NEREUS- tradipitant capsule
NEREUS by
Drug Labeling and Warnings
NEREUS by is a Prescription medication manufactured, distributed, or labeled by Vanda Pharmaceuticals Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use NEREUS safely and effectively. See full prescribing information for NEREUS.
NEREUS™ (tradipitant) capsules, for oral use
Initial U.S. Approval: 2025INDICATIONS AND USAGE
NEREUS is a substance P/neurokinin 1 (NK1) receptor antagonist indicated for the prevention of vomiting induced by motion in adults. (1)
DOSAGE AND ADMINISTRATION
- The recommended dosage of NEREUS is 85 mg or 170 mg as a single oral dose approximately 60 minutes before an event expected to cause vomiting induced by motion. The safety of NEREUS for the prevention of vomiting induced by motion in adults for more than 90 doses has not been established in clinical trials. (2.1)
- The maximum dosage in a 24-hour period is a single dose of 85 mg or 170 mg. (2.1)
- Administer on an empty stomach, at least 1 hour prior to or 2 hours after a full meal. (2.1)
DOSAGE FORMS AND STRENGTHS
Capsules: 85 mg (3)
CONTRAINDICATIONS
None. (4)
WARNINGS AND PRECAUTIONS
Effects on the Ability to Drive or Operate Machinery: May impair mental and/or physical abilities required for driving a motor vehicle or operating heavy machinery. Concomitant use of other drugs that cause central nervous system depression and strong CYP3A4 inhibitors may increase this effect. If concomitant use is unavoidable, warn patients against driving and other activities requiring complete mental alertness. (5.1, 7.1)
ADVERSE REACTIONS
Most common adverse reactions (incidence ≥5%) are: somnolence, headache, and fatigue. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Vanda Pharmaceuticals Inc. at 1-844-GO-VANDA (1-844-468-2632) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
Strong CYP3A4 Inhibitors: May increase tradipitant exposure and the risk of adverse reactions. (7.1)
USE IN SPECIFIC POPULATIONS
Lactation: Monitor breastfed infants for somnolence. (8.2)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 3/2026
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage and Administration
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Effects on the Ability to Drive or Operate Machinery
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
7 DRUG INTERACTIONS
7.1 Effects of Other Drugs on NEREUS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
8.7 Hepatic Impairment
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage and Administration
- The recommended dosage of NEREUS is 85 mg or 170 mg as a single oral dose. Use the lowest effective dose. The safety of NEREUS for the prevention of vomiting induced by motion in adults for more than 90 doses has not been established in clinical trials [see Adverse Reactions (6.1)].
- Administer NEREUS orally approximately 60 minutes before an event expected to cause vomiting induced by motion.
- The maximum dosage in a 24-hour period is a single dose of 85 mg or 170 mg.
- Administer NEREUS on an empty stomach, at least 1 hour prior to or 2 hours after a full meal [see Clinical Pharmacology (12.3)].
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Effects on the Ability to Drive or Operate Machinery
In placebo-controlled clinical trials, somnolence (6%, 12%) and fatigue (6%, 8%) were adverse reactions reported in subjects who took a single dose of 85 mg or 170 mg NEREUS, respectively [see Adverse Reactions (6.1)]. NEREUS may impair the mental and/or physical abilities required for driving a motor vehicle or operating heavy machinery. Concomitant use of other drugs that cause central nervous system depression and strong CYP3A4 inhibitors may increase this effect [see Drug Interactions (7.1)]. If concomitant use is unavoidable, warn patients against driving and other activities requiring complete mental alertness.
-
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of a single 85 mg or 170 mg dose of NEREUS was evaluated in adult subjects with a history of motion sickness in two randomized, double-blind, placebo-controlled trials, Study 1 and Study 2 [see Clinical Studies (14)]. Additional safety data for the 170 mg dose of NEREUS were obtained from a randomized, double-blind, placebo-controlled trial, Study 3 (NCT03772340).
Adverse reactions reported in at least 5% of subjects treated with a single NEREUS 85 mg or 170 mg dose and at a higher frequency than subjects who received placebo, are shown in Table 1.
