Paulas Choice Hydralight Shine Free Daily Mineral Complex by Paula's Choice, LLC. Paula's Choice
Paulas Choice Hydralight Shine Free Daily Mineral Complex by
Drug Labeling and Warnings
Paulas Choice Hydralight Shine Free Daily Mineral Complex by is a Otc medication manufactured, distributed, or labeled by Paula's Choice, LLC.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
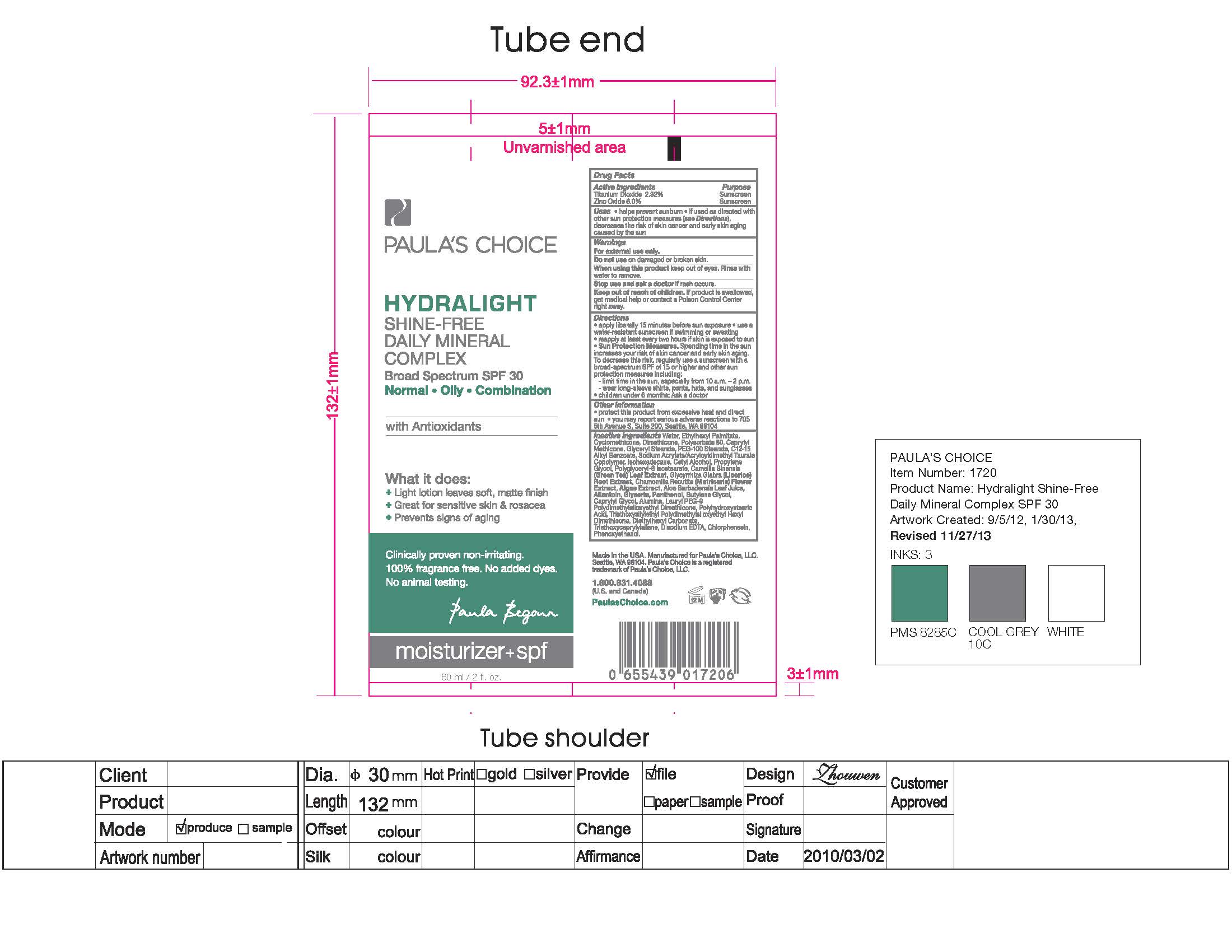
PAULAS CHOICE HYDRALIGHT SHINE FREE DAILY MINERAL COMPLEX- titanium dioxide, zinc oxide lotion
Paula's Choice, LLC.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Paula's Choice
Keep out of reach of children. If product is swallowed, get medical help or contact a Poison Control Center right away.
Directions
Apply liberally 15 minutes before sun exposure. Use a water resistant sunscreen if swimming or sweating. Reapply at least every two hours if skin is exposed to sun.
Sun Protection Measures Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a broad-spectrum SPF of 15 or higher and other sun protection measures including:
- Limit time in the sun, especially from 10 a.m. - 2 p.m.
- Wear long-sleeve shirts, pants, hats, and sunglasses
- Children under 6 months: ask a doctor
Protect this product from excessive heat and direct sun. You may report serious adverse reaction to 705 5th Avenue, South, Suite 200, Seattle, WA 98104
Water, Ethylhexyl Palmitate, Cyclomethicone, Dimethicone, Polysorbate 80, Caprylyl Methicone, Glyceryl Stearate, PEG-100 Stearate, C12-15 Alkyl Benzoate, Sodium Acrylate/Acryloyldimethyl Taurate Copolymer, Isohexadecane, Cetyl Alcohol, Propylene Glycol, Polyglyceryl-6 Isostearate, Camellia Sinensis (Green Tea) Leaf Extract, Glycyrrhiza Glabra (Licorice) Root Extract, Chamomilla Recutita (Matricaria) Flower Extract, Algae Extract, Aloe Barbadensis Leaf Juice, Allantoin, Glycerin, Panthenol, Butylene Glycol, Caprylyl Glycol, Alumina, Lauryl PEG-9 Polydimethylsiloxyethyl Dimethicone, Polyhydroxystearic Acid, Triethoxysilylethyl Polydimethylsiloxyethyl Hexyl Dimethicone, Diethylhexyl Carbonate, Triethoxyxaprylylsilane, Disodium EDTA, Chlorphenesin, Phenoxyethanol
| PAULAS CHOICE HYDRALIGHT SHINE FREE DAILY MINERAL COMPLEX
titanium dioxide, zinc oxide lotion |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - Paula's Choice, LLC. (029583981) |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.