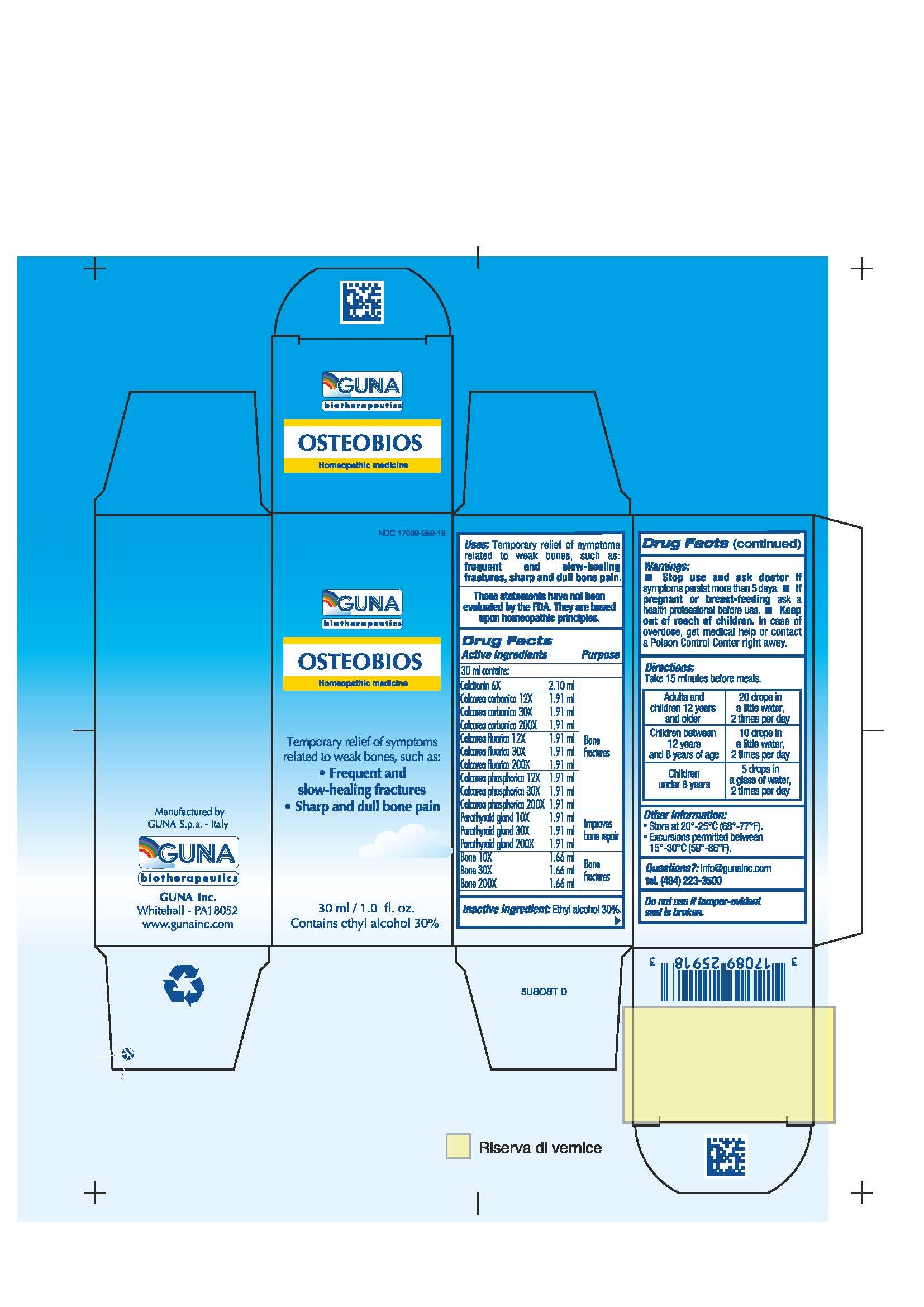
OSTEOBIOS- calcitonin human - calcium carbonate - calcium fluoride - calcium phosphate - isoleucine - leucine - lysine - methionine - phenylalanine - sus scrofa bone - sus scrofa parathyroid gland - threonine - tryptophan - valine - solution/ drops
OSTEOBIOS by
Drug Labeling and Warnings
OSTEOBIOS by is a Homeopathic medication manufactured, distributed, or labeled by Guna spa. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- ACTIVE INGREDIENTS/PURPOSE
- USES
- WARNINGS
- PREGNANCY
- WARNINGS
- DIRECTIONS
- QUESTIONS
- INACTIVE INGREDIENT
- INDICATIONS & USAGE
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
OSTEOBIOS
calcitonin human - calcium carbonate - calcium fluoride - calcium phosphate - isoleucine - leucine - lysine - methionine - phenylalanine - sus scrofa bone - sus scrofa parathyroid gland - threonine - tryptophan - valine - solution/ dropsProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 17089-259 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CALCITONIN HUMAN (UNII: I0IO929019) (CALCITONIN HUMAN - UNII:I0IO929019) CALCITONIN HUMAN 6 [hp_X] in 30 mL ISOLEUCINE (UNII: 04Y7590D77) (ISOLEUCINE - UNII:04Y7590D77) ISOLEUCINE 3 [hp_X] in 30 mL LEUCINE (UNII: GMW67QNF9C) (LEUCINE - UNII:GMW67QNF9C) LEUCINE 3 [hp_X] in 30 mL LYSINE (UNII: K3Z4F929H6) (LYSINE - UNII:K3Z4F929H6) LYSINE 3 [hp_X] in 30 mL METHIONINE (UNII: AE28F7PNPL) (METHIONINE - UNII:AE28F7PNPL) METHIONINE 3 [hp_X] in 30 mL PHENYLALANINE (UNII: 47E5O17Y3R) (PHENYLALANINE - UNII:47E5O17Y3R) PHENYLALANINE 3 [hp_X] in 30 mL THREONINE (UNII: 2ZD004190S) (THREONINE - UNII:2ZD004190S) THREONINE 3 [hp_X] in 30 mL TRYPTOPHAN (UNII: 8DUH1N11BX) (TRYPTOPHAN - UNII:8DUH1N11BX) TRYPTOPHAN 3 [hp_X] in 30 mL VALINE (UNII: HG18B9YRS7) (VALINE - UNII:HG18B9YRS7) VALINE 3 [hp_X] in 30 mL CALCIUM CARBONATE (UNII: H0G9379FGK) (CALCIUM CATION - UNII:2M83C4R6ZB, CARBONATE ION - UNII:7UJQ5OPE7D) CALCIUM CARBONATE 30 [hp_X] in 30 mL CALCIUM FLUORIDE (UNII: O3B55K4YKI) (FLUORIDE ION - UNII:Q80VPU408O) CALCIUM FLUORIDE 30 [hp_X] in 30 mL CALCIUM PHOSPHATE (UNII: 97Z1WI3NDX) (CALCIUM CATION - UNII:2M83C4R6ZB) CALCIUM PHOSPHATE 30 [hp_X] in 30 mL SUS SCROFA BONE (UNII: 597RLJ09RG) (SUS SCROFA BONE - UNII:597RLJ09RG) SUS SCROFA BONE 30 [hp_X] in 30 mL SUS SCROFA PARATHYROID GLAND (UNII: 2KBE35NE8S) (SUS SCROFA PARATHYROID GLAND - UNII:2KBE35NE8S) SUS SCROFA PARATHYROID GLAND 30 [hp_X] in 30 mL Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) 9 mL in 30 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 17089-259-18 1 in 1 BOX 05/01/2018 1 30 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 05/23/2006 Labeler - Guna spa (430538264) Establishment Name Address ID/FEI Business Operations Guna spa 338587646 manufacture(17089-259)
Trademark Results [OSTEOBIOS]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 OSTEOBIOS 78902198 3352224 Live/Registered |
GUNA, INC. 2006-06-06 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.