Table 1: Adverse Reactionsa in Adult Subjects with a History of Motion Sickness in Single-Dose, Placebo-Controlled Studies Among Subjects Receiving NEREUS aReported in at least 5% of subjects and at a higher frequency than placebo.
bNEREUS was administered as a single 85 mg dose approximately 60 minutes prior to a boat trip and without food.
cNEREUS was administered as a single 170 mg dose approximately 60 minutes prior to a boat trip and without food.
Studies 1 and 2 Studies 1, 2, and 3 Adverse
ReactionNEREUS 85 mgb
N=227
n (%)Placebo
N=228
n (%)NEREUS 170 mgc
N=289
n (%)Placebo
N=291
n (%)Somnolence 14 (6) 9 (4) 36 (12) 17 (6) Headache 16 (7) 12 (5) 28 (10) 17 (6) Fatigue 14 (6) 6 (3) 24 (8) 6 (2) In a 12-month randomized, open-label study, 382 subjects with a history of motion sickness were instructed to administer NEREUS 85 mg (N=199) or 170 mg (N=183) as a single dose 60 minutes before a travel event of at least 60 minutes in duration expected to induce symptoms of motion sickness. Subjects were allowed to take up to 90 doses during the study. The median exposure was 18 doses and the majority of subjects (69%) did not take more than 30 doses. Most subjects (95%) did not take more than 8 doses in any 30-day period. Adverse reactions were consistent with those observed in Study 1, Study 2, and Study 3.
- 7 DRUG INTERACTIONS
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Available data from clinical trials with NEREUS use in pregnant women are insufficient to inform a drug-associated risk of major birth defects, miscarriage, or other adverse maternal or fetal outcomes.
In animal reproduction studies, no adverse developmental effects were observed with oral administration of tradipitant to pregnant rats during organogenesis through lactation or to pregnant rabbits during organogenesis at doses up to approximately 3.3 and 1.4 times the exposure to tradipitant at the maximum recommended human dose (MRHD), respectively.
The background risk of major birth defects and miscarriage for the indicated population(s) is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Data
Animal Data
In a combined fertility and embryo-fetal development study in pregnant rats, tradipitant was administered at oral doses of 10, 100, or 1000 mg/kg (approximately 1.4, 1.9, and 2.2 times the exposure to tradipitant at the MRHD) during the periods of mating and organogenesis, through gestation day 17. No adverse effects on maternal performance or embryo-fetal development were observed at any tested dose.
In an embryo-fetal development study in pregnant rabbits, tradipitant was administered at oral doses 30, 175, or 1000 mg/kg/day (approximately 0.2, 0.4, and 1.4 times the exposure to tradipitant at the MRHD) during the period of organogenesis, from gestation day 7 to 19. No adverse effects on maternal performance or embryo-fetal development were observed at any tested dose.
In a pre- and post-natal development study in pregnant rats, tradipitant was administered at oral doses of 100, 300, or 1000 mg/kg/day (approximately 2.3, 2.4, and 3.3 times the exposure to tradipitant at the MRHD) from gestation day 6 through lactation day 20. No maternal toxicity or developmental effects on the offspring were observed at any tested dose.
8.2 Lactation
Risk Summary
Lactation studies have not been conducted to assess the presence of tradipitant or its metabolites in human milk, the effects on the breastfed infant, or the effects on milk production. Tradipitant is present in rat milk (see Data). When a drug is present in animal milk, it is likely that the drug will be present in human milk. Monitor breastfed infants for somnolence. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for NEREUS and any potential adverse effects on the breastfed child from NEREUS or the underlying maternal condition.
Tradipitant was excreted in milk of lactating rats that received tradipitant at 100, 300, or 1000 mg/kg (2.3, 2.4, and 3.3 times the exposure to tradipitant at the MRHD) during pregnancy from gestation day 6 through lactation day 20. Serum concentrations of tradipitant and its major metabolites in pups were approximately 1% to 3% of maternal serum concentrations on lactation day 4 and up to 1% of those in the dams on lactation day 11.
8.4 Pediatric Use
The safety and effectiveness of NEREUS have not been established in pediatric patients.
8.5 Geriatric Use
Of the total number of NEREUS-treated subjects in controlled clinical studies for vomiting induced by motion, 69 (13%) were 65 years of age and older, while 4 (0.8%) were 75 years of age and older [see Adverse Reactions (6.1) and Clinical Studies (14)]. No overall differences in safety or effectiveness were observed between subjects 65 years of age and older and younger adult subjects, and other reported clinical experience has not identified differences in responses between geriatric and younger adult subjects, but greater sensitivity of some older individuals cannot be ruled out.
