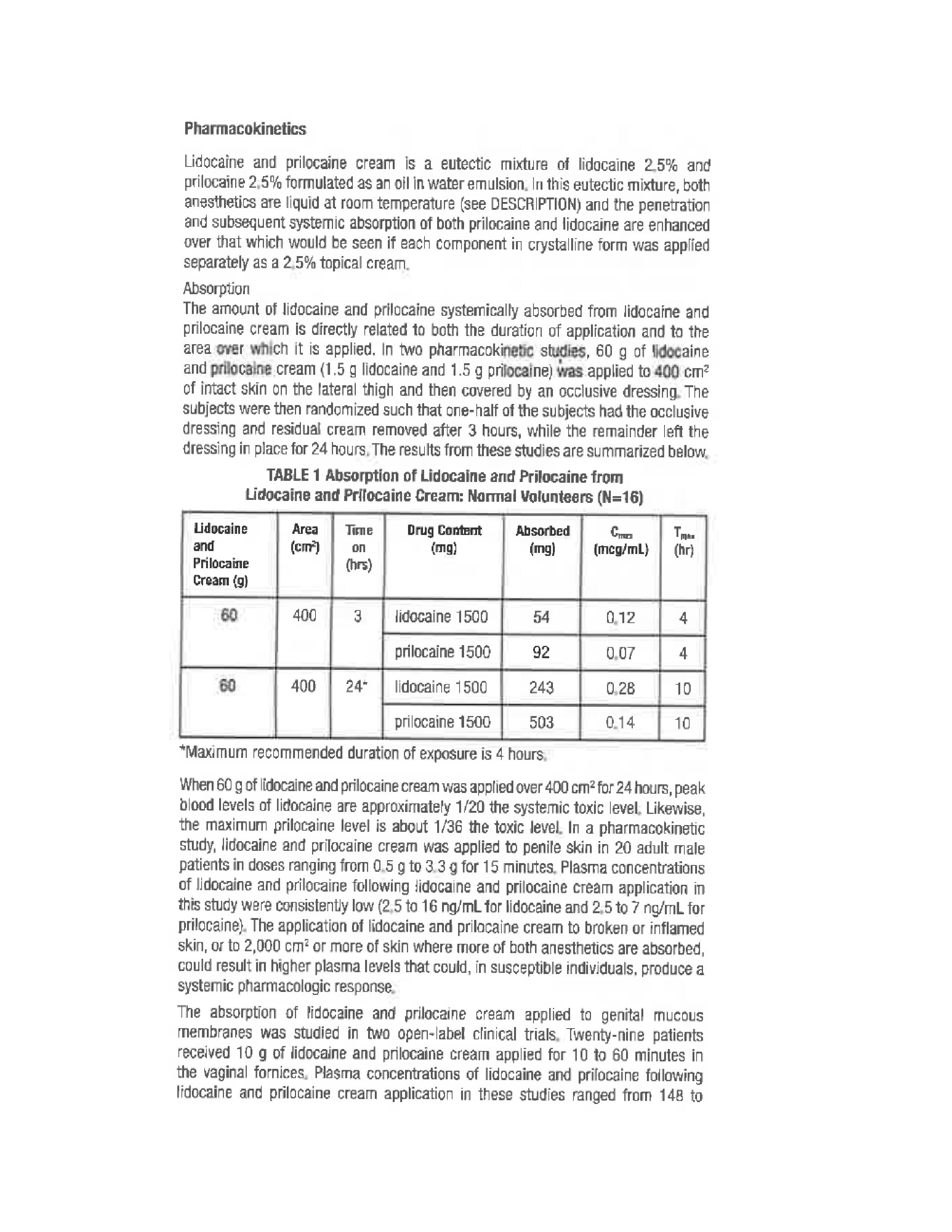
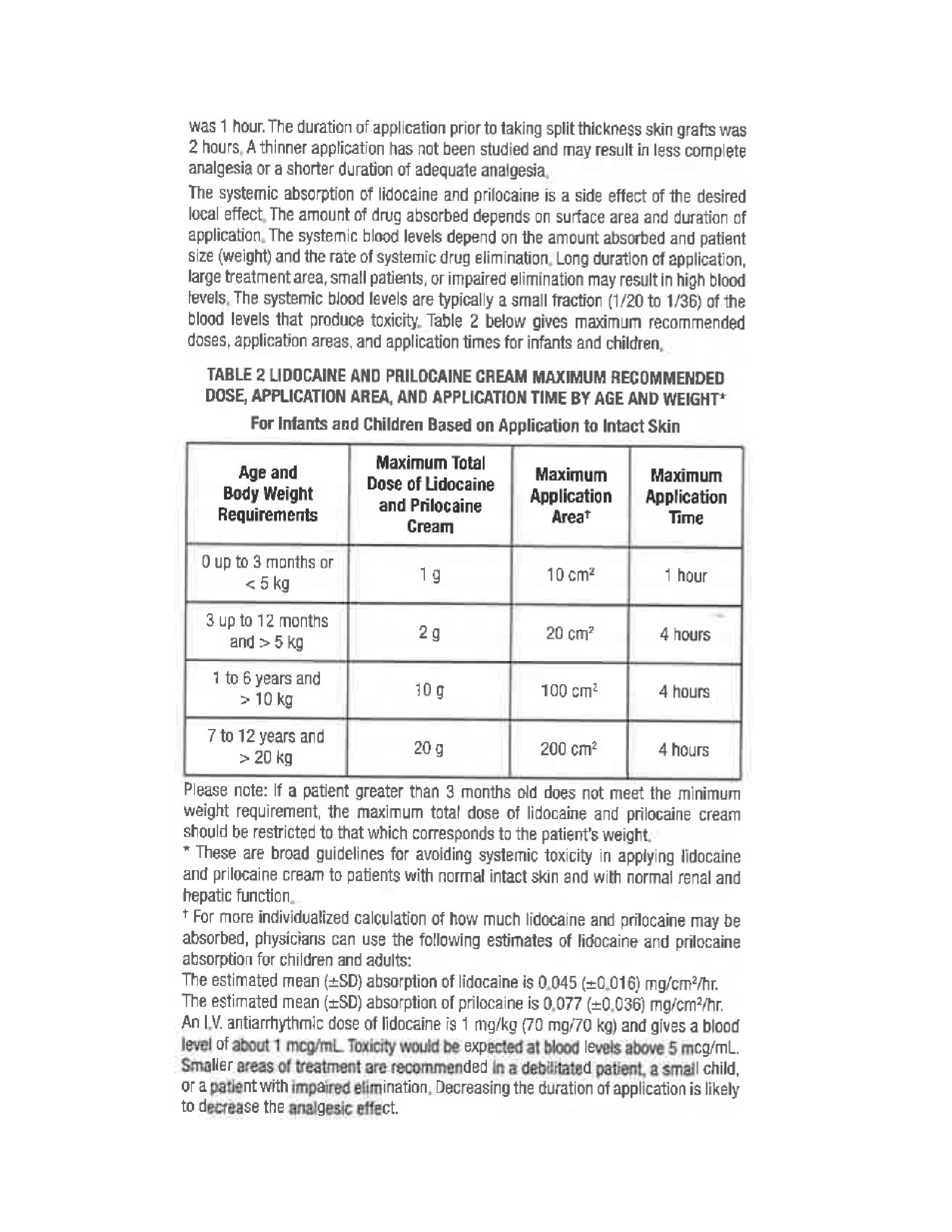
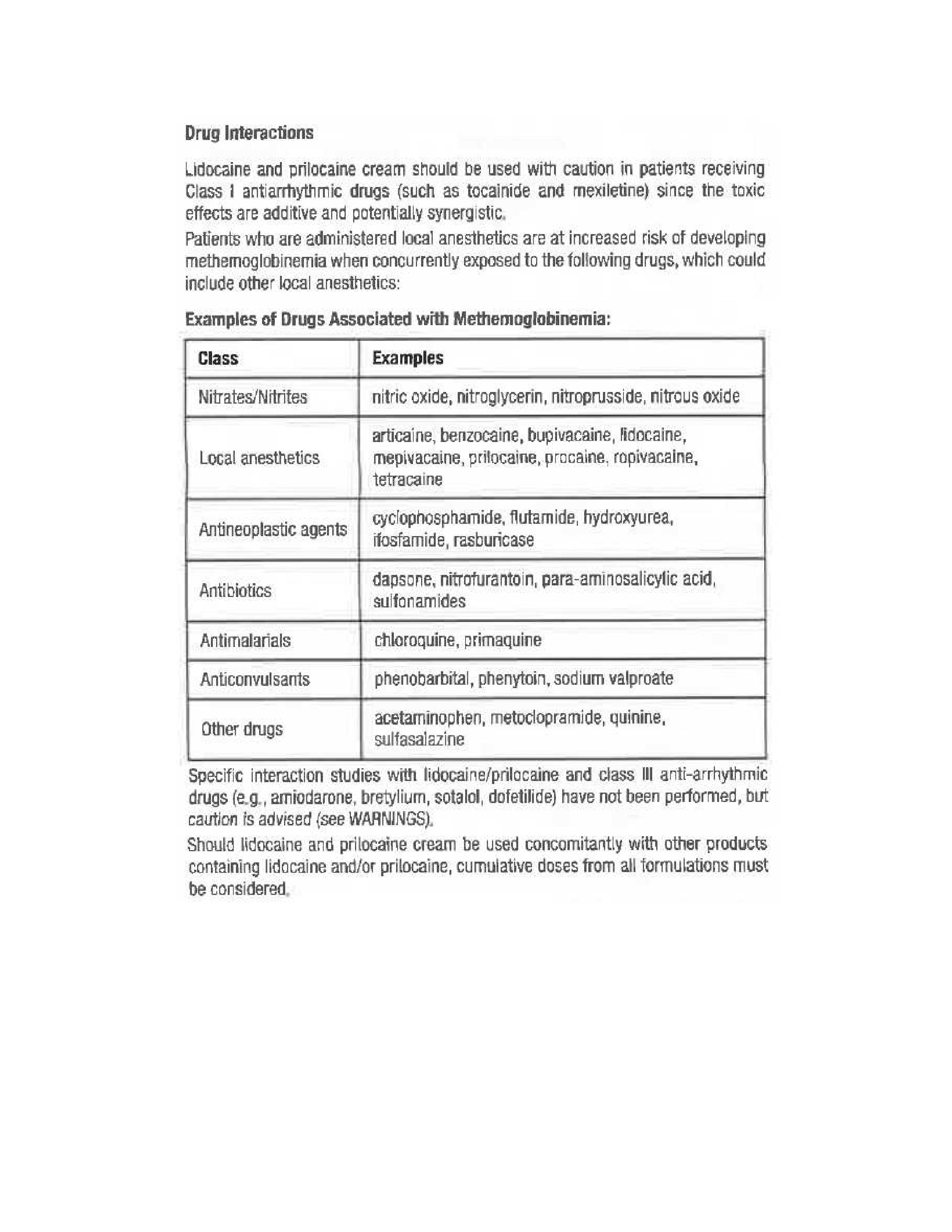
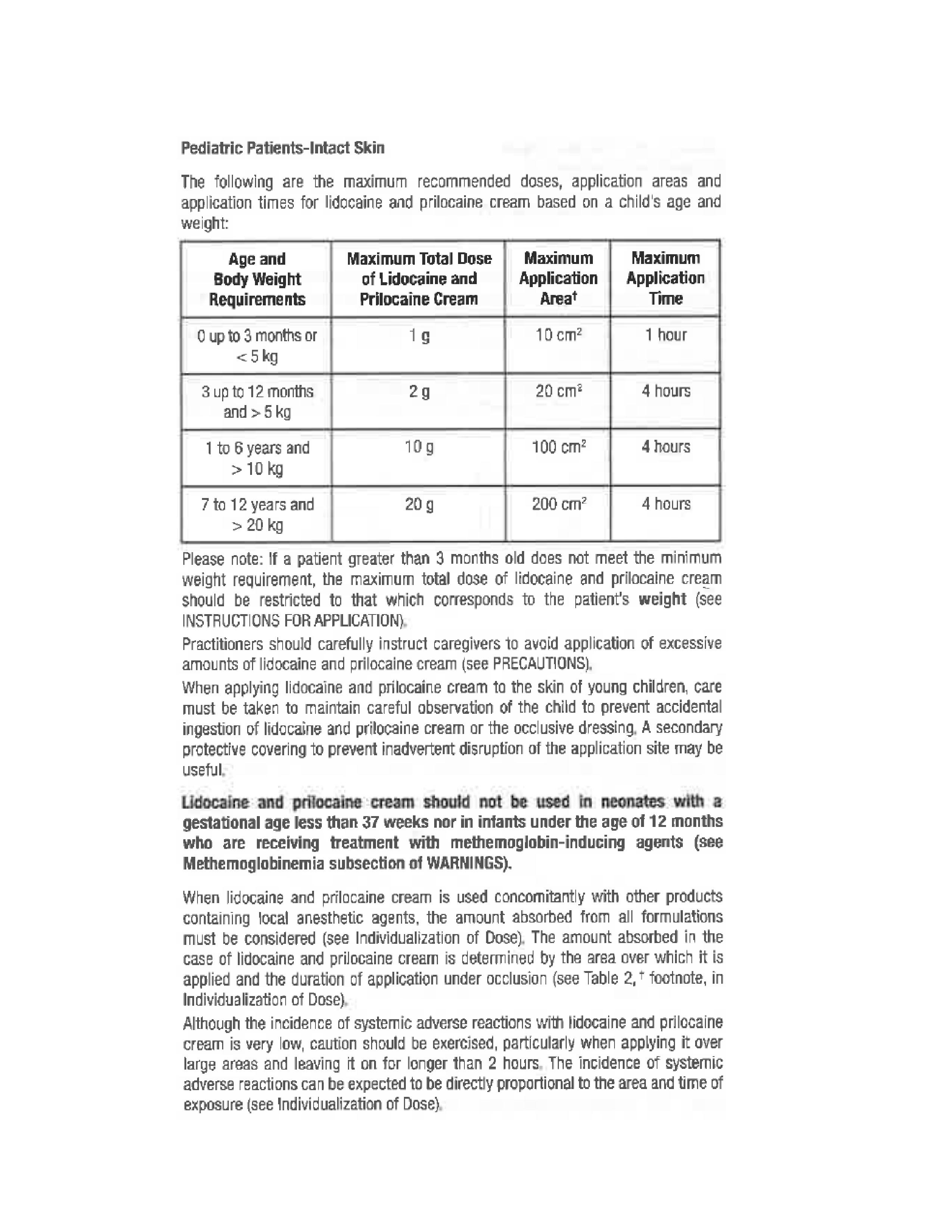
Rhodes Pharmaceuticals Lidocaine and Prilocaine Cream
Lidocaine and Prilocaine Cream by
Drug Labeling and Warnings
Lidocaine and Prilocaine Cream by is a Prescription medication manufactured, distributed, or labeled by TriRx Huntsville Pharmaceutical Services. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
LIDOCAINE AND PRILOCAINE CREAM- lidocaine and prilocaine cream cream
TriRx Huntsville Pharmaceutical Services
----------
Rhodes Pharmaceuticals Lidocaine and Prilocaine Cream
| LIDOCAINE AND PRILOCAINE CREAM
lidocaine and prilocaine cream cream |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - TriRx Huntsville Pharmaceutical Services (117090286) |
Revised: 6/2024
Document Id: 1a013e76-e5e3-56b2-e063-6394a90a5f22
Set id: 19fb6823-fcd8-7c08-e063-6294a90aa62e
Version: 2
Effective Time: 20240603
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.