of milk. The incidence of retinopathy has been reported to be higher when this maintenance dose is exceeded.
Should a relapse occur after medication is withdrawn, therapy may be resumed or continued on an intermittent schedule if there are no ocular contraindications.
Corticosteroids and salicylates may be used in conjunction with this compound, and they can generally be decreased gradually in dosage or eliminated after the drug has been used for several weeks. When gradual reduction of steroid dosage is indicated, it may be done by reducing every four to five days the dose of cortisone by no more than from 5 mg to 15 mg; of hydrocortisone from 5 mg to 10 mg; of prednisolone and prednisone from 1 mg to 2.5 mg; of methylprednisolone and triamcinolone from 1 mg to 2 mg; and of dexamethasone from 0.25 mg to 0.5 mg.
HOW SUPPLIED
Hydroxychloroquine sulfate tablets, USP are white, to off-white, capsule-shaped tablets, debossed "HCQS" on one side and plain on the reverse side and are available in bottles of 100 and 180. Each tablet contains 200 mg hydroxychloroquine sulfate, USP (equivalent to 155 mg base).
NDC: 42291-318-90 bottles of 90
NDC: 42291-318-01 bottles of 100
NDC: 42291-318-18 bottles of 180
NDC: 42291-318-50 bottles of 500
Dispense in a tight, light-resistant container as defined in the USP/NF.
Store at 20° - 25° C (68° - 77° F) excursions permitted to 15° - 30° C (59° - 86° F) [See USP Controlled Room Temperature].
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
Manufactured for:
AvKARE, Inc.
Pulaski, TN 38478
Mfg. Rev. 11/13
AV Rev. 09/14 (P)
Repackaging Information
Please reference the How Supplied section listed above for a description of individual tablets. This drug product has been received by Aphena Pharma - TN in a manufacturer or distributor packaged configuration and repackaged in full compliance with all applicable cGMP regulations. The package configurations available from Aphena are listed below:
| Count | 200mg |
| 60 | 43353-972-53 |
| 90 | 43353-972-60 |
| 180 | 43353-972-80 |
| 9000 | 43353-972-09 |
Store between 20°-25°C (68°-77°F). See USP Controlled Room Temperature. Dispense in a tight light-resistant container as defined by USP. Keep this and all drugs out of the reach of children.
Repackaged by:

Cookeville, TN 38506
20160621JH
| HYDROXYCHLOROQUINE SULFATE
hydroxychloroquine sulfate tablet, film coated |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Aphena Pharma Solutions - Tennessee, LLC (128385585) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Aphena Pharma Solutions - Tennessee, LLC | 128385585 | REPACK(43353-972) | |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.