8.6 Renal Impairment
The recommended dosage of NEREUS in patients with mild and moderate renal impairment (estimated glomerular filtration rate (eGFR) of at least 30 mL/minute/1.73m2) is the same as in patients with normal renal function. Tradipitant has not been studied in subjects with severe renal impairment (eGFR ≤ 29 mL/min/1.73m2) [see Clinical Pharmacology (12.3)]. Avoid use of NEREUS in patients with severe renal impairment.
-
11 DESCRIPTION
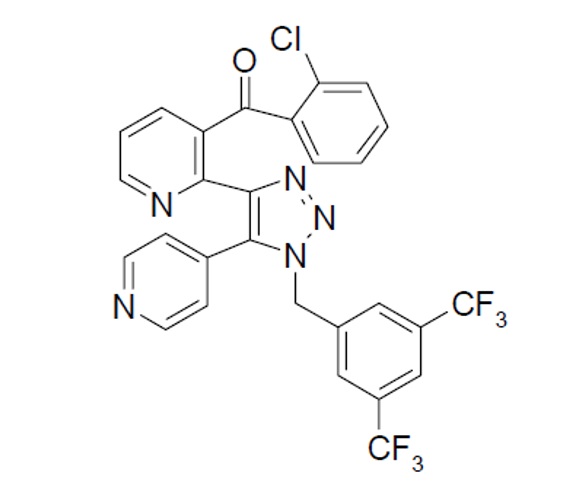
NEREUS (tradipitant) is a substance P/neurokinin-1 (NK-1) receptor antagonist designated by the chemical name, as {2-[1-(3,5-Bistrifluoromethylbenzyl)-5-pyridin-4-yl-1H-[1,2,3]triazol-4- yl]-pyridin-3-yl}-(2-chlorophenyl)-methanone, with molecular formula C28H16ClF6N5O, and molecular weight 587.9. The chemical structure for tradipitant is:
Tradipitant is a white to off-white crystalline powder. It is practically insoluble in water, soluble in methanol, and slightly soluble in isopropyl alcohol and simulated gastric fluid.
NEREUS is supplied as white opaque body capsules with “VANDA 85mg” printed in white on the black opaque cap for oral administration, each containing 85 mg tradipitant. Inactive ingredients are croscarmellose sodium, lactose monohydrate, magnesium stearate, microcrystalline cellulose, povidone, and sodium lauryl sulfate. The capsule contains FD&C blue No 1, FD&C red No 40, and FD&C yellow No 6, gelatin, and titanium dioxide.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Tradipitant is a selective, high-affinity antagonist of human substance P/neurokinin 1 (NK1) receptors. Tradipitant does not have affinity for NK2 and NK3 receptors, serotonin (5-HT3), dopamine (D2), cholinergic, or histamine (H1) receptors.
Tradipitant has been shown in animal models to inhibit drug induced emesis. Human Positron Emission Tomography (PET) studies with tradipitant have shown that tradipitant crosses the blood brain barrier and occupies brain NK-1 receptors.
The exact mechanism by which tradipitant exerts its therapeutic effect is not fully established.
12.2 Pharmacodynamics
Tradipitant inhibits the NK1 receptor with a Ki of 0.062 ± 0.01 nM and inhibits substance P (SP) induced intracellular calcium mobilization with a Kb of 0.095 ± 0.025 nM. The major metabolites of tradipitant (M2, M3, M4, and M8) showed a similar degree of binding at the NK-1 receptor.
NK1 Receptor Occupancy
A clinical study using PET imaging demonstrated a dose- and concentration-dependent increase in frontal cortex NK-1 receptor occupancy of tradipitant. The maximum receptor occupancy was 93% after multiple administrations of 100 mg NEREUS.
Cardiac Electrophysiology
At the mean maximum concentration provided by the maximum recommended single dose (170 mg) administered without food, clinically significant QTc prolongation was not observed.
