DALBA VEGAN ESSENCE SUNSCREEN- homosalate, octocrylene, ethylhexyl methoxycinnamate, ethylhexyl salicylate cream
dAlba Vegan Essence Sunscreen by
Drug Labeling and Warnings
dAlba Vegan Essence Sunscreen by is a Otc medication manufactured, distributed, or labeled by ADMTMAX LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- General Precautions
- WARNINGS AND PRECAUTIONS
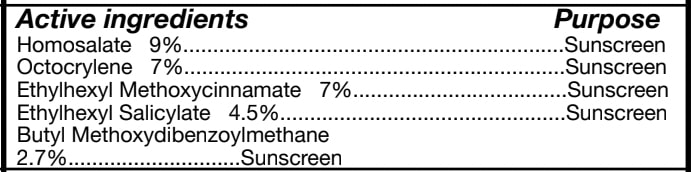
- ACTIVE INGREDIENT
- INACTIVE INGREDIENT
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
- DOSAGE & ADMINISTRATION
- WARNINGS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
DALBA VEGAN ESSENCE SUNSCREEN
homosalate, octocrylene, ethylhexyl methoxycinnamate, ethylhexyl salicylate creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 84662-020 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 9 mg in 50 mL Inactive Ingredients Ingredient Name Strength 1,3-DIMETHYLBUTYL SALICYLATE (UNII: 47OL39K42M) Product Characteristics Color Score Shape FREEFORM Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 84662-020-01 50 mL in 1 TUBE; Type 0: Not a Combination Product 09/16/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 08/27/2024 Labeler - ADMTMAX LLC (101826554)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.