12.3 Pharmacokinetics
Absorption
Absolute oral bioavailability has not been studied in humans. Following a single dose of 85 mg tradipitant in healthy subjects under fasting conditions, tradipitant geometric mean Cmax was 84.7 ng/mL, AUC 0-inf was 1839 ng*h/mL, and the median Tmax was 2.0 hours. Following a single dose of 170 mg tradipitant in healthy subjects under fasting conditions, tradipitant geometric mean Cmax was 112 ng/mL, AUC0-inf was 3,526 ng*h/mL, and the median Tmax was 1.50 hours.
Effect of food
Tradipitant Cmax and AUC0-inf increased approximately 4.7-fold and 2.4-fold, respectively, and Tmax was delayed by 2 hours, when 85 mg tradipitant was administered with a high-fat meal (800 to 1,000 calories, 50% fat) compared to fasted conditions in healthy subjects. After administration of 170 mg tradipitant with a high-fat meal, Cmax and AUC0-inf increased approximately 6.9-fold and 2.9-fold, respectively, and Tmax was delayed by 2.5 hours [see Dosage and Administration (2.1)].
Distribution
The apparent volume of distribution (Vd/F) of tradipitant is 1956 L in healthy subjects. Tradipitant plasma protein binding ranged from 96% to >99% in vitro.
Elimination
The observed mean elimination half-life for tradipitant is approximately 34 hours and apparent oral clearance of tradipitant is 41.7 L/hour in healthy subjects.
Metabolism
Tradipitant is extensively metabolized; however, the metabolic pathways have not been fully characterized. In vitro findings suggest that non-CYP450-mediated processes and CYP enzymes (CYP3A4, CYP2C19, and to a lesser extent by CYP2C8) are involved in the metabolism of tradipitant. Tradipitant is also glucuronidated by UGT1A4 and UGT2B7.
Four major metabolites (M2, M3, M4, and M8) were identified in plasma. Following a single dose of 85 mg tradipitant in healthy subjects under fasting conditions, the AUCs of M2, M3, M4, and M8 represent 40%, 33%, 43%, and 42% of the parent AUC, respectively.
Excretion
Following a single oral dose of radiolabeled tradipitant 25 mg, approximately 88% of the dose was recovered, with 80% of the dose in feces and 7% in urine. Unchanged tradipitant was minimal in feces and was not detected in urine.
Specific Populations
Patients with Renal Impairment
Based on population pharmacokinetic analysis, no clinically significant difference was observed in pharmacokinetics of tradipitant between subjects with normal renal function and subjects with mild to moderate renal impairment (estimated glomerular filtration rate (eGFR) of at least 30 mL/minute/1.73m2). The impact of severe renal impairment (eGFR ≤29 mL/min/1.73m2) on the pharmacokinetics of tradipitant is unknown [see Use in Specific Populations (8.6)].
Patients with Hepatic Impairment
Tradipitant has not been studied in subjects with any degree of hepatic impairment (Child-Pugh Class A to C). The impact of mild, moderate, or severe hepatic impairment on the pharmacokinetics of tradipitant is unknown. [see Use in Specific Populations (8.7)]
Drug Interaction Studies
Clinical Studies
Midazolam (CYP3A4 Substrate)
Co-administration of tradipitant did not result in clinically significant change in pharmacokinetics of midazolam or 1-OH-midazolam.
Ethanol
Co-administration of tradipitant with ethanol (20% w/v alcohol in orange juice (240 mL for men and 200 mL women) increased tradipitant Cmax by 9% and AUC0-24s by 14%. This increase in exposure is not considered to be clinically relevant.
In Vitro Studies
Based on in vitro studies, tradipitant does not inhibit CYP1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, nor induces CYP1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19.
Based on in vitro studies, tradipitant may inhibit and induce CYP3A4. However, an in vivo study showed no clinically significant differences in midazolam pharmacokinetics when co-administered with tradipitant.
Tradipitant is a P-gp substrate and is not a substrate of BCRP, OATP1B1, OATP1B3, MATE1 or MATE2-K. In vitro, tradipitant did not inhibit P-gp, BCRP, OATP1B1, OATP1B3, OCT2, OAT1, OAT3, MATE1, and MATE2K at clinically relevant concentrations.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
In transgenic rasH2 mice orally administered tradipitant (15, 50, or 1500 mg/kg/day) for 26 weeks, there was no increase in tumors.
In rats orally administered tradipitant (10, 30, or 150 mg/kg/day) for at least 104 weeks, a dose- related increase in the incidence of thyroid follicular cell tumors (adenomas or adenomas combined with carcinomas), correlating with the findings of thyroid masses and cysts, was observed at all tested doses in both males (less than the exposure at the MRHD) and females (1.4 times the exposure to tradipitant at the MRHD). These findings in the thyroid are likely rodent specific, secondary to the induction of hepatic metabolic enzymes and not relevant to humans.
Mutagenesis
Tradipitant was not genotoxic in a battery of in vitro (Ames, chromosomal aberration in human peripheral blood lymphocytes) and in vivo (mouse micronucleus) assays.
Impairment of Fertility
In a combined fertility and embryofetal development study, no effects were observed on fertility or reproductive performance in male or female rats administered oral doses of tradipitant at 10, 100, or 1000 mg/kg/day (up to 2.2 times the exposure to tradipitant at the MRHD) prior to and through mating, conception, and implantation.
-
14 CLINICAL STUDIES
The efficacy of NEREUS for the prevention of vomiting induced by motion was evaluated in two randomized, double-blind, placebo-controlled studies: Study 1 (NCT04327661) and Study 2 (NCT05903924). In both studies, subjects were randomized 1:1:1 to receive a single dose of NEREUS 85 mg, 170 mg, or placebo approximately 60 minutes prior to a boat trip scheduled to last approximately 2 to 5 hours. Subjects with conditions causing chronic nausea were not eligible for enrollment. Subjects were instructed to take study medication without food. Use of anti-nausea or anti-emetic medications was not allowed during the studies. The efficacy of NEREUS for the treatment of established nausea and vomiting was not evaluated in the studies.
There were 365 subjects in Study 1 and 316 in Study 2 with a mean age of 48 years (range from 18 to 75 years), 62% were female, 12% identified as Hispanic or Latino, 81% identified as White, 9% as Asian, 5% as Black or African American, and 4% identified as another racial group.
The duration of the boat trip in these studies ranged from 2 to 4.4 hours. The peak wave height ranged from 0.3 to 2.5 meters.
The primary endpoint in both studies was the percentage of subjects with vomiting during the boat trip. Vomiting was assessed every 30 minutes using a 1-item questionnaire completed by subjects indicating whether they had vomited in the last 30 minutes.
Prevention of Vomiting Endpoint Results
The percentage of subjects with vomiting during a boat trip in Studies 1 and 2, is shown in Table 2.
Table 2: Percentage of Subjects with Vomiting During a Boat Trip CI = confidence interval
aNEREUS was administered as a single 85 mg or 170 mg dose approximately 60 minutes prior to a boat trip and without food.
bThe difference (%) for NEREUS minus placebo is based on the unadjusted risk difference. The 95% CI is calculated using the Wald method.
NEREUS 85 mga NEREUS 170 mga Placebo Study 1 N=123 N=120 N=122 Incidence of
Vomiting (%)20% 18% 44% Treatment -25% -26% Difference (95%
CI)b(-36%, -14%) (-37%, -15%) Study 2 N=104 N=106 N=106 Incidence of
Vomiting (%)18% 10% 38% Treatment -19% -27% Difference (95%
CI)b(-31%, -8%) (-38%, -16%) Prevention of Nausea Endpoint Results
In Study 1 and Study 2 during a boat trip, a secondary endpoint of worst nausea score, assessed over the preceding 30 minutes on a 5-point scale where 0 indicates no nausea and 4 indicates very severe nausea, was 2.4 for NEREUS 85 mg and 2.3 for NEREUS 170 mg compared to 2.4 for placebo in Study 1 and 2.5 for NEREUS 85 mg and 2.2 for NEREUS 170 mg compared to 2.4 for placebo in Study 2. These differences did not reach statistical significance in either study.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
NEREUS (tradipitant) capsules 85 mg are supplied as white opaque body with “VANDA 85mg” printed in white on the black opaque cap and packaged as 36-count in an HDPE bottle with child-resistant cap and a 2-g desiccant pouch and as 60-count in an HDPE bottle with child resistant cap and a 2-g desiccant canister.
- NDC: 43068-585-02 Bottles of 36
- NDC: 43068-585-01 Bottles of 60
Store NEREUS capsules at controlled room temperature, 25°C (77°F); excursions permitted to 15°C to 30°C (59° - 86°F) [See USP Controlled Room Temperature].
Protect NEREUS capsules from exposure to light and moisture.
-
17 PATIENT COUNSELING INFORMATION
Effects on the Ability to Drive or Operate Machinery
Advise patients NEREUS may cause somnolence and/or fatigue and impair the mental and/or physical abilities required for driving a motor vehicle or operating heavy machinery and to avoid concomitant use of other drugs with NEREUS as it may increase this effect [see Drug Interactions (7.1)]. If concomitant use is unavoidable, warn patients against driving and other activities requiring complete mental alertness [see Warnings and Precautions (5.1)].
Administration Information
Advise the patient:
- Take NEREUS approximately 60 minutes before an event expected to cause vomiting induced by motion.
- The maximum dosage in a 24-hour period is a single dose of 85 or 170 mg.
- Administer NEREUS on an empty stomach, at least 1 hour prior to or 2 hours after a full meal [see Clinical Pharmacology (12.3)].
Lactation
Advise breastfeeding women using NEREUS to monitor infants for somnolence and to seek medical care if they notice this sign [see Use in Specific Populations (8.2)].
Distributed By:
Vanda Pharmaceuticals Inc.
Washington, D.C. 20037 USA - PRINCIPAL DISPLAY PANEL - NDC: 43068-585-02 - 85mg Tradipitant Capsules - Carton Label
- PRINCIPAL DISPLAY PANEL - NDC: 43068-585-02 - 85mg Tradipitant Capsules - Container Label
- PRINCIPAL DISPLAY PANEL - NDC: 43068-585-01 - 85mg Tradipitant Capsules - 60 Count Container Label
-
INGREDIENTS AND APPEARANCE
NEREUS
tradipitant capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 43068-585 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TRADIPITANT (UNII: NY0COC51FI) (TRADIPITANT - UNII:NY0COC51FI) TRADIPITANT 85 mg Product Characteristics Color BLACK (BLACK) , WHITE (WHITE) Score no score Shape CAPSULE (CAPSULE) Size 19mm Flavor Imprint Code VANDA;85mg Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 43068-585-02 36 in 1 BOTTLE 03/31/2026 1 1 in 1 CARTON; Type 0: Not a Combination Product 2 NDC: 43068-585-01 60 in 1 BOTTLE 03/31/2026 2 1 in 1 CARTON; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA220152 03/31/2026 Labeler - Vanda Pharmaceuticals Inc. (133501556)
Trademark Results [NEREUS]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 NEREUS 98023555 not registered Live/Pending |
Impact Immobilier Inc 2023-06-01 |
 NEREUS 97650292 not registered Live/Pending |
ASTREA UK SERVICES LIMITED 2022-10-27 |
 NEREUS 97006921 not registered Live/Pending |
Shanghai Xuchao Electronic Commerce Co.,Ltd. 2021-09-01 |
 NEREUS 87729615 5549526 Live/Registered |
SHENZHEN HOPESTAR SCI-TECH CO.,LTD. 2017-12-21 |
 NEREUS 87483173 5370054 Live/Registered |
ShenZhen L-Rider technology Co., Ltd 2017-06-09 |
 NEREUS 87476280 5369656 Live/Registered |
ShenZhen L-Rider technology Co., Ltd 2017-06-06 |
 NEREUS 85906316 4526922 Live/Registered |
QINGDAO DITRIP TYRE CO., LTD. 2013-04-17 |
 NEREUS 85221035 4129544 Dead/Cancelled |
Nereus Environmental LLC 2011-01-19 |
 NEREUS 79370712 not registered Live/Pending |
FXA Media GmbH 2023-03-15 |
 NEREUS 79338821 not registered Live/Pending |
Nereus London Holdings Limited 2022-01-17 |
 NEREUS 79054404 3643708 Dead/Cancelled |
ATLANTIS RESOURCES CORPORATION; PTE. LIMITED 2008-04-30 |
 NEREUS 78551396 not registered Dead/Abandoned |
Nereus Pharmaceuticals, Inc. 2005-01-21